
Concept explainers
(a)
Interpretation:
Among the following compounds the compound which reacts faster in SN2 reaction is to be identified.
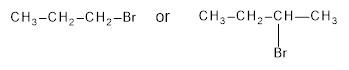
Concept Introduction:
Primary substitution, unless there is a steric hindrance in the
(b)
Interpretation:
Among the following compounds the compound which reacts faster in E1 reaction is to be identified.
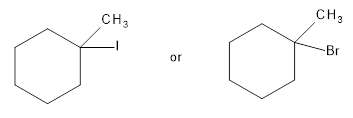
Concept Introduction:
For an E1/E2 reaction, when same alkyl group is present with the leaving group, the relative reactivity of alkyl halide is,
(c)
Interpretation:
Among the following which reacts faster in SN1 reaction is to be identified.
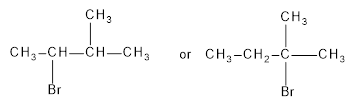
Concept Introduction:
Secondary alkyl halide do not undergo SN1 reaction, tertiary can undergo both substitution and elimination product, and substitution is mostly favored.
Trending nowThis is a popular solution!

Chapter 9 Solutions
Organic Chemistry (8th Edition)
- a. which reaction is faster ? b. complete the enerqy diagram c. labei the diagram by drawing the structures of the Starting a. NaSEt NaOEt Vs. Br Br reactionarrow_forwardWhat starting materials are needed to prepare each alkene using a Heckreaction?arrow_forwardWhich is an energy diagram for a concerted reaction (SN2 and E2)? A B C Darrow_forward
- Draw the product formed when A is treated with each series of reagents.a. [1] H2O; [2] NaH; [3] CH3Brb. [1] CuCN; [2] DIBAL-H; [3] H2O c. [1] C6H5NH2; [2] CH3COClarrow_forwardHow do you identify if a reaction is Sn1 Sn2 E1 or E2? Draw a flowchart to identify if reactions are SN1 SN2 E1 or E2.arrow_forwardDraw the product of each reaction (d).arrow_forward
- Draw the products formed in each Wittig reaction. Draw the major stereoisomer when applicable.arrow_forwardDraw a reaction coordinate diagram for a reaction in which a. the product is thermodynamically unstable and kinetically unstable. b. the product is thermodynamically unstable and kinetically stable.arrow_forward16. Draw in the curved arrows for the SN2 reaction shown. CI s 17. Draw in the curved arrows for the E2 reaction shown. (CH3)3COK (CH3)3COH Oll ·xarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





