
Concept explainers
Write structural formulas for each of the following:
Vinylcycloheptane
Interpretation:
The structural formula for each of the given compounds is to be written.
Concept introduction:
When writing the structural formula of any compound, first the functional group from the suffix of the given name is identified.
The longest carbon chain containing the functional group is located.
The carbon atoms of the chain are numbered in a such way that the functional group is at the lowest numbered carbon atom.
Substituents are attached to the parent chain according to their positions given in the name.
In alkenes, the Z isomers have the higher ranked substituents on the same side of the double bond, and in E isomers, higher ranked substituents are on opposite sides of the double bond.
Answer to Problem 31P
Solution:
Explanation of Solution
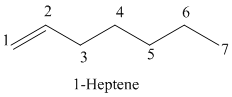
a)
The parent chain has seven carbon atoms since the name has the word “hept.” The functional group is alkene. The double bond is present between
The structural formula is as follows:
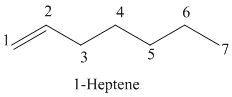
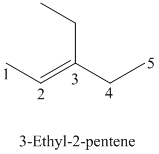
b)
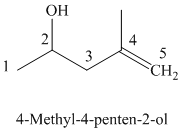
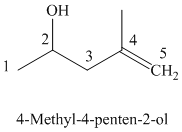
The parent chain has five carbon atoms since the name has the word “pent.” The functional group is alkene. The double bond is present between
The structural formula is as follows:
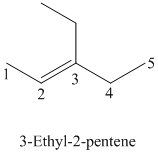
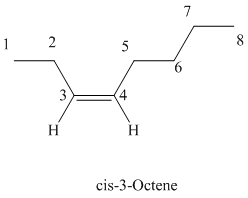
c)
The parent chain has eight carbon atoms since the name has the word “oct.” The functional group is alkene. The double bond is present between
The structural formula is as follows:
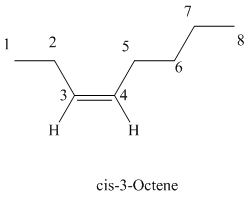
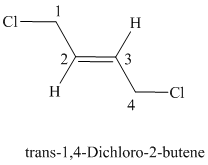
d)
The parent chain has four carbon atoms since the name has the word “but.” The functional group is alkene. The double bond is present between
The structural formula is as follows:
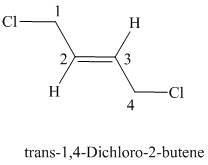
e)
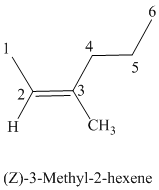
The parent chain has six carbon atoms since the name has the word “hex.” The functional group is alkene. The double bond is present between
The higher ranked substituents attached to the double bonded carbon atoms must be on the same side of the double bond since the conformation is Z. The structural formula is as follows:
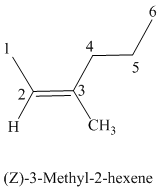
f)
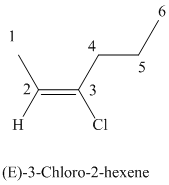
The parent chain has six carbon atoms since the name has the word “hex.” The functional group is alkene. The double bond is present between
The higher ranked substituents attached to the double bonded carbon atoms must be on the opposite side of the double bond since the conformation is E.
The structural formula is as follows:
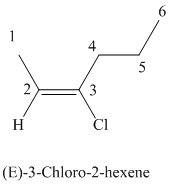
g)
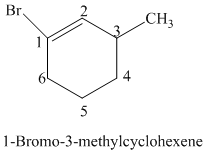
The parent is a six carbon atom ring since the name has the word “cyclohex.” The functional group is alkene. The double bond is present between
The structural formula is as follows:
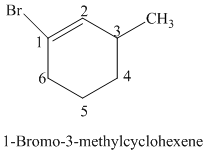
h)
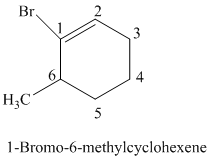
The parent is a six carbon atom ring since the name has the word “cyclohex.” The functional group is alkene. The double bond is present between
The structural formula is as follows:
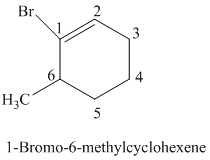
i)
The parent chain has five carbon atoms since the name has the word “pent.” A double bond is present between
The structural formula is as follows:
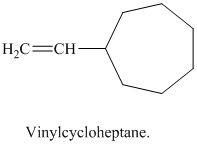
