
Concept explainers
Most modern scanning electron microscopes (SEMs) are equipped with energy-dispersive X-ray detectors for the purpose of chemical analysis of the specimens. This X-ray analysis is a natural extension of the capability of the SEM because the electrons that are used to form the image are also capable of creating characteristic X-rays in the sample. When the electron beam hits the specimen, X-rays specific to the elements in the specimen are created. These can be detected and used to deduce the composition of the specimen from the well-known wavelengths of the characteristic X-rays of the elements. For example:
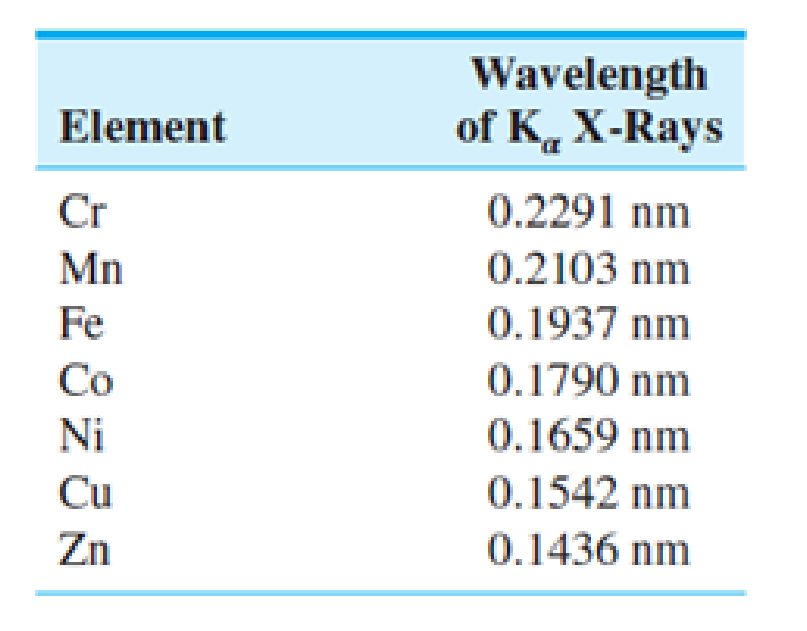
Suppose a metallic alloy is examined in an SEM and three different X-ray energies are detected. If the three energies are 7492, 5426, and 6417 eV, what elements are present in the sample? What would you call such an alloy? (Look ahead to Chap. 9 in the textbook.)
The element present in the sample when the X-ray energy is 7462 eV.
Answer to Problem 70SEP
The element present in the sample when the X-ray energy is 7462 eV is
Explanation of Solution
Write the equation to calculate the wavelength of the characteristic X-rays of the element
Here, energy of the radiation is
Conclusion:
The values of Planck constant and speed of light are
Substitute
From the given table, the wavelength value of
Thus, the element present in the sample when the X-ray energy is 7462 eV is
The element present in the sample when the X-ray energy is 5426 eV.
Answer to Problem 70SEP
The element present in the sample when the X-ray energy is 5426 eV is
Explanation of Solution
Conclusion:
Substitute
From the given table, the wavelength value of
Thus, the element present in the sample when the X-ray energy is 5426 eV is
The element present in the sample when the X-ray energy is 6417 eV.
Answer to Problem 70SEP
The element present in the sample when the X-ray energy is 6417 eV is
Explanation of Solution
Conclusion:
Substitute
From the given table, the wavelength value of
Thus, the element present in the sample when the X-ray energy is 6417 eV is
The elements, chromium, iron and nickel present in the sample represent the principal constituents of the austenitic stainless steels.
Want to see more full solutions like this?
Chapter 2 Solutions
Foundations of Materials Science and Engineering
- You can use 1kV-30kV in a scanning electron microscope (SEM). You are asked to analyze the surface topography as well as bulk microstructure of a cast iron. Keep working distance and spot size constant for your experiment. CALCULATE the ‘electron interaction’ volume or depth of penetration of electron beams to the sample for the 2kV voltage (applied). Which voltage (2kV, 5kV, 10kV, 20kV, or 30kV) do you prefer for surface topography and which one for bulk microstructure? For calculation, consider the cast iron as just iron. Show your work.arrow_forwardA cell culture is covered with a 1 cm sheet of Lucite (density=1.19 g/cm^3). What thickness of lead (in cm) is needed on top of the Lucite to prevent 10MeV beta rays from reaching the culture? Use the approximate empirical formulas relating range Rin g/cm^2 to electron kinetic energy T in MeV:arrow_forward(a) how much time is needed to measure the kinetic energy of an electron whose speed is 10 metre per second with an uncertainty of and no more than 0.1 percent. How far will the electron have travel in this period of time. (b) make the same calculation for a one gram insect whose speed is the same what do the set of figure indicate?arrow_forward
- Q1 In a parallel plate diode, the anode is at 230 V with respect to the cathode which is 4.5 mm away from it. An electron is emitted from cathode with an initial velocity of 3 x 106 m/s towards the anode. Calculate; (i) the arrival velocity of the electron at anode. (ii) time of transit. (iii) the velocity and distance travelled by the electron after 6 x 10-10 s.arrow_forwardConsider the Bragg equation in the x-ray diffraction. If the value of the wavelength is doubled, which of the following is NOT true?Required to answer. Single choice. Bragg angles of reflections increase The d spacings become smaller. The diffraction pattern expands. Some previously accessible reflections can no longer be measured. None of the abovearrow_forwardWhat do you mean by characteristic x rays?arrow_forward
- What kind of material would you expect to show an x-ray diffraction profile like the one shown below? 130K 120K 110K 100K- 90K- 80K- 70K- 60K- 50K- 40K- 30K- 20K 10K Intensity (counts) 20⁰ O a. A material containing iron O b. A crystalline solid O c. O d. A single crystal solid A liquid A mixed amorphous and crystalline solid O e. O f. An amorphous solid A grain boundary is an example of a A vacancy is an example of a An interstitial is an example of a A dislocation is a perfect crystal 1-dimensional (line) defect 3-dimensional (volume) defect 0-dimensional (point) defect 2-dimensional (plane) defect Angle (degrees 20arrow_forward8. An electron has a rest mass of 9.11x10^(-28) g. What is the energy contained in the electron provided it is accelerated to the speed of light in kJ? Express the answer in 4 significant figures.arrow_forwardWhy do quantum effects only happen on the atomic scale?arrow_forward
- What thickness of aluminum is required to reduce 200Kev gamma rays to 10% of its original value? Note that the thickness of half the value at energy Kev 200 in aluminum is 2.14 cmarrow_forwardExplain the main postulate of the VSEPR model. List the five base geometries (along with bond angles) that most molecules or ions adopt to minimize electron-pair repulsions. Why are bond angles sometimes slightly less than predicted in actual molecules as compared to what is predicted by the VSEPR model?arrow_forwardPolarizability is defined as the extent to which the electron cloud surrounding an atom or molecule can be distorted by an external charge. Rank the halogens (F2,Cl2,Br2,I2) and the noble gases (He,Ne, Ar, Kr, Xe) in order of polarizability (from least polarizable to most polarizable). What characteristics of these substances could be used to determine this ranked order?arrow_forward
 Understanding Motor ControlsMechanical EngineeringISBN:9781337798686Author:Stephen L. HermanPublisher:Delmar Cengage Learning
Understanding Motor ControlsMechanical EngineeringISBN:9781337798686Author:Stephen L. HermanPublisher:Delmar Cengage Learning
