
Concept explainers
- a. Draw the Lewis structure for ethene (ethylene), H2CCH2, a small hydrocarbon with a
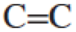 double bond.
double bond. - b. Based on this structure, predict the H–C–H bond angle. Explain your reasoning.
- c. Sketch the molecule showing the predicted bond angles.
(a)
Interpretation:
The Lewis structure for ethylene has to be drawn.
Concept Introduction:
Lewis structures are diagrams that represent the chemical bonding of covalently bonded molecules and coordination compounds.
The chemical bonding present in covalently bonded molecules and in coordination compounds are represented using Lewis structures.
It is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure. In these cases, the chemical bonding are described by delocalization of electrons and is known as resonance.
All the possible resonance structures are imaginary whereas the resonance hybrid is real.
These structures will differ only in the arrangement of the electrons not in the relative position of the atomic nuclei.
Lewis structure for any molecule is drawn by using the following steps,
First the skeletal structure for the given molecule is drawn then the total number of valence electrons for all atoms present in the molecule is determined
The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it.
Finally, the electrons which got after subtractions have to be equally distributed such that each atom contains eight electrons in its valence shell.
Molecular geometry is the shape of a molecule predicted by considering only bond pair of electrons.
Geometry of different type of molecules with respect to the number of electron pairs are mentioned below,
Explanation of Solution
The Lewis electron dot structure for given molecules are determined by first drawing the skeletal structure for the given molecules, then the total number of valence electrons for all atoms present in the molecules are determined.
The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it.
Finally, the electrons which got after subtractions have to be equally distributed considering each atom contains eight electrons in its valence shell.
The given moleucle is ethylene
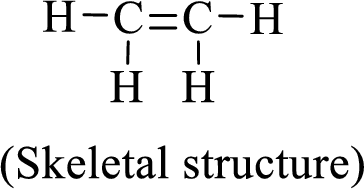
Thus, the Lewis structure of given compound is,
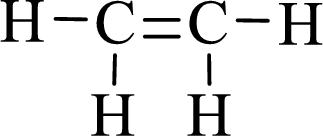
(b)
Interpretation:
The bond angle value in
Concept Introduction:
Molecular geometry is the shape of a molecule predicted by considering only bond pair of electrons.
Geometry of different type of molecules with respect to the number of electron pairs are mentioned below,
Explanation of Solution
Lewis structure for the given compound ethylene is,
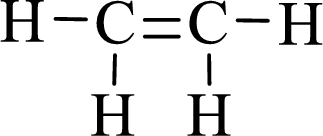
Here, the carbon atom does not have lone pair of electrons and the geometry around the carbon atom is triogonal planar, thus the bond angle in between
(c)
Interpretation:
The ethylene molecule has to be sketched and the predicted bond angle should be shown in the structure.
Concept Introduction:
Molecular geometry is the shape of a molecule predicted by considering only bond pair of electrons.
Geometry of different type of molecules with respect to the number of electron pairs are mentioned below,
Explanation of Solution
Lewis structure for the given compound ethylene is,
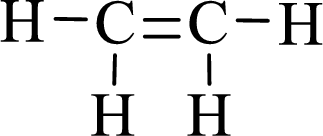
Here, the carbon atom does not have lone pair of electrons and the geometry around the carbon atom is triogonal planar, thus the bond angle in between
According to the predicted bond angle the structure can be sketched as follows,
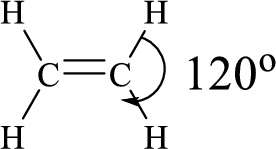
Want to see more full solutions like this?
