
Human Physiology: An Integrated Approach (8th Edition)
8th Edition
ISBN: 9780134605197
Author: Dee Unglaub Silverthorn
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8.3, Problem 7CC
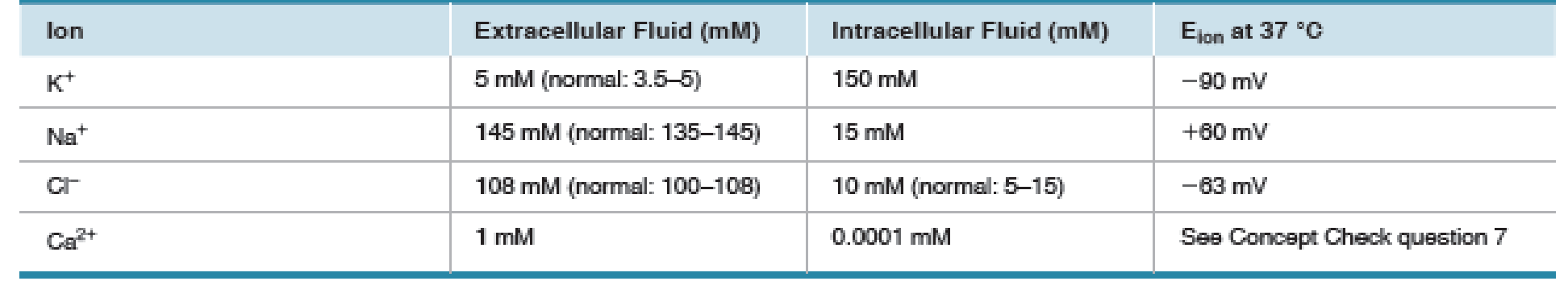
Given the values in Table 8.2, use the Nernst equation to calculate the equilibrium potential for Ca2+. Express the concentrations as powers of 10 and use your knowledge of logarithms [p. A-00] to try the calculations without a calculator.
TABLE 8.2 Ion Concentrations and Equilibrium Potentials
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Given that the extracellular concentration of Cl- is approximately 120 mM, what is the intracellular concentration if the Nernst
potential for Cl- is 39 mV. (T=298 K, R=1.987 cal/K·mol, F=23,062 cal/mol·V)
Calculate the free energy of transport for the movement of potassium by the sodium/potassium pump under normal physiological conditions: 4 mM serum potassium, 135 mM intracellular potassium, 37.1 °C, and resting potential -82 mV. Express your answer in kJ/mol. Show all work.
Calculate the free energy of transport for the movement of potassium by the sodium/potassium pump under disturbed conditions of 2 mM serum potassium. Assume all other parameters remain the same. Express your answer in kJ/mol. Show all work.
What factors could limit the continued action of the sodium/potassium pump when only 2 mM potassium is present in the blood plasma? Note that under normal physiological conditions, the cell interior contains 11 mM sodium and the blood contains 140 mM sodium.
Table Q1(a) shows typical values for the intracellular and extracellular
concentrations of the major ion species (in millimoles per litre) for frog skeletal
muscle.
Table Q1(a)
Permeability (cm/s)
2 x 10-8
2х 10
Ions
Intracellular
Extracellular
Na*
12
145
K+
155
4
4
120
4 x 106
By referring to Table Q1(a), compute the equilibrium resting potential for this
membrane by assuming the room temperature is 20° C. Given the Boltzman's
constant, k = 1.38 x 1023 J/K and an electronic charge, q = 1.602 x 10-19 C.
Chapter 8 Solutions
Human Physiology: An Integrated Approach (8th Edition)
Ch. 8.1 - Organize the following terms describing functional...Ch. 8.2 - Where do neurohormone-secreting neurons terminate?Ch. 8.2 - What is the difference between a nerve and a...Ch. 8.2 - Draw a chain of three neurons that synapse on one...Ch. 8.2 - What is the primary function of each of the...Ch. 8.2 - Name the two glial cell types that form myelin....Ch. 8.3 - Given the values in Table 8.2, use the Nernst...Ch. 8.3 - Would a cell with a resting membrane potential of...Ch. 8.3 - Would the cell membrane depolarize or...Ch. 8.3 - Match each ions movement with the type of graded...
