
Concept explainers
(a)
Interpretation:
The condensed formula of
Concept Introduction:
Condensed structural formula: Condensed structural formula shows the arrangement of atoms in grouped form.
(a)
Answer to Problem 11A.11E
The condensed formula of
Explanation of Solution
The given compound is,
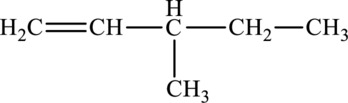
The parent chain of the compound is pentane. One methyl group is present in the carbon third position and a double bond is found in carbon first position. Hence, the systematic name of the compound is
(b)
Interpretation:
The condensed formula of
Concept Introduction:
Refer to part (a).
(b)
Answer to Problem 11A.11E
The condensed formula of
Explanation of Solution
The given compound is,
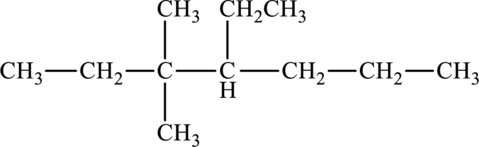
The parent chain of the compound is heptane. One ethyl group is present in the carbon fourth position and two methyl groups are present in carbon third position. Hence, the systematic name of the compound is
(c)
Interpretation:
The condensed formula of
Concept Introduction:
Refer to part (a).
(c)
Answer to Problem 11A.11E
The condensed formula of
Explanation of Solution
The given compound is,
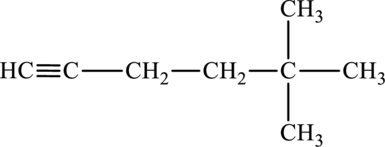
The parent chain of the compound is hexane. Two methyl groups are present in carbon fifth position and a triple bond is seen at the carbon first position. Hence, the systematic name of the compound is
(d)
Interpretation:
The condensed formula of
Concept Introduction:
Refer to part (a).
(d)
Answer to Problem 11A.11E
The condensed formula of
Explanation of Solution
The given compound is,
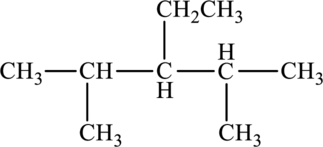
The parent chain of the compound is pentane. One ethyl group is present in the carbon third position and two methyl groups are present in carbon second and fourth position respectively.
Hence, the systematic name of the compound is
Want to see more full solutions like this?
Chapter 11 Solutions
Chemical Principles: The Quest for Insight
- Draw the product resulting from mild oxidation of (a) 2-butanol; (b) 2-methylpropanal; (c) cyclopentanol.arrow_forwardDraw the organic product formed when the following compounds undergo a substitution reaction: (a) acetic acid and 1-hexanol; (b) propanoic acid and dimethyl-amine; (c) ethanoic acid and diethylamine.arrow_forwardWrite a complete chemical equation showing reactants, products, and catalysts needed (if any) for the following reaction and (2) Draw and name the organic compound found in every reaction.(Use condensed structural formula) (a) Complete hydrogenation of 2-Methylhexa-1,5-diene (b) Complete halogenation (Br2) of 3-Ethyl-2,2-dimethylhept-3-ene(c) Reaction of (4E)-2,4-Dimethylhexa-1,4-diene with a mole of waterarrow_forward
- Illustrate the chemical structural formula for 3-methyl-3-ethylpentane. (b) Identify its chemical family as an isomer. (c) Provide the balanced chemical reaction equation for the combustion of one mole of this fuel with an equivalence ratio of ϕ=0.735arrow_forwardWrite the condensed structural formula for each of thefollowing compounds: (a) 2-ethyl-1-hexanol, (b) methylphenyl ketone, (c) para-bromobenzoic acid, (d) butyl ethylether, (e) N,N-dimethylbenzamide.arrow_forwardDraw complete structural diagrams of the following compounds. (a) 2-methyl,pent-2-ene (b) propyne (c) 2,3-dimethyl butane (d) cycloheptanearrow_forward
- Write the condensed formulas of the following names of organic compounds. (a). 4-methyl-3,6-octadiene (b). N-ethyl-N-methyl-3,4-dimethylaniline (c). 4-cyclopropyl-3-methylcyclopentanalarrow_forwardGive the molecular formula of a hydrocarbon containingfive carbon atoms that is (a) an alkane, (b) a cycloalkane,(c) an alkene, (d) an alkyne.arrow_forwardWhat product is formed from the hydrogenation of2-methylpropene? (a) propane, (b) butane, (c) 2-methylbutane,(d) 2-methylpropane, (e) 2-methylpropyne.arrow_forward
- Write the condensed formulas of the following names of organic compounds. (a). 4-methyl-3,6-octadiene (b). N-ethyl-N-methyl-3,4-dimethylanilinearrow_forwardDraw the organic product formed when the following compounds undergo a substitution reaction: (a) acetic acid and methyl-amine; (b) butanoic acid and 2-propanol; (c) Formica acid and 2-methyl-1-propanol.arrow_forwardDraw the organic product formed when the following compounds undergo a substitution reaction: (a) acetic acid and methylamine; (b) butanoic acid and 2-propanol; (c) formic acid and 2-methyl-1-propanol.arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
