
Concept explainers
(a)
Interpretation:
cis isomer should be drawn for the given trans isomer.
Concept Introduction:
The arrangement of groups attached to the carbo atoms involved in a
Cis Isomer: The two groups attached on same side of
In cis isomer the hydrogen atoms are on the same side of the two carbon atom.
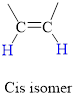
Trans isomer: The two groups are attached on opposite sides of the carbon-carbon double bond;.
The hydrogen atoms are on the opposite side of the two carbon atom.
(b)
Interpretation:
trans isomer should be drawn for the given cis isomer.
Concept Introduction:
The arrangement of groups attached to the carbo atoms involved in a
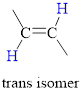
Cis Isomer: The two groups attached on same side of
In cis isomer the hydrogen atoms are on the same side of the two carbon atom.

Trans isomer: The two groups are attached on opposite sides of the carbon-carbon double bond;.
The hydrogen atoms are on the opposite side of the two carbon atom.

Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
Chemistry & Chemical Reactivity
- Draw as many isomers as you can for C4H7Cl using bond-line structuresarrow_forwardIsomers are different compounds that have the same molecular formula. If the atoms are connected in different ways, they are called constitutional or structural isomers. Geometric isomers are a type of isomer where the order of the atoms in the two compounds is the same but their arrangement in space is different. The most common types of geometric isomers are cis- and trans- isomers. PART 5: Constitutional/Structural Isomers Draw (using any method you wish) and give the IUPAC name for any four isomers with the molecular formula C6H12Brz. Structure: - Br CH₂ сна CH₂CH CH CH₂ CH₂ Br. Name: bromo-3 methyl peature Name: Structure: Structure: Name: Structure: comors Name:arrow_forwarda.Give all the structural isomers of the compound C3H5Cl which contains one C=C bond. (Hint: 3 isomers) b.One of the structural isomers in above question shows cis-trans isomerism. Draw and name this pair of isomers.arrow_forward
- What is the total number of single bonds, double bonds, and triple bonds in the best Lewis structure of C2H2? 0 single, 2 double, 1 triple 2 single, 1 double, 0 triple 3 single, 0 double, 0 triple 1 single, 2 double, 0 triple 2 single, 0 double, 1 triplearrow_forwardThere are at least three different molecules with the formula C3H8O. Draw a Lewis structure for each possible constitutional isomer. Be careful not to duplicate any structures.arrow_forwardIs this compound a cis or trans isomer? name the isomerarrow_forward
- Draw the Lewis structure of acetic acid, CH3CO2H, clearly indicating all non-bonding pairs of electrons. Even though nitrogen and phosphorus have the same number of valence electrons, nitrogen can only bond to chlorine four times forming NCl4+ but phosphorus can bond with chlorine five times forming PCl5. Explain. Draw all the isomers of C4H9Br, using bond-line formulas. Draw the Lewis structure of the nitrite ion, NO2–, clearly indicating resonance contributors as well as non-bonding pairs of electrons and formal charges, as relevant. Nitromethane is a polar molecule but contains 2 equal polar covalent bonds. Briefly explain why and draw a relevant 3-dimensional structure to show the overall dipole moment of the molecule. Draw all isomers of C6H12O that are aldehydes.arrow_forwardThere are seven possible dichloropropene isomers (molecular formula C3H4Cl2). Draw their structures and name each isomer. (Hint: Don't over-look cis-trans isomers.)arrow_forwardA triple bonded carbon can only bond with one other atom. O True O Falsearrow_forward
- If you rotate the hydrogen atoms to a different position in a a model of C2H6 by turning about the central C—C bond, do you get different structural (or constitutional) isomers of C2H5Cl?arrow_forwardUnshared, or lone, electron pairs play an important role in determining the chermical and physical properties of organic compounds. Thus, it is important to know which atoms carry unshared pairs. Use the structural formulas below to determine the number of unshared pairs at each designated atom. Be sure your answers are consistent with the formal charges on the formulas. The number of unshared pairs at atom a is The number of unshared pairs at atom b is HyC CH The number of unshared pairs at atom e is The number of unshared pairs at atom a is The number of unshared pairs at atom b is HC CH The number of unshared pairs at atom e isarrow_forwardStructural Isomers: In the next part you will learn a little about structural isomers and how to draw them. Structural isomers are compounds with the same molecular formula, but different bonding arrangements or connectivities. In other words, the atoms are connected in a different order. For example, there are 2 different compounds with the molecular formula C4H10. They are given below: H H-C- H -H H H HHH H- H H-C -H Η ΗΗ H H Do you see how the atoms are bonded in a different order in the 2 structures? So they are structural isomers. C3H12 Find three different molecular structures, isomers, with this formula. (Hint: Remember the C rule where every C atom needs to have 4 bonds.) C2H60 Find two different molecules with this formula. (Hint: What are the 2 ways you can C5H10 How many structural isomers can you find for this formula? (Hint: Notice that 2 H atoms have been removed. This means that there is EITHER 1 double bond OR there is a ring structure.) Try to provide at least 4…arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning



