
Concept explainers
The sketch of the graph for the given function and to explain the trends in the graph.
Answer to Problem 4.1P
The graph for the function
Explanation of Solution
Given:
The Planck distribution function for blackbody
Calculation:
Consider the term,
The value of the term is calculated as,
Consider the term,
The value of the term is calculated as,
Thus, the Planck's distribution function of a blackbody radiation for a fixed temperature
The following table shows the Planck's intensity distribution for different wavelengths at a fixed temperature.
| Wavelength | |
The following figure shows the Planck's intensity distribution for different wavelengths at a fixed temperature.
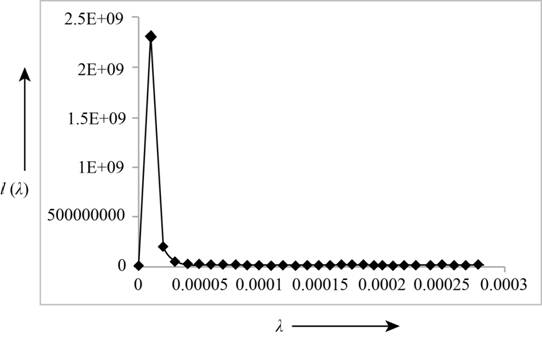
Figure (1)
From the graph, it can be concluded that the intensity of blackbody radiation for a fixed temperature, is maximum at a particular wavelength and it becomes zero with the increase in wavelength. With the increase in temperature, the maximum intensity of a radiation shift towards the shorter wavelengths.
Conclusion:
Therefore, the graph for the function
Want to see more full solutions like this?
Chapter 4 Solutions
Modern Physics For Scientists And Engineers
- Solar radiation falls on Earth's surface at a rate of 1900 W/m². Assuming that the radiation has an average wavelength of 580 nm, how many photons per square meter per second fall on the surfaces? The speed of light is 3 × 10° m/s and Planck's constant is 6.62607 × 10-34 J. s. Answer in units of photon/m² · s. 2arrow_forwardA photon in a laboratory experiment has anenergy of 4.1 eV.What is the frequency of this photon?Planck’s constant is 6.63 × 10−34 J · s.Answer in units of Hz.arrow_forwardAn X-ray photon with a wavelength of 0.999 nmnm strikes a surface. The emitted electron has a kinetic energy of 990 eV. What is the binding energy of the electron in kJ/molkJ/mol? [Note that KEKE = 12mv212mv2 and 1 electron volt (eVeV) = 1.602×10−19J1.602×10−19J.] Express your answer using three significant figures.arrow_forward
- An infrared satellite measures outgoing radiation that leaves Earth's surface through an atmospheric window. The observed spectral irradiance at a wavelength of 10 μm is 2.199×107 W m-2 m-1. What is the temperature of the surface? Give your answer in K.arrow_forwardProblem-1: An asteroid is hurtling toward earth at 150,000“. The temperature of the asteroid is about 100 K, meaning that its peak emission is 2 = 29 µm. The speed of light is c = 3E[8]. a) What is the wavelength of light that we receive from the asteroid? (Answer: 2.89855E[-05] m)arrow_forwardA log in the fire is glowing red (λ = 629 nm). What is the temperature of the log, in kelvin?arrow_forward
- In an experiment on the photoelectric effect, a metal is illuminated by visible light of different wavelengths. A photoelectron has a maximum kinetic energy of 0.9 eV when red light of wavelength 640 nm is used. With blue light of wavelength 420 nm, the maximum kinetic energy of the photoelectron is 1.9 eV. Use this information to calculate an experimental value for the Planck constant h. [arrow_forwardThe photoelectric equation for the kinetic energy of a photoelectron is, following Einstein, E < hf – W, whereh is Planck's constant, f is the frequency of the light, and W is the work-function. Sodium has W = 3.2×10-19 J. When sodium is illuminated by monochromatic light of a particular frequency, electrons are emitted with speeds up to 8 x 105 ms-1. a) Calculate the wavelength of the light. b) Calculate the stopping potential.arrow_forwardThe photoelectric equation for the kinetic energy of a photoelectron is, following Einstein, E < hf – W, where h is Planck's constant, f is the frequency of the light, and W is the work-function. Sodium has W = 3.2×10-19 J. When sodium is illuminated by monochromatic light of a particular frequency, electrons are emitted with speeds up to 8 x 105 ms-1. a) Calculate the wavelength of the light. b) Calculate the stopping potential.arrow_forward
- The peak wavelength of radiation emitted by a black body at a temperature of 2000 K is 1.45 μm. If the peak wavelength of emitted radiation changes to 2.90 μm, then the temperature (in K) of the black body isarrow_forwardThrough what potential difference ΔVΔV must electrons be accelerated (from rest) so that they will have the same wavelength as an x-ray of wavelength 0.130 nmnm? Use 6.626×10−34 J⋅sJ⋅s for Planck's constant, 9.109×10−31 kgkg for the mass of an electron, and 1.602×10−19 CC for the charge on an electron. Express your answer using three significant figures. =89.0 V Through what potential difference ΔVΔV must electrons be accelerated so they will have the same energy as the x-ray in Part A? Use 6.626×10−34 J⋅sJ⋅s for Planck's constant, 3.00×108 m/sm/s for the speed of light in a vacuum, and 1.602×10−19 CC for the charge on an electron. Express your answer using three significant figures. Second question is what I need help on! Thanks!arrow_forwardPART A: A metal surface is illuminated with photons with a frequency f=1.5×10^15 Hz. The stopping potential for electrons photoemitted from the surface is 3.6 V. What is the work function of the metal? Answer= 2.6 eV PART B: A certain metal has a work function ϕ. What is the maximum photon wavelength that will produce photoemission? Express your answer in terms of ϕ,Planck's constant h, and the speed of light c. *Please answer Part B*arrow_forward
 College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