Chapter 4 Solutions
Chemistry In Context
Additional Science Textbook Solutions
Chemistry & Chemical Reactivity
CHEMISTRY-TEXT
Inorganic Chemistry
Inorganic Chemistry
Chemistry: Matter and Change
Principles of General, Organic, Biological Chemistry
- Consider the molecules — BrF5. A. Draw the best Lewis structure for this molecule. Label any atoms with nonzero formal charge. B. Label each bond angle. As part of your answer be sure to include if it is more or less than the ideal bond angle. C. What is the electron geometry around the bromine atom? D. Are the bonds in the molecule polar? E. Is the overall molecule polar? — CH2 F2 . A. Draw the best Lewis structure for this molecule. B. Label each bond angle. Answers for A-D here: C. Redraw the shape of the molecule. Draw all dipoles. D. Is the overall molecule polar? — Consider the molecule CH2 CF2 . A. Draw the best Lewis structure for this molecule. B. Label each bond angle. C. Redraw the shape of the molecule (according to the exacting specifications of your instructor). Draw all dipoles. D. Is the overall molecule polar?arrow_forwardChemistry: Bonding 4a. Draw the Lewis structure for CH4, (methane), NH3, (ammonia), and H20 (water). b. Calculate the bond polarity for the C-H bond, N-H bond, and O-H bond. c. Classify these molecules as either polar or nonpolar. If the molecule is polar, label the partial negative and partial positive sides of the molecules you drew above.arrow_forwardSodium Azide is an ionic compound that is used in automotive air bags. It has the chemical formula NaN3 (The Azide ion is N , and should not be confused with the Nitride ion, N°). a. Give the LEWIS diagram of the Azide ion. b. What is the geometry of the Azide ion? What is the bond angle of the Azide ion?arrow_forward
- Draw the lewis structure for SH2. Then identify the electron pair geometry (EPG), molecular geometry (MG) bond angle (BA), and polarity (P). Select one: a. EPG: tetrahedral, MG: Bent, BA: 120*, P: non-polar b. EPG: tetrahedral, MG: bent, BA: <<109.5*, P: polar c. EPG: tetrahedral, MG: Bent, BA: <<109.5*, P: non-polar d. EPG: bent, MG: tetrahedral, BA: <<109.5*, P: polararrow_forwardChange 1,2-ethenediol into 1,2 ethynediol (HOCCOH) by removing one hydrogen atom from each carbon atom and replacing with a triple bond between the carbon atoms. Be sure to add the third rod to represent the triple bond. Draw a Lewis structure for 1,2-ethynediol. A. Identify the electron-pair and molecular geometry around each carbon and oxygen atom. B. What are the bond angles for C-C-O? For C-O-H? C. Can the molecule interconvert or “flip” between different structures in which the –OH group on the two carbon atoms occupy different orientations with respect to each other? If so, how does this happen? If not, why?arrow_forwardFill in the table. Central atom is listed first. A. Write the number of valence electrons below the formulaB. Draw the Lewis structureC & D. Write the Electron Group Geometry and Molecular Shape NamesE. Write the bond angleF. Write the molecular polarity. "P" for polar and "NP" for nonpolar. SpeciesValenceElectrons(1 pt.) LewisStructure(2 pt.) Electron PairGeometryName(1 pt.) Molecular ShapeName (1 pts.) BondAngle (1 pt.) Molecular Polarity(1 pt.) PO43- NOBr Uploadarrow_forward
- The formula for nitryl chloride is CINO2 (in which N is the central atom). a.Draw the Lewis structure for the molecule, including all resonance structures. b.What is the N-O bond order? c.Describe the electron-pair and molecular geometries and give values for all bond angles. d.What is the most polar bond in the molecule? Is the molecule polar? e.The computer program used to calculate electrostatic potential surfaces gave the following charges on atoms in the molecule: A =-0.03, B = -0.26, and C = +0.56. Identify the atoms A, B, and C. Are these calculated charges in accord with your predictions?arrow_forward4. Multiple structures with the same chemical formula (number and type of each element present) but differing in the atom connectivity are called structural isomers. Use the skeletal structure below to draw a structural isomer of what you have already drawn in Table 4. H O H 0 CN H a. Label the central atoms like you did in Table 4 and name the geometries of each of them.arrow_forwardDraw the lewis structure for SH2. Then identify the electron pair geometry (EPG), molecular geometry (MG) bond angle (BA), and polarity (P). Select one: a. EPG: tetrahedral, MG: Bent, BA: <<109.5*, P: non-polar b. EPG: bent, MG: tetrahedral, BA: <<109.5*, P: polar c. EPG: tetrahedral, MG: bent, BA: <<109.5*, P: polar d. EPG: tetrahedral, MG: Bent, BA: 120*, P: non-polararrow_forward
- What is the chemical formula for ethane? Observe your ethane molecule. Are there any polar covalent bonds present? Based on your answer would you predict that ethane would or would not dissolve easily in water? Now imagine taking off one hydrogen from ethane and adding a hydroxyl group in its place. Draw your completed ethanol molecule below. ouy? Ethanol is an alcohol found in drinks such as beer and wine. Examine this molecule. Are there any polar cov bonds present? Based on your answer, would you predict that ethanol would or would not dissolve easily in water?arrow_forward3. Draw the Lewis-dot structure for C4H10 below. Remember the Lewis-dot structure is the same as the Expanded structure. A. What is the main intermolecular force acting on this molecule? B. Is this molecule polar or nonpolar? Provide a brief explanation (2-3 sentences) for your answer.arrow_forwardDraw the 2D and the 3D Lewis structure for each molecule. You do not need to show resonance structures here. Determine the electron pair geometry (EPG), the molecular geometry (MG), the bond angle (BA) around the central atom, and the hybridization of the central or bold atom. Circle the formula of any polar molecule. For molecules with more than one central atom (e.g. CHCH20H) give the structure around the bold atom.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





