
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5.9, Problem 12P
Carbocations are key intermediates in petroleum refining. Of particular importance is one having the carbon skeleton shown.
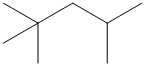
How many different
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
N,N-diethyl-m-toluamide (DEET) is the active ingredient in many insect repellent preparations. Following is one of the steps in its synthesis. In the box below draw the
structure of the product of this reaction.
H3C
MgBr
1. CO₂
2. H₂O*
product
• You do not have to consider stereochemistry.
• You do not have to explicitly draw H atoms.
• Do not include lone pairs in your answer. They will not be considered in the grading.
• Draw the Grignard reagent as a covalent magnesium bromide.
N,N-diethyl-m-toluamide (DEET) is the active ingredient in many insect repellent preparations. Following is one of the steps in its synthesis. In the box below
draw the structure of the product of this reaction.
H3C
MgBr
1. CO2
2. H30*
product
• You do not have to consider stereochemistry.
• You do not have to explicitly draw H atoms.
• Do not include lone pairs in your answer. They will not be considered in the grading.
• Draw the Grignard reagent as a covalent magnesium bromide.
P
ору
aste
O O O- []#
Previous
Next
Email Instructor
Save and Exit
Draw all the isomers of C5H₁0. Clearly show stereochemistry if stereoisomers are possible.
Step 1: Start by drawing all the isomers with double bonds.
• Consider constitutional isomers, then stereoisomers.
Step 2: Draw all isomers containing a ring.
• Consider constitutional isomers, then stereoisomers.
Step 2: Next consider isomers containing a ring. The largest ring possible with this molecular formula contains 5 carbons, while
the smallest possible ring contains 3 carbons.
Draw an isomer containing a 5-carbon ring.
Rings More
Select Draw
/
C
H
Erase
Consider a 4-carbon ring of C5H₁0. Draw stereoisomers,
if applicable.
Select Draw Rings
More
C
H
Erase
Chapter 5 Solutions
Organic Chemistry - Standalone book
Ch. 5.1 - Prob. 1PCh. 5.1 - Prob. 2PCh. 5.1 - Many compounds contain more than one functional...Ch. 5.2 - Prob. 4PCh. 5.3 - Prob. 5PCh. 5.4 - Classify the isomeric C4H10O alcohols as being...Ch. 5.5 - Bromine is less electronegative than chlorine, yet...Ch. 5.6 - Prob. 8PCh. 5.7 - Prob. 9PCh. 5.8 - Prob. 10P
Ch. 5.8 - Prob. 11PCh. 5.9 - Carbocations are key intermediates in petroleum...Ch. 5.9 - Prob. 13PCh. 5.9 - Prob. 14PCh. 5.11 - Prob. 15PCh. 5.13 - Prob. 16PCh. 5.14 - For the reaction of a primary alcohol RCH2OH with...Ch. 5.15 - Prob. 18PCh. 5 - Write structural formulas for each of the...Ch. 5 - Prob. 20PCh. 5 - Prob. 21PCh. 5 - Write structural formulas for all the...Ch. 5 - Prob. 23PCh. 5 - Prob. 24PCh. 5 - Epichlorohydrin is the common name of an...Ch. 5 - Prob. 26PCh. 5 - Prob. 27PCh. 5 - Prob. 28PCh. 5 - Some of the most important organic compounds in...Ch. 5 - Prob. 30PCh. 5 - Prob. 31PCh. 5 - Prob. 32PCh. 5 - Prob. 33PCh. 5 - Prob. 34PCh. 5 - Prob. 35PCh. 5 - Prob. 36PCh. 5 - Prob. 37PCh. 5 - Prob. 38PCh. 5 - Prob. 39PCh. 5 - Prob. 40PCh. 5 - The reaction of 2,2-dimethyl-1-propanol...Ch. 5 - (a) Assuming that the rate-determining elementary...Ch. 5 - The reaction of 3-tert-butyl-3-pentanol with...Ch. 5 - Prob. 44PCh. 5 - Prob. 45PCh. 5 - Prob. 46DSPCh. 5 - Prob. 47DSPCh. 5 - Prob. 48DSPCh. 5 - Prob. 49DSPCh. 5 - Prob. 50DSPCh. 5 - Prob. 51DSPCh. 5 - Prob. 52DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw all the isomers of C,H0. Clearly show stereochemistry if stereoisomers are possible. Step 1: Start by drawing all the isomers with double bonds. • Consider constitutional isomers, then stereoisomers. Step 2: Draw all isomers containing a ring. • Consider constitutional isomers, then stereoisomers. The longest chain does not have to contain all five carbon atoms. Draw the alkene isomers that have a 4-carbon main chain.arrow_forwardN,N-diethyl-m-toluamide (DEET) is the active ingredient in many insect repellent preparations. Following is one of the steps in its synthesis. In the box below draw the structure of the product of this reaction. MgBr H3C 0 ▾ + 1. CO₂ 2. H3O+ • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • Draw the Grignard reagent as a covalent magnesium bromide. n [ ]# product ChemDoodleⓇ 28arrow_forwardHBr addition to ethylene is exothermic. What does this mean? Options: The rate determining step is the first step. The newly formed bonds are more stable (higher dissociation energy) than those broken in the reactants. The carbocation intermediate is higher energy than the alkene. The reaction is very fast.arrow_forward
