
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7.4, Problem 7.5P
Give the systematic (IUPAC) names of the following
- a. CH2=CH—CH2—CH(CH3)2
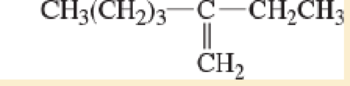
- b. CH2=CH-CH2-CH=CH2
- c. CH2=C=CH—CH=CH2
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Name each alkane.
a. CH3-CH-CH3
ČH;
CH;
CH3
b. CH3-CH-CH2-CH-CH2
ČH3
CH, CH,
c. CH3-C-C-CH3
ČH, ČH3
CH3
CH,
CH2
d. CH3-CH-CH2-CH-CH-CH2-CH2-CH3
ČH3
. Give the IUPAC names of the following compounds.
1. CH2=CH-CH,–CH(CH3)2
2. CH2=CH–CH2-CH=CH2
3. CH;(CH2);-C–CH,CH3
5.
CH2
4.
CH2=C=CH-CH=CH2
Name each alkyne.
CH,-C=C-CH,
CH3
b. CH;-C=C-C-CH,-CH,
CH3
c. CH=C-CH-CH2-CH2-CH,
CH-CH,
ČH,
Chapter 7 Solutions
Organic Chemistry (9th Edition)
Ch. 7.3A - Prob. 7.1PCh. 7.3A - Prob. 7.2PCh. 7.3B - Draw five more compounds of formula C4H6NOC1.Ch. 7.3B - For each of the following molecular formulas,...Ch. 7.4 - Give the systematic (IUPAC) names of the following...Ch. 7.5B - The following names are all incorrect. Draw the...Ch. 7.5B - Prob. 7.8PCh. 7.5B - a. How many stereogcmc double bonds are in...Ch. 7.6 - Teflon-coated frying pans routinely endure...Ch. 7.7B - Prob. 7.11P
Ch. 7.8B - Use the data in Table7-2 to predict the energy...Ch. 7.8C - Prob. 7.13PCh. 7.8E - Explain why each of the following alkenes is...Ch. 7.8F - Prob. 7.15PCh. 7.10 - Prob. 7.16PCh. 7.10A - SN1 substitution and E1 elimination frequently...Ch. 7.10C - Prob. 7.18PCh. 7.10C - Prob. 7.19PCh. 7.10C - Prob. 7.20PCh. 7.11 - Prob. 7.21PCh. 7.11 - Prob. 7.22PCh. 7.12 - Prob. 7.23PCh. 7.12 - Prob. 7.24PCh. 7.13 - Prob. 7.25PCh. 7.14B - Prob. 7.26PCh. 7.14B - Make models of the blowing compounds, and predict...Ch. 7.15 - Prob. 7.28PCh. 7.15 - Prob. 7.29PCh. 7.15 - Prob. 7.30PCh. 7.15 - Prob. 7.31PCh. 7.16 - Predict the major and minor elimination products...Ch. 7.17B - Predict the products and mechanisms of the...Ch. 7.18 - Propose mechanisms for the following reactions.Ch. 7.18 - Prob. 7.35PCh. 7.19B - The dehydrogenation of butane to trans-but-2-ene...Ch. 7.19B - Prob. 7.37PCh. 7.19B - Prob. 7.38PCh. 7.19B - Prob. 7.39PCh. 7 - Prob. 7.40SPCh. 7 - Prob. 7.41SPCh. 7 - Prob. 7.42SPCh. 7 - Prob. 7.43SPCh. 7 - Prob. 7.44SPCh. 7 - Prob. 7.45SPCh. 7 - Prob. 7.46SPCh. 7 - The energy difference between cis- and...Ch. 7 - Prob. 7.48SPCh. 7 - Prob. 7.49SPCh. 7 - Prob. 7.50SPCh. 7 - What halides would undergo E2 dehydrohalogenation...Ch. 7 - Prob. 7.52SPCh. 7 - Prob. 7.53SPCh. 7 - Write a balanced equation for each reaction,...Ch. 7 - Prob. 7.55SPCh. 7 - Using cyclohexane as your starting material, show...Ch. 7 - Show how you would prepare cyclopentene from each...Ch. 7 - Prob. 7.58SPCh. 7 - E1 eliminations of alkyl halides are rarely useful...Ch. 7 - Prob. 7.60SPCh. 7 - Propose mechanisms for the following reactions....Ch. 7 - Prob. 7.62SPCh. 7 - Prob. 7.63SPCh. 7 - Prob. 7.64SPCh. 7 - Prob. 7.65SPCh. 7 - Prob. 7.66SPCh. 7 - Prob. 7.67SPCh. 7 - Prob. 7.68SPCh. 7 - Prob. 7.69SPCh. 7 - Explain the dramatic difference in rotational...Ch. 7 - One of the following dichloronorbornanes undergoes...Ch. 7 - A graduate student wanted to make...Ch. 7 - Prob. 7.73SPCh. 7 - Prob. 7.74SPCh. 7 - Prob. 7.75SPCh. 7 - Prob. 7.76SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach (4th Edition)
Fully developed conditions are known to exist for water flowing through a 25-nim-diameer tube at 0.01 kg/s and ...