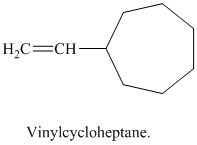
j) Vinylcycloheptane
The structure of a vinyl group is
A cycloheptane ring is attached to this vinyl group. The ring has seven carbon atoms. The structural formula is as follows:
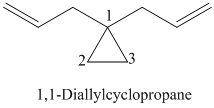
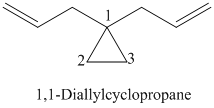
k)
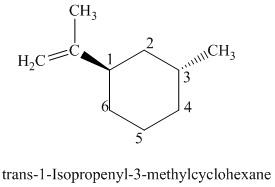
l)
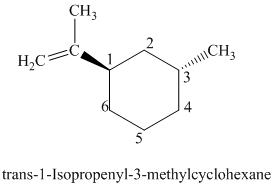
The parent is a six carbon atom ring since the name has the word “cyclohex.” An isopropenyl group is attached to the
The structural formula is as follows:
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry - Standalone book
- Draw structural formulas for these alkenes. (a) trans-2-Methyl-3-hexene (b) 2-Methyl-2-hexene (c) 2-Methyl-1-butene (d) 3-Ethyl-3-methyl-1-pentene (e) 2,3-Dimethyl-2-butene (f) cis-2-Pentene (g) (Z)-1-Chloropropene (h) 3-Methylcyclohexene (i) 1-Isopropyl-4-methylcyclohexene (j) (E)-2,6-Dimethyl-2,6-octadiene (k) 3-Cyclopropyl-1-propene (l) Cyclopropylethene (m) 2-Chloropropene (n) Tetrachloroethylene (o) 1-Chlorocyclohexenearrow_forwardWhat is the molecular formula of 5-chloro-1-ethylcyclohexene? O C8H12CI none of these O C8H13CI O C₂H₁6CI O C6H₁4 CIarrow_forwardWhich are constitutional isomers ? hexanal and 2 hexanone (R)-2-chlorobutane and (S)-2-chloropropane (E)-2-hexene and (Z)-2-hexene 1-bromobutane and 1-bromoethanearrow_forward
- Which of the following structures is 3-ethyl-2,2-dimethylpentane? CH3 CH3-C-CH-CH2-CH3 CH3 CH2-CH3 O CH3-CH-CH-CH2-CH3 CH3 CH2-CH3 CH3 CH3-CH-CH-CH-CH3 CH3 CH2-CH3 CH3 CH3-CH-C-CH2-CH3 CH3 CH2-CH3arrow_forwardDraw the structure of the following compounds. Use line- angle structural formula. 1.2-bromo-4- methylbicyclo[3.2.1]octane 2.5-chloro-1-ethylspiro[2.5]octane 3.3,7-dimethyl-5-propylnonane 4.1-bromo-4-ethyl-5- methylcyclohexane 5.5-cyclobutyl-2-cyclopropyl-6,8- diethyl-3,7,7-trimethyldecanearrow_forwardName the following molecule. CH₂CH3 H-CH3 H3C H Br (2R, 3R)-2-bromo-3-methylpentane (2S,3S)-2-bromo-3-methylpentane O (2R,3S)-2-bromo-3-methylpentane (1R, 2S)-1-bromo-1,2-dimethylbutane (1S,2R)-1-bromo-1,2-dimethylbutanearrow_forward
- Give the IUPAC name for the following compound. To preview the image click here Br O 5-bromo-3,6-dimethyl-(3Z)-heptene O 5-bromo-3,6-dimethyl-(3E)-heptene O 3-bromo-2,5-dimethyl-(4Z)-heptene O 3-bromo-2,5-dimethyl-(4E)-heptenearrow_forwardWhich of the following is a correct IUPAC name for the molecule shown. CH, H3C CH,CH,CH2 CH3 O (2E, 4Z)-4,5-dimethyloctadiene O (2E, 4E)-4,5-dimethyloctene • (2E, 4E)-4,5-dimethyloctadiene o (4E, 6E)-4,5-dimethyloctadiene (22, 4Z)-4,5-dimethyloctadienearrow_forwardA student drew the structural diagram shown below. CH, 2 HHHHHHHH 1 H-C-C-C-C-C-C-C-C-H III HHHHHHHH 3 CH₂CH₂CH₂ oct-3-ene Compounds that Contain Eight Carbons 2,3-dimethylhex-2-ene I 5 CH₂CH₂ 6 2,3-dimethyl-1-propylcyclopropane 7 CH₂ CH₂ 4 3,3-dimethylhexane The compounds above that are isomers of the structural diagram that the student drew are numbered ______ '__—_ › andarrow_forward
- c) For the IUPAC names below, provide an unambiguous structure! 1) (1R, 2R, 4S)-1,2-dibromo-4-methyl-cyclohexane 2) (2R, 3Z)-5-Fluoro-3-penten-1,2-diolarrow_forwardWhich will exhibit geometric isomerism? O 11-dimethylcyclohexane Methylcyclohexane 1-ethyl-3-methylcyclohexane 2,3-dimethyl-1-hexenearrow_forwardWhat is the IUPAC name for the molecule shown below? CI (4R,5Z)-4-chlorohept-5-en-2-yne (2E,4S)-4-chlorohept-2-en-5-yne (4R, 5E)-4-chlorohept-5-en-2-yne O (2E,4S)-4-chlorooct-2-en-5-ynearrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning



