
(a)
Interpretation:
The complete and detailed mechanism of the given reaction is to be drawn, and the product is to be predicted.
Concept introduction:
There are two locations in an
Answer to Problem 18.6P
The product of given reaction is
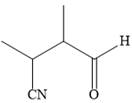
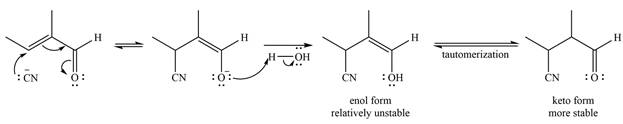
The mechanism is drawn as
Explanation of Solution
Even though
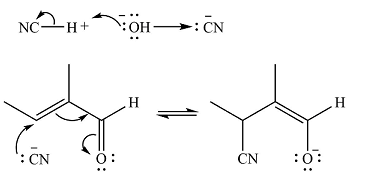
The negatively charged oxygen extracts a proton from the solvent molecule (
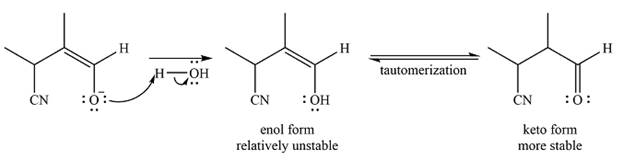
The enol form being relatively unstable rapidly tautomerizes to the more stable keto form of the product.
Thus, the final product of the reaction is
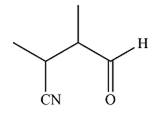
And the complete mechanism can be drawn as
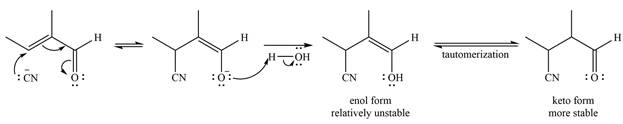
The mechanism and the product of the given reaction were determined on the basis of reversible addition of cyanide ion to the
(b)
Interpretation:
The complete and detailed mechanism of the given reaction is to be drawn, and the product is to be predicted.
Concept introduction:
There are two locations in an
Answer to Problem 18.6P
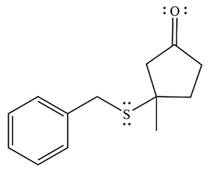
The product of the given reaction is
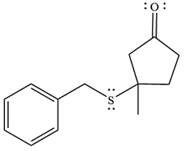
The mechanism is drawn as
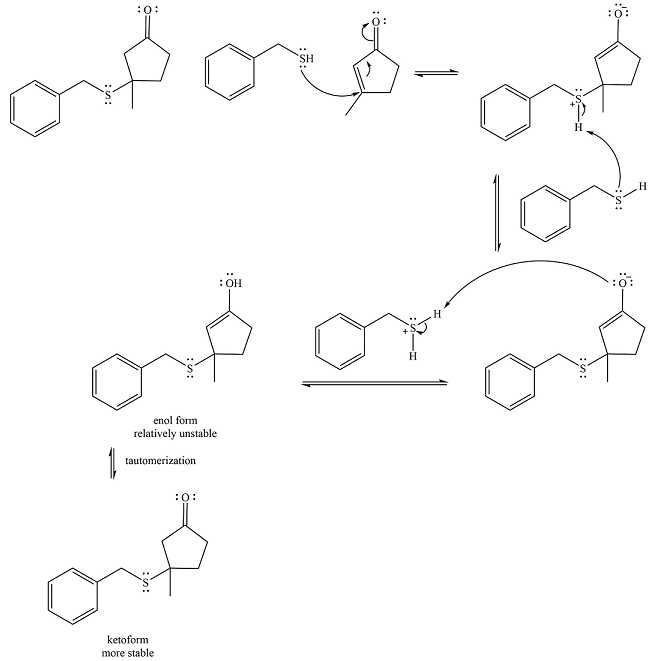
Explanation of Solution
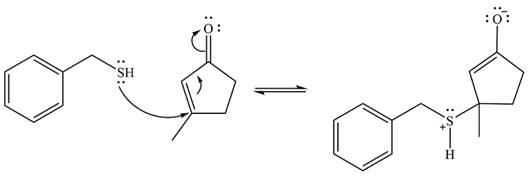
Two proton transfer steps follow and produce an uncharged enol. The enol form is relatively unstable and rapidly tautomerizes to the more stable keto form.
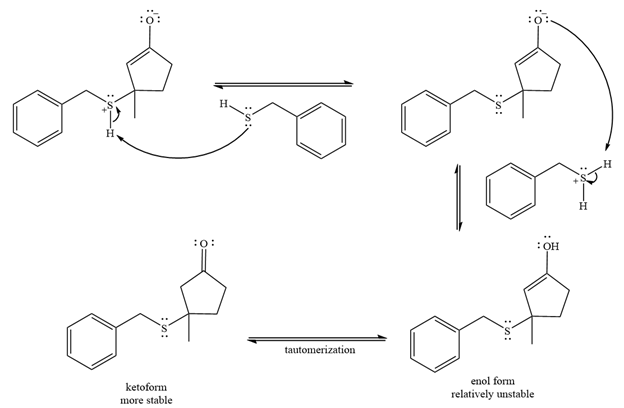
Thus, the final product of the reaction is
And the complete mechanism can be drawn as