- Draw a structural formula for the substitution product of the reaction shown below. Br H NH3 CH₂OH • Use the wedge/hash bond tools to indicate stereochemistry where it exists. If more than one stereoisomer of product is formed, draw both. • Separate multiple products using the + sign from the drop-down menu. • Products that are initially formed as ions should be drawn in their neutral forms.arrow_forwardClassify the following alkyl halide as primary, secondary, tertiary? and Why? CH₂Cl benzyl chloride cr chlorobenzenearrow_forwardDraw structural formulas for the cis and trans isomers of hydrindane. Show each ring in its most stable conformation. Which of these isomers is more stable?arrow_forward
- 1. Consider the following reaction - Dehydrogenation of Methyl Cyclohexane (MCH) to Toluene (TOL) using a Precious Metal (Pt) supported on Alumina. CH3 CH3 2 () + 3 H₂ Typically, hydrogenation of a cyclo-alkane, like MCH, occurs in 3 or more stages, where one bond is dehydrogenated per stage, eventually resulting in the dehydrogenation of 3 bonds. Strong adsorption of MCH is likely to occur, but the adsorption of hydrogen (either as a molecule on a single site OR as an atom on a single site) is unclear. Therefore, in this question, two types of reaction mechanisms are to be considered, as discussed below: (a) You are to first propose a reaction mechanism which includes adsorption, surface reaction, and desorption. For each of the following cases, please write down a full set of elementary reaction mechanisms. (0) Extraction of Hydrogen in 3 stages, based on dehydrogenation of each bond-where the ensuing hydrogen product is adsorbed as a molecule on a single site, in one step.…arrow_forwardCarvone is the major constituent of spearmint oil. Draw the major organic product of the reaction of carvone with HOCH2CH2OH, HCI. Carvone • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • In cases where there is more than one answer, just draw one. • If no reaction occurs, draw the organic starting material. + Sn 1arrow_forward3. Please draw the most important (stable) resonance form of the carbonation that mediates the following reaction: OH H3O+ O 4. Draw the structure of the alkene isomer (with the molecular formula of C5H10) that is most reactive one in the addition reaction with Br₂.arrow_forward
- The two alkenes below react with HI at different rates. CH3CH=CHCH3 and CH₂=CH₂ Draw the structural formula of the MAJOR product formed by the alkene having the HIGHER reaction rate. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. TAYY [ ] در ChemDoodle Ⓡarrow_forwardDraw a structural formula for the substitution product of the reaction shown below. +- Na SCH3 CH3CN • Use the wedge/hash bond tools to indicate stereochemistry where it exists. If more than one stereoisomer of product is formed, draw both. ● Separate multiple products using the + sign from the drop-down menu. • Products that are initially formed as ions should be drawn in their neutral forms.arrow_forwardDraw a structural formula for the substitution product of the reaction shown below. ● H3C HH Br + O • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • If more than one stereoisomer of product is formed, draw both. Separate multiple products using the + sign from the drop-down menu. • Products that are initially formed as ions should be drawn in their neutral forms. ▼ In [F OCH 3 -85 ? Na CN CH3CH₂OH ChemDoodleⓇ >arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License