Fundamentals of Heat and Mass Transfer
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Describe the orbitals used in bonding and the bond angles in the following compounds: a. CH3O b. CO2 c. H2CO d....
Organic Chemistry (8th Edition)
Consider a sample of ideal gas initially in a volume V at temperature T and pressure P. Does the entropy of thi...
General Chemistry: Principles and Modern Applications (11th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 17 of 59 > Name the alkene. CH2 H2 .C. H3C .C. CH3 H2 name: about us I privacy policy terms of use contact careersarrow_forwardName each of the following cyclic alkanes, and indicate the formula of the compound. CHCH3 а. CH3 CH3 | CCH3 b. CH3 CH3 с. СHз СH-CH-CH3 CH3arrow_forwardUsing the IUPAC system, name the following alkanes: A. CH3-CH2-CH-CH2-CH3 | CH3 B. CH3-CH-CH2-CH2-CH3 | CH2 | CH3 CH3 CH3 | | C. CH3-C-CH2-C-CH3 | | CH3 CH3arrow_forward
- Name the alkene. CH2 H2 .C. CH3 H3C H2 name:arrow_forwardGeneral Chemistry 1 Name the follouing alkenes and alkynes, 3. CH=CCH,CH=CH, 6. 10 er babn Shemcel en CH, CH, 4. CH,CCH,C=CH 7. CH,C=CCHCHCH, CH, CH, 8. CH,=CHCH,CH,CH,C=CH 5. CH,CH,CCH,CH, CH,arrow_forward5. Give the IUPAC NAME of the following compound: H2C=CH(CH2)2CH=CH2 б. 6. Give the COMMON NAME of the following compound CH3 CH3-C-CH2-CH3 CH3arrow_forward
- 8. Name each alkyne. A. CH3CH2CH2C=CH B. CH3CH2CH2C=CCH3arrow_forwardP. CH3-CH2-CH3 Q. CH3-CH2-CH=CH-CH3 R. CH3-CH2-CH2-CH2-CH3 S. CH3-CH=CH-CH2-CH3 Which compounds belong to the same homologous series? P and Q Q and R R and S P and R Which structures show unsaturated hydrocarbons? P and Q P and R Q and S R and Sarrow_forward46. Name the following alkenes. CH,CH3 CH2=CHCH,CHCH,CHCH3 1. CH3 , CH3 CH2=CHCH2CH3 CH2=CHCH,CHCH3 a Carrow_forward
- Part II - Isomerism 1. Octane, C8H18, has 18 different constitutional or chain isomers. One of them, isooctane, is used as a standard in determining the octane rating of gasoline a. Draw the structural formulas for at least ten chain isomers of octane. b. Give the IUPAC name of each. c. Which of the isomers that you have drawn has the highest boiling point? Which has the lowest boiling point? Rationalize. 2. Which of the following structural formulas represent identical compounds and which represent constitutional/structural isomers? Identical compounds: Constitutional isomers: CH3 i). CH3-C-CI ČH3 a). CH3CH2CHCH3 e). CH2CH2CHCH3 CI CI CI CH,CI b). CH3-C-CH3 f). CH3CH2CH2CH,CI j). CICH2 CI CH3 g). CICH,CHCH3 CH2CI k). CH3-CH-CH3 CI c). CH3CHCHCH3 CH2CH3 1). CH3CHCI h). CH3CHCH2CH2CI CI d). -CIarrow_forwardPart II - Isomerism 1. Octane, C8H18, has 18 different constitutional or chain isomers. One of them, isooctane, is used as a standard in determining the octane rating of gasoline a. Draw the structural formulas for at least ten chain isomers of octane. b. Give the IUPAC name of each. c. Which of the isomers that you have drawn has the highest boiling point? Which has the lowest boiling point? Rationalize.arrow_forward3. Name the following alkenes and alkynes. HC-CH-C=C-CH-CH--CH3 CH-CH H H H H. H C= C C C-H H. H.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License