
Concept explainers
Interpretation:
The Lewis structure of the molecule whose Newman projection is given is to be drawn.
Concept introduction:
In a Newman projection, the bond of interest connecting the atoms is not shown. But the atoms connected are shown explicitly. The front atom is shown as a point while the back atom is shown as a circle. The atoms attached to the front carbon converge at the point while the atoms attached to the back carbon are shown connected to the circle.
Answer to Problem 4.1P
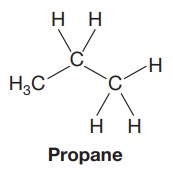
The Lewis structure of the molecule is:
Explanation of Solution
The given Newman projection is:
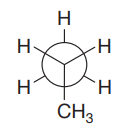
In this Newman projection, the front carbon atom is shown by a point. It is attached to two hydrogen atoms and a methyl group. The back carbon atom is shown by a circle. It is attached to three hydrogen atoms. Thus, in the Lewis structure, there will be a chain of three carbon atoms. The molecule is propane. Its Lewis structure is shown below:

The Lewis structure of the molecule is drawn as shown above.
Want to see more full solutions like this?
Chapter 4 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Problem Draw Lewis structures for the following:(a) Ethylene (C2H4), the most important reactant in the manufacture of polymers(b) Nitrogen (N2), the most abundant atmospheric gasPlan We show the structure resulting from steps 1 to 4: placing the atoms, counting the total valence electrons, making single bonds, and distributing the remaining valence electrons in pairs to attain octets. Then we continue with step 5, if needed.arrow_forwardWould each end of the bond line structure be carbon? Is problem (a) correct?arrow_forwardBelow are two sets of resonance structures. Where applicable provide the missing curved arrow notation, lone pair electrons, and nonzero formal charge. Do not delete or add any bonds or atoms.arrow_forward
- Problem Which of the following substances exhibits H bonding? For any that do, show the H bonds between two of its molecules. (a) C̟H, (b) CH;OH (c) CH3C–NH,arrow_forwardPlease answer this asap.. you can see the example on the other picture Instruction: Identify the hybrid orbitals used in the given moleculesarrow_forwardIdentify all functional groups that are present in strychnine, a highly toxic alkaloid used as a pesticide to kill rodents, whose line structure is shown here. What compound class is characteristic of each of those functional groups? Strychninearrow_forward
- Consider the Lewis structure shown for thionitromethane. Draw the major resonance structure for the compound shown; include lone pairs of electrons, formal charges, and condensed hydrogen atoms (located in the More menu). Then draw curved arrows to show how this can be converted to the Lewis structure given.arrow_forwardPlease draw a more stable resonance structure for the following molecule. Use a curved arrow to show how to transform the original structure to the new one and please specify charges.arrow_forward4)On the following molecules, draw the curved arrows to convert the left-hand resonance structure form to the right-hand resonance structure formarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
