
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3.12, Problem 12P
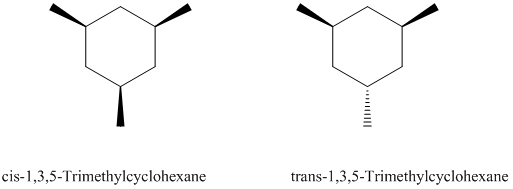
Based on what you know about disubstituted cyclohexanes, which of the following two stereoisomeric
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Why is a substituted cyclohexane ring more stable with a larger group in the equatorial position?
Draw the most stable conformers of trans-1-Bromo-4-methylcyclohexane and cis-1-Bromo-4-methylcyclohexane. Which is more stable? Explain the reason for your answer.
can we convert this Trans-1-tert-butyl-3-methylcyclohexane into (the most stable version of)cis-1-tert-butyl-3-methylcyclohexane?what would cis-1-tert-butyl-3-methylcyclohexane be like?
Chapter 3 Solutions
Organic Chemistry - Standalone book
Ch. 3.1 - Identify the alkanes corresponding to each of the...Ch. 3.1 - Find the conformations in Figure 3.4 in which the...Ch. 3.2 - Sketch a potential energy diagram for rotation...Ch. 3.2 - Acetylcholine is a neurotransmitter in the central...Ch. 3.2 - Prob. 5PCh. 3.5 - The heats of combustion of ethylcyclopropane and...Ch. 3.8 - Prob. 7PCh. 3.10 - The following questions relate to a cyclohexane...Ch. 3.10 - Draw the most stable conformation of...Ch. 3.11 - Prob. 10P
Ch. 3.11 - Prob. 11PCh. 3.12 - Based on what you know about disubstituted...Ch. 3.12 - Write structural formulas for the most stable...Ch. 3.14 - Cubane (C4H8) is the common name of the polycyclic...Ch. 3.14 - Prob. 15PCh. 3.14 - Prob. 16PCh. 3.14 - Prob. 17PCh. 3.14 - Prob. 18PCh. 3.15 - Prob. 19PCh. 3 - Give the IUPAC names of each of the following: (a)...Ch. 3 - Draw Newman projections for the gauche and...Ch. 3 - Identify all atoms that are (a) anti and (b)...Ch. 3 - Prob. 23PCh. 3 - Prob. 24PCh. 3 - Prob. 25PCh. 3 - Prob. 26PCh. 3 - Prob. 27PCh. 3 - Prob. 28PCh. 3 - Oxidation of 4-tert-butylthiane proceeds according...Ch. 3 - The following are representations of two forms of...Ch. 3 - Draw (a) a Newman projection of the most stable...Ch. 3 - Write a structural formula for the most stable...Ch. 3 - Sight down the C-2-C-3 bond, and draw Newman...Ch. 3 - Prob. 34PCh. 3 - Sketch an approximate potential energy diagram for...Ch. 3 - Prob. 36PCh. 3 - Even though the methyl group occupies an...Ch. 3 - Which do you expect to be the more stable...Ch. 3 - Arrange the trimethylcyclohexane isomers shown in...Ch. 3 - Identify the more stable stereoisomer in each of...Ch. 3 - One stereoisomer of 1,1,3,5-tetramethylcyclohexane...Ch. 3 - One of the following two stereoisomers is...Ch. 3 - In each of the following groups of compounds,...Ch. 3 - The heats of combustion of the more and less...Ch. 3 - The measured dipole moment of ClCH2CH2Cl is 1.12D....Ch. 3 - Prob. 46PCh. 3 - Prob. 47PCh. 3 - Prob. 48DSPCh. 3 - Prob. 49DSPCh. 3 - Prob. 50DSPCh. 3 - Prob. 51DSPCh. 3 - Prob. 52DSPCh. 3 - Prob. 53DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The heats of hydrogenation of cycloheptene and 1,3,5-cycloheptatriene are 110 kJ/mol (26.3 kcal/mol) and 305 kJ/mol (73.0 kcal/mol), respectively. In both cases cycloheptane is the product. What is the resonance energy of 1,3,5-cycloheptatriene? How does it compare with the resonance energy of benzene?arrow_forwardDetermine which cyclohexane structure has the MOST energy (is the LEAST stable)?arrow_forwardHow many possible stereoisomers can be written for 1-ethyl-3- methylcyclohexane? O 2 O4 8arrow_forward
- Which will be more stable, cis or trans-1,4-tert-butylcyclohexane? Explain by drawing their structures?arrow_forwardWhich compound is more stable: cis-1,2-dimethylcyclopropane or trans-1,2- dimethylcyclopropane? Explain your answer.arrow_forwardWhich of the following two conformations of cis-1-bromo-4-methylcyclohexane is more stable? Use the strain energy increments in the table below estimate the energy difference (a positive number) between them.arrow_forward
- What is the systematic name for the 1,2,4-trichlorocyclohexane stereoisomer with the smallest heat of combustion?arrow_forwardWhich has a higher percentage of the diequatorial-substituted conformer compared with the diaxial-substituted conformer: trans-1,4-difluorocyclohexane or cis-1-tert-butyl-3- fluorocyclohexane? O trans-1,4-difluorocyclohexane O cis-1-tert-butyl-3-fluorocyclohexanearrow_forwardStereoisomers share the same connectivity and differ only in the way their atoms are arranged in space. Draw the structure of a compound that is a stereoisomer of trans-1,4-dibromocyclohexane cis-1,2-dimethylcyclobutane cis-1,3-dimethylcyclobutanearrow_forward
- Which of the following compounds has a stereoisomer that is a meso compound? A 2,4-dibromohexane B 2,4-dibromopentane C 2,4-dimethylpentane D 1,3-dichlorocyclohexane E 1,4-dichlorocyclohexane F 1,2-dichlorocyclobutanearrow_forwardProvide a systematic name of the following compound: 3-butyl-2,5-dimethyl-6-(1-methylethyl)octane O 3-ethyl-2,4-dimethyl-6-(1-methylethyl)decane 3-ethyl-6-(2-propyl)-2,4-dimethyldecane 3-butyl-2,5-dimethyl-6-(2-propyl)octane 3,5-diisopropyl-4-methyldecane O O O O Oarrow_forwardWhich would you predict to have the larger (more negative) heat of combustion, cis-1,4-dimethylcyclohexane or trans-1,4-dimethylcyclohexane?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License