Ch. 8.3 - Prob. 11CCCh. 8.3 - What is the difference between conductance and...Ch. 8.3 - If you put ouabain, an inhibitor of the Na+-K+...Ch. 8.3 - The pyrethrin insecticides, derived from...Ch. 8.3 - When Na+ channel gates are resetting, is the...Ch. 8.3 - A stimulating electrode placed halfway down an...Ch. 8.3 - Place the following neurons in order of their...Ch. 8.4 - Prob. 18CCCh. 8.4 - Prob. 19CCCh. 8.4 - Prob. 20CCCh. 8.4 - Prob. 21CCCh. 8.4 - Prob. 22CCCh. 8.4 - Classify the H+-neurotransmitter exchange as...Ch. 8.4 - Prob. 24CCCh. 8.4 - Prob. 25CCCh. 8.4 - Is Na+-dependent neurotransmitter reuptake...Ch. 8.5 - In Figure 8.24e, assume the postsynaptic neuron...Ch. 8.5 - In the graphs of Figure 8.24a, b, why doesnt the...Ch. 8.5 - Prob. 29CCCh. 8.5 - Prob. 30CCCh. 8 - List the three functional classes of neurons, and...Ch. 8 - Somatic motor neurons control __________, and...Ch. 8 - Prob. 3RQCh. 8 - Prob. 4RQCh. 8 - Prob. 5RQCh. 8 - Prob. 6RQCh. 8 - Axonal transport refers to the (a) release of...Ch. 8 - Match the numbers of the appropriate...Ch. 8 - Arrange the following events in the proper...Ch. 8 - List the four major types of ion channels found in...Ch. 8 - Prob. 11RQCh. 8 - An action potential is (circle all correct...Ch. 8 - Choose from the following ions to fill in the...Ch. 8 - What is the myelin sheath?Ch. 8 - List two factors that enhance conduction speed.Ch. 8 - Prob. 16RQCh. 8 - Draw and label a graph of an action potential....Ch. 8 - Prob. 18RQCh. 8 - Prob. 19RQCh. 8 - Create a map showing the organization of the...Ch. 8 - Prob. 21RQCh. 8 - Prob. 22RQCh. 8 - Prob. 23RQCh. 8 - Prob. 24RQCh. 8 - The presence of myelin allows an axon to (choose...Ch. 8 - Define, compare, and contrast the following...Ch. 8 - Prob. 27RQCh. 8 - Prob. 28RQCh. 8 - Prob. 29RQCh. 8 - Prob. 30RQCh. 8 - An unmyelinated axon has a much greater...Ch. 8 - The GHK equation is sometimes abbreviated to...Ch. 8 - In each of the following scenarios, will an action...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Using the Goldman-Hodgkin-Katz equation, determine what happens to the resting membrane potential if the ECF K+ concentration doubles to 10 mM.arrow_forward(a) Table Q1(a) shows typical values for the intracellular and extracellular concentrations of the major ion species (in millimoles per litre) for frog skeletal Q1 muscle. Table Q1(a) Permeability (cm/s) 2 x 10-8 2 x 106 4 x 10-6 Ions Intracellular Extracellular Na* 12 145 K* 155 4 4 120 By referring to Table Q1(a), compute the equilibrium resting potential for this membrane by assuming the room temperature is 20° C. Given the Boltzman's constant, k= 1.38 x 1023 J/K and an electronic charge, q = 1.602 x 1019 C.arrow_forwardAt the peak of the action potential, Vm is approximately -65 mV. Assuming normal intracellular and extracellular K+ concentrations (refer to the table), (1) calculate the driving force (in mV) that acts on K+ ions and (2) use the information obtained in part 1 to determine the direction in which K+ ions will flow (i.e., into the cell or out of cell)arrow_forward
- Which factors determine the resting membrane potential? Explain why the resting membrane potential is never really the value calculate by the Nernst equation for the potassium equilibrium potential. comments : please answer me in a correct way to hence my knowledge and for my better understandingarrow_forward1. Given the following concentrations: K* Na* ICF 140 mM 14 mM 10 mM ECF 5 mM 145 mM 140 mM Calculate the equilibrium potentials of K, Na and Cl using the Nernst equation. EK = ENa = ECi =arrow_forwardA cell has an actual membrane potential (Em) at rest of -75mV. The equilibrium potential for Na+ is +120mV and the equilibrium potential for K+ is -95mV. Calculate the net driving force for Na+ in mV.arrow_forward
- With regard to Na+ and K+ equilibrium potentials and the resting and active membrane potentials, write down (a- D the directions of the forces indicated, acting on the ion in the table below under the respective condition(s). NB the examples given. lon Condition Electrical/Chemical force Direction of force Chemical e.g....inward.. ****** Na ENa+ = + 60 mV Electrical a. Chemical e.g. ...outward.. EK+ =- 90 mV Electrical b. Chemical C. Na" Emp = 0 mV Electrical d. Chemical K+ Emp =-20 mV Electricalarrow_forwardHow long would it take to reduce 1 mole of each of the following ions using the current indicated? Assume the voltage is sufficient to perform the reduction.(a) Al3+, 1.234 A(b) Ca2+, 22.2 A(c) Cr5+, 37.45 A(d) Au3+, 3.57 Aarrow_forwardThe ion flows across neuronal membranes at rest and duringan action potential do not significantly change bulk ionconcentrations, except for that of Ca2+ ions. Resting Ca2+ ionconcentrations in cells are usually about 10–7 M, and Ca2+ ionsexert physiological effects at concentrations of perhaps 10–5 M.Explain why relative changes of intracellular [Ca2+] are muchgreater than for, say, [Na+] (12–50 mM).arrow_forward
- Consider a suspension of particles (isoelectric point is at pH 6) in water at pH 2 and a NaCl concentration of 0.001 M. Describe how the strength of repulsion varies with the following changes, assuming all other conditions remain constant. Give a description (more than just increase or decrease) in terms of the effect on the double layer thickness and the zeta potential. (a) Change from 0.001 M NaCl to 0.1 M NaCl, (b) Change from pH = 2 to pH = 5.arrow_forwardThe resting membrane potential (RMP) is the voltage measured in a cell during resting state. It is particularly critical for the proper functioning highly excitable cells such as neurons and skeletal muscle cells. Identify the major ionic player then describe how it gives rise to and maintains RMP.arrow_forwardCalculate the equilibrium membrane potentials to be expected across a membrane at 37 ∘C, with a NaCl concentration of 0.50 M on the "right side" and 0.08 M on the "left side", given the following conditions. In each case, state which side is (+) and which is (−). Membrane equally permeable to both ions.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning

Human Physiology: From Cells to Systems (MindTap ...
Biology
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Cengage Learning

Enzyme Kinetics; Author: MIT OpenCourseWare;https://www.youtube.com/watch?v=FXWZr3mscUo;License: Standard Youtube License