The mechanism and the product of the given reaction were determined on the basis of reversible addition of the uncharged nucleophile to the
(c)
Interpretation:
The complete and detailed mechanism of the given reaction is to be drawn and the product is to be predicted.
Concept introduction:
There are two locations in an
Answer to Problem 18.6P
The product of given reaction is
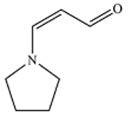
The mechanism of the reaction is
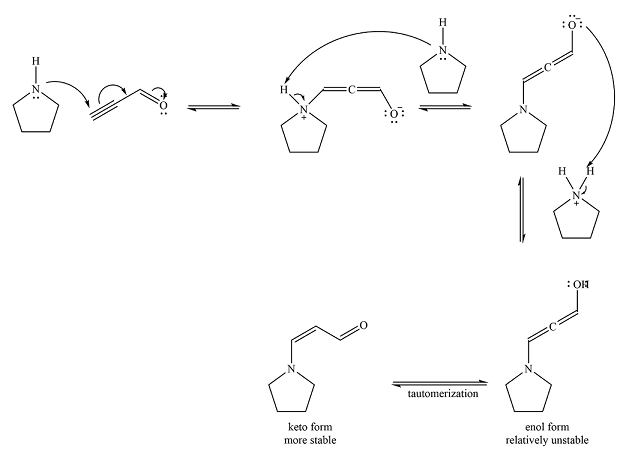
Explanation of Solution
Pyrrolidine is uncharged nucleophile and favors conjugate addition to an
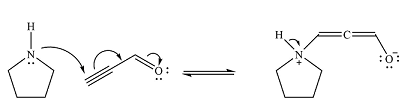
After the nucleophilic addition, two proton transfer steps occur to produce an uncharged enol. The enol form is relatively unstable and rapidly tautomerizes to the more stable keto form.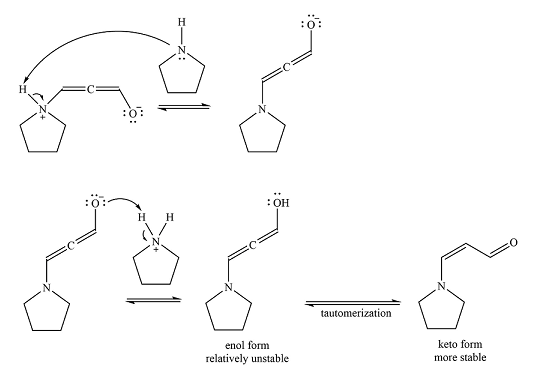
Thus, the final product of the reaction is

And the complete mechanism of the reaction can be drawn as

The mechanism and the product of the given reaction were determined on the basis of reversible addition of the uncharged nucleophile to the
Want to see more full solutions like this?
Chapter 18 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Draw the complete, detailed mechanism and the products for each of the following reactions.arrow_forwardDraw the complete, detailed mechanism for the reaction shown here. Will the product be optically active? Explain.arrow_forwardDraw the complete, detailed mechanism for the reaction shown here and predict the major product. CH3ONA Explain. DMSOarrow_forward
- Draw the complete, detailed mechanism for the reaction shown here and give the major product. CH3I (excess) ? NH2arrow_forwardDraw a complete, detailed mechanism for this reaction.arrow_forwardPredict the major product of each of the reactions shown here and provide the complete, detailed mechanism.arrow_forward
- The reaction shown here is a halosulfonation, which is a useful variation of the sulfonation reaction. Draw the complete mechanism for this reaction.arrow_forwardDraw a complete, detailed mechanism for the following reaction. A key intermediate is provided.arrow_forwardPropose a mechanism for the reaction shown here, which takes place under conditions that favor anarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





