
Concept explainers
We now continue the use of
To make your own roadmap for Chapters 20–23, take a blank sheet of paper and write the following
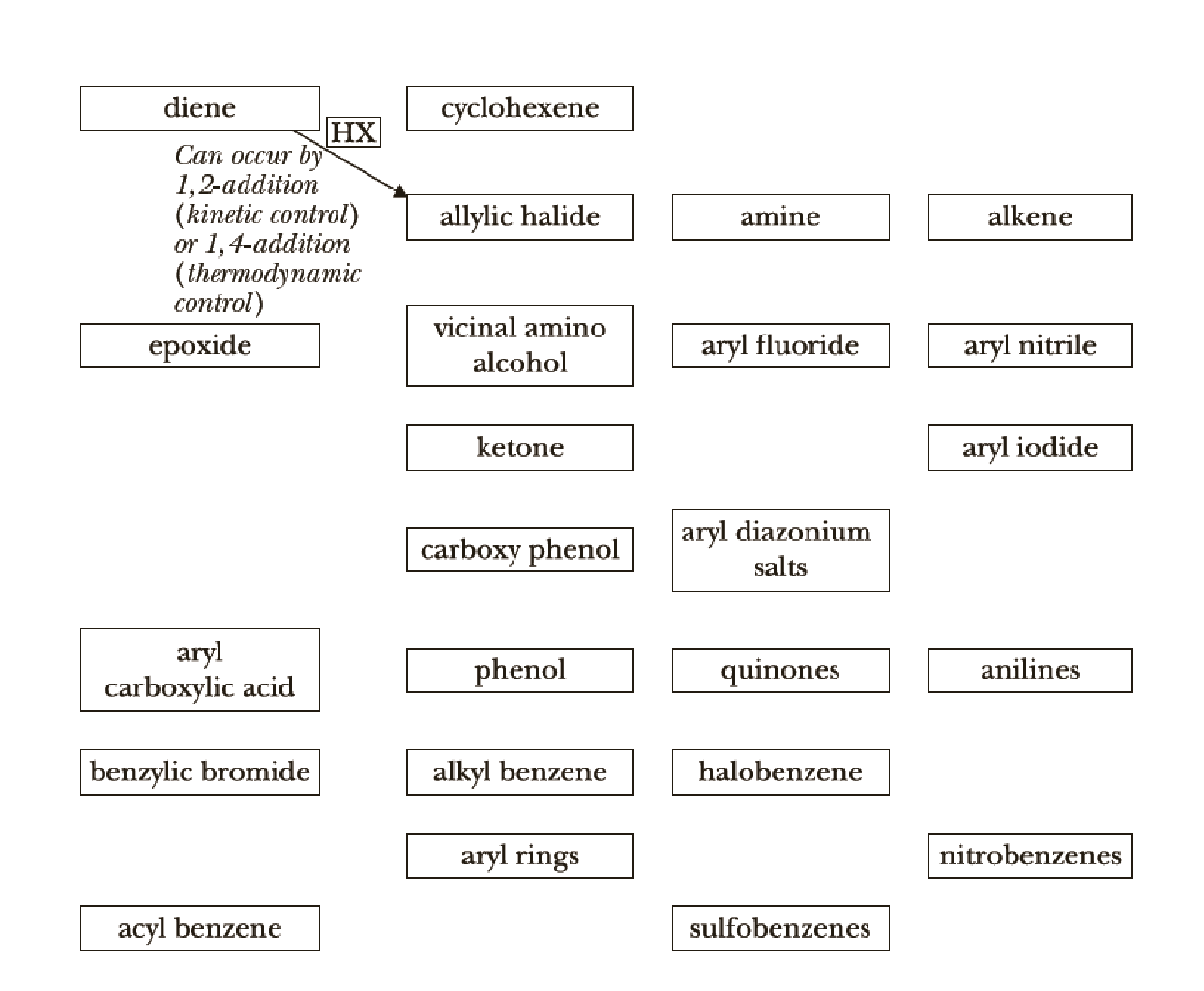
We now continue the use of organic chemistry reaction roadmaps. Because of the unique nature of the new reactions presented, we recommend that you make a new roadmap only for Chapters 20–23.
