
Concept explainers
Interpretation:
It should be determined that the given [1, 7] sigmatropic rearrangement involves whether suprafacial or antarafacial rearrangement.
Concept introduction:
In a sigmatropic reaction “ one new sigma-bond is formed as another breaks.”
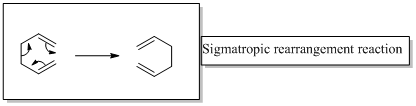
Sigmatropic rearrangement reactions are named with digits. For example a [1, 3] sigmatropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3.
Migration of carbon and hydrogen will occur in a sigmatropic rearrangement reaction.
When hydrogen migrates in a sigmatropic rearrangement, the s orbital of the hydrogen is partially bonded to both the migration origin and the migration terminus in the transition state.
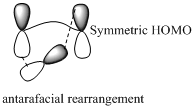
Migration of hydrogen in suprafacial and antarafacial rearrangement can be represented as follows,


Migration of carbon occurs through two ways because it has a two lobed p orbital. Carbon can simultaneously interact with the migration origin and the migration terminus using one lobe of its p orbital. Migration of carbon in suprafacial and antarafacial rearrangement can be represented as follows,
Carbon migrating with one lobe of its p orbital interacting


Carbon migrating with both lobe of its p orbital interacting

Trending nowThis is a popular solution!

Chapter 28 Solutions
Organic Chemistry
- 3a. 3b. 3c 3d. 3e. CO₂ clavulanic acid CH₂-OH H Answer the following questions about clavulanic acid. Does clavulanic acid inhibit D-alanyl-D-alanine transpeptidase? Does clavulanic acid contain a ß-lactam? Does clavulanic acid contain a thiazolium ring? What is the result of the treatment of penicillinase with clavulanic acid? Does clavulanic acid form a covalent acyl-enzyme intermediate with penicillinase?arrow_forwardThe anticoagulant heparin is a polysaccharide that contains alternating residues of -D- glucuronic acid-6- sulfate and N-sulfo-D-glucosamine-6sulfate connected by (1 B 4)- glycosidic linkages. Draw a part of heparin that shows one each of the two residues.arrow_forwardDo the equilibria of the following acid–base reactions lie to the right or the left? (The pKa of H2O2 is 11.6.)arrow_forward
- subunits D-J pleasearrow_forwardWhen CaC2 reacts with water, what carbon-containing compoundforms?(a) CO (b) CO2 (c) CH4 (d) C2H2 (e) H2CO3arrow_forwardThere are 20 common, naturally occurring amino acids from which all proteins are derived (as will be discussed in Chapter 25), although many other less common amino acids have been isolated from natural sources. Valine is one of the 20 common amino acids, and it was used as a starting material in the laboratory synthesis of an uncommon amino acid (Tetrahedron 1997, 53, 1151-1156). During one of the steps in the synthesis, compound 1 was treated with HBr under conditions that favor radical addition, giving stereoisomers 2 and 3. Draw the structures of 2 and 3, and describe their stereoisomeric relationship. H3C- CH₂ H N HT H (S)-valine -CH3 00 Modify the given copies of compound 1 to drawthe structures of 2 and 3. You can use the single bond tool to add/remove double bonds. H3C- -N & N CH₂ HBr OCH₂ hv -CH3 2 + 3arrow_forward
- Which answer describes the relationship between these compounds? (a) OH H OH H (b) H O They are the same molecule. Enantiomers Diastereomers Both Meso OH OH Harrow_forwardTrue or False Beta (1 -> 4) glycosidic linkages are prevalent in carbohydrates considered to be more digestible, such as amylose and maltosearrow_forwardChp15-4(5)arrow_forward
- Draw the structure of 2-azacyclopentanone, g-butyrolactam:arrow_forwardCH-ST15 A mixture of equal amounts of two enantiomers A. is called a racemic mixture B. is optically inactive O.C implies that the enantionmers are meso forms D. is both a racemic mixture and is optically inactive O E. is both a racemic mixture and is optically activearrow_forward5:51 Draw examples of the following: (a) A meso compound with the formula C8H18 (b) A meso compound with the formula C9H20 (c) A compound with two chirality centers, one R and the other Sarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





