
Concept explainers
Interpretation:
Two compounds are to be identified on the basis of their chemical formulas, and IR and
Concept introduction:
Peaks in an IR spectrum help in determining
In 13C NMR spectroscopy, we get information about the different types of carbon present in the given molecular formula.
13C NMR spectroscopy can even differentiate between primary, secondary, tertiary, and quaternary carbons.
If the compound only contains C, H, and O, the index of hydrogen deficiency is calculated as
If a compound contains C, H, O, and a halogen, the halogen can be treated as if it were a hydrogen for the purpose of calculating index of hydrogen deficiency.
Answer to Problem 35P
Solution:
a) The structure of the compound
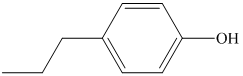
b) The structure of the compound
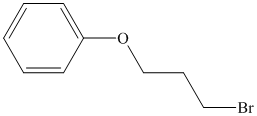
Explanation of Solution
The chemical formula of the compound is
The count of four indicates the possibility of a benzene ring. If a benzene ring is present, the oxygen atom would be singly bonded, either as a hydroxyl group or as an ether functional group.
The IR and 13C NMR spectra of the compound are as follows:
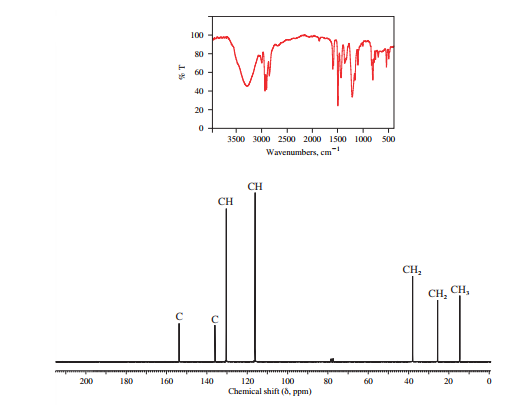
The presence of a broad peak around 3200
Chemical formula of the compound is
Therefore this compound also is likely to have a benzene ring.
The IR and 13C NMR spectra of the compound are as follows:
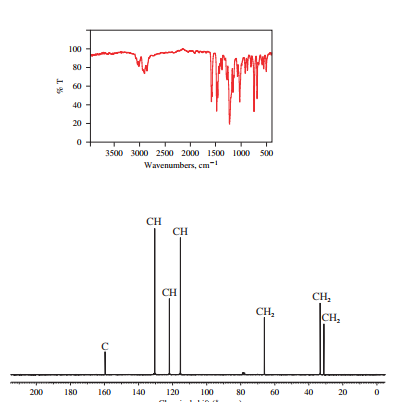
Two peaks in the IR spectrum, at about 700 and 750
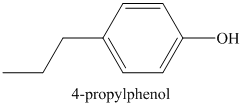
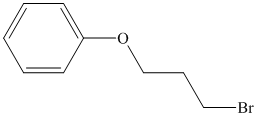
Therefore, the structure of the compound is
Want to see more full solutions like this?
Chapter 23 Solutions
Organic Chemistry - Standalone book
- A ¹H NMR spectrum is shown for a molecule with the molecular formula of C8H10O. Draw the structure that best fits this data. 11 10 9 8 5H 2H 4 3 3H 1 ppmarrow_forwardAssume a compound with the formula C4H8O. a) How many double bonds and/or rings does your compound contain? b) If your compound shows an infrared absorption peak at 1715 cm-1, what functional group does it have? c) If your compound shows a single 1H NMR absorption peak at 2.1 δ, what is its structure?arrow_forwardCompound 2 has molecular formula C6H12. It shows three signals in the 1H-NMR spectrum, one at 0.96 ppm, one at 2.03 ppm, and one at 5.33 ppm. The relative integrals of these three signals are 3, 2, and 1, respectively. Provide structure for compound 2, explain how you reached your conclusion.arrow_forward
- Identify the compound with molecular formula C8H10O that gives the IR and 1H NMR spectra shown here.arrow_forwardidentify the compound with molecular formula C2H6O that gives this 1H NMR spectrum.arrow_forwardAn unknown compound has the molecular formula C9H10O and the Carbon-13 NMR spectrum shown below. Using this information, draw the structure of the unknown compoundarrow_forward
- Compound B has molecular formula C9H12. It shows five signals in the 1H-NMR spectrum - a doublet of integral 6 at 1.22 ppm, a septet of integral 1 at 2.86 ppm, a singlet of integral 1 at 5.34 ppm, a doublet of integral 2 at 6.70 ppm, and a doublet of integral 2 at 7.03 ppm. The 13C-NMR spectrum of B shows six unique signals (23.9, 34.0, 115.7, 128.7, 148.9, and 157.4). Identify B and explain your reasoning.arrow_forwardThe compound A that gives the following IR and NMR spectra has the formula CsH1002. Based on these spectra, determine the structure of A. For NMR analysis, indicate clearly the signals originated from all types of protons in your answer. 100 80 60 %T 40 20 4000 3500 300e 2500 2000 1500 cm-1 зн 3H 2H 2H 4.5 4.0 3.5 3.0 2.5 2.0 1.5 1.0 PPM 928 098 1884 26TT 99T 2945 1962arrow_forwardCompound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively. Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forward
- 1Compound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively. Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forwardTunc 201 Identify the isomer of C3H5Br that has the 13C NMR spectrum shown. Select the single best answer. 220 200 180 160 140 120 100 O Z-1-bromopropene O 2-bromopropene O 3-bromopropene O E-1-bromopropene O bromocyclopropane ppm X 80 S 60 40 CH₂ CH 20 0arrow_forwardA compound with molecular formula C3H8O produces a broad signal between 3200 and 3600 cm³¹ in its IR spectrum and produces two signals in its 13C NMR spectrum. Draw the structure of the compound. Draw Your Solutionarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

