
Concept explainers
How many
How many yield 2,3-dimethylbutane?
How many yield methylcyclobutane?
Interpretation:
The number of alkenes which are required to produce the given alkanes on catalytic hydrogenation is to be determined.
Concept introduction:
On catalytic hydrogenation, alkenes get converted to the corresponding alkanes with the same number of carbon atoms.
In hydrogenation reaction, one hydrogen atom gets attached to each of the double bonded carbon atoms.
To accelerate the rate of hydrogenation, the metal catalyst provides an alternative pathway involving low activation energy steps.
The alkane that is formed contains two hydrogen atoms more than the corresponding alkene and these two hydrogen atoms are added on the adjacent carbon atoms in the alkene.
Answer to Problem 25P
Solution:
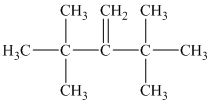
a) Only one alkene can produce
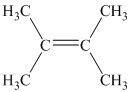
b) Two alkenes can produce
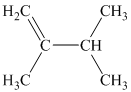
c) Three alkenes can produce methylcyclobutane upon catalytic hydrogenation.
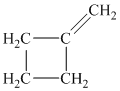
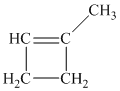
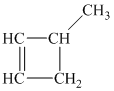
Explanation of Solution
a) The structure of
Hydrogenation of alkenes adds one hydrogen atom on each carbon atom of the double bonded carbon atoms. So in the alkane, these two carbon atoms (previously double bonded) should be attached to at least one hydrogen atom each.
In
The alkane structure is symmetric, and carbon atoms
b) The structure of
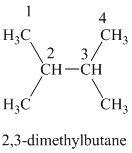
The hydrogen of alkenes adds one hydrogen atom on each carbon atom of the double bonded carbon atoms. So in the alkane, these two carbon atoms (previously double bonded) should be attached to at least one hydrogen atom each.
In
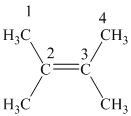
The
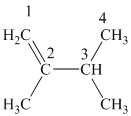
The alkane structure is symmetric, and the carbon atoms
Hence, two alkenes can produce
c) The structure of methylcyclobutane is shown below:
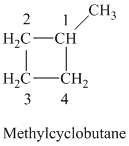
The hydrogen of alkenes adds one hydrogen atom on each carbon atom of the double bonded carbon atoms. So in the alkane, these two carbon atoms (previously double bonded) should be attached to one at least one hydrogen atom each.
In methylcyclobutane, the
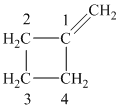
The
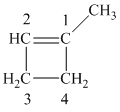
The
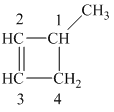
The alkane structure is symmetric. This means, there cannot be any other distinct alkene structure which would produce methylcyclobutane upon catalytic hydrogenation.
Hence, three alkenes can produce methylcyclobutane upon catalytic hydrogenation.
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry - Standalone book
- Draw the alkene that would react with the reagent given to account for the product formed. ? + HCI CH3 CH3CCH3 CI • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. #[ ] در ChemDoodlearrow_forwardMatch each alkene to its heat of hydrogenation.Alkenes: 3-methylbut-1-ene, 2-methylbut-1-ene, 2-methylbut-2-ene?H° (hydrogenation) kJ/mol: –119, –127, –112arrow_forwardDraw the alkene that would react with the reagent given to account for the product formed. ? + H₂O H₂SO4 CH3 CH3CCH3 OH You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. Sn [F ChemDoodleⓇarrow_forward
- a cyclation? 7. Draw the structure of the product(s) for the following AICI, catalyzed reactions; 3-chloro-2,2-dimethlbutane + isopropylbenzene CI benzene + 1,4-dichlorobutane benzene + a six carbon acid chloridearrow_forwardIdentify two alkenes that react with HBr to form 1-bromo-1-methylcyclohexane without undergoing a carbocation rearrangement.arrow_forwardWhich represents an efficient synthetic route to go from an alkane to an alkene? O elimination with NaNH2, followed by a water workup O anti-Markovnikov hydrohalogenation, followed by elimination O radical bromination, followed by elimination O hydration, followed by elimination O hydration, followed by ozonolysis of the double bondarrow_forward
- Draw the structure for an alkene that gives the following reaction product. CH3 HCI CH3CHCH2CHCH3 CI • Ignore alkene stereochemistry. •You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. P. opy aste ChemDoodle"arrow_forwardDraw the alkene that would react with the reagent given to account for the product formed. ? + HCI My 3 You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. CH3 CH₂ CHOCH3 TT CI CH3 L ▼ {n [F ? ChemDoodleⓇarrow_forwardDraw the organic product that is expected to form when the following compound is oxidized under biological conditions. oxidation SH reduction • You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. • If no reaction occurs, draw the organic starting material.arrow_forward
- Draw the cycloalkene that would react with the reagent given to account for the product formed. ? + H₂O H₂SO4 CH3 ▼ • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. » [ ]# CH3 ? OHarrow_forward2. Explain the selectivity of the following reaction, which produces a single product despite both alkene carbons being equally substituted. H3C CF3 HBr CF3 H3C CF3 H3C Br- H3C only product -H CF3arrow_forwardReview Topics] [References] Draw a structural formula for the major product of the reaction shown. CH;CH2 c=CHCH3 CH;CH2 Br2 H20 • Show product stereochemistry IF the reactant alkene has both carbons of the double bond within a ring. • Do not show stereochemistry in other cases. • If the reaction produces a racemic mixture, just draw one stereoisomer. Previous Next ChemDoodle Save and Ex tv ill I 11 МacBook Air DII DD F12 F11 F10 80 888 F9 F7 FB F6 F5 F4 F3 * & $ %3D 5 6. 7 8 9. 4 { P E R T Y H J K F + * COarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