To make your own roadmap for Chapters 20–23, take a blank sheet of paper and write the following functional groups in the orientations shown. Fill the entire sheet of paper and leave plenty of room between functional groups. Most students find it helpful to use a poster-sized sheet of paper filled out in landscape orientation.
Interpretation:
A reaction roadmap has to be made for the reactions in the study Guide sections of chapters 20-23.
Concept Introduction:
Aromatic substitutions:
Aromatic substitution can be of two types, those are electrophilic and nucleophilic. Aromatic electrophilic substitution is an organic reaction in which an atom that is attached to an aromatic system is replaced by an electrophile. For example nitration, sulphonation, acylation, alkylation, halogenation to aromatic ring is aromatic electrophilic substitution reaction.
Aromatic nucleophilic substitution reactions are those reactions where nucleophile replaces a good leaving group.
Sigmatropic reaction:
A sigmatropic reaction is a pericyclic reaction where the net result is change of one sigma bond to another sigma bond in an uncatalysed intramolecular process.
Explanation of Solution
The reaction road maps for the reactions in the study Guide section of chapters (20-23) will help us to find out the reactions in easy way. The section number indicates the respective reaction segments involved in the study guide section.
Chapters 20 roadmap reaction legend,
Reaction 20.1:
Electrophilic addition to conjugate diene:
Conjugated diene undergoes both 1,2-addition and 1,4-addition reactions with electrophiles often giving mixtures of both kinds of products. The ratio of 1,2-addition to 1,4-addition is temperature dependent. 1,2-addition products are kinetically controlled product and 1,4 addition products are thermodynamically controlled product. When a conjugated diene reacts with
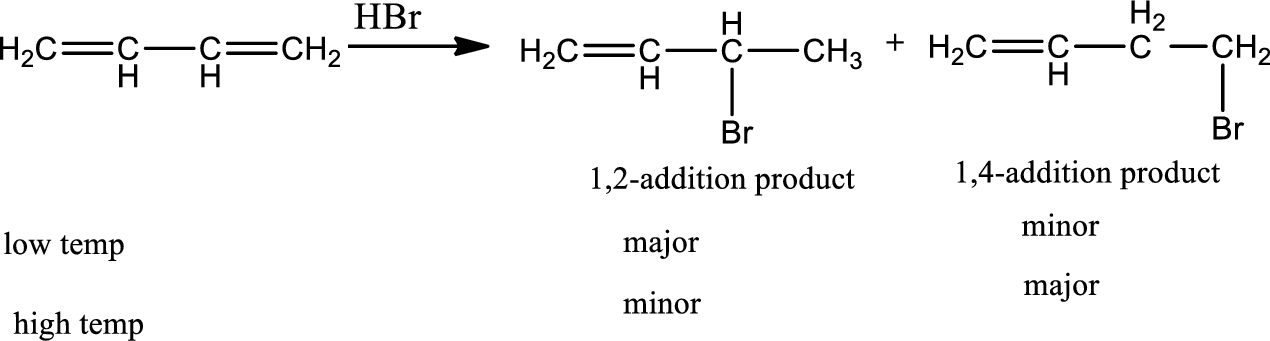
Reaction 20.2:
Diels-Alder reaction:
Conjugated dienes react with certain type of molecules having double or triple bonds, known as dienophile, to form two new sigma bond and a ring structure on the lose of pi bond in a reaction called Diels-Alder reaction. This reaction takes place in a concerted mechanism without intermediates and involves a redistribution of six pi electrons in a cyclic transition state where the pi bond breaking and new sigma bond making occurs simulteneously. The configuration of the diene and dienophile is preserved. Formation of the endo adduct is favoured. This reaction is facilitated by having electron withdrawing groups on dienophile and electron donating groups on diene. The diene must be on s-cis conformation to react. Diels-Alder reaction is an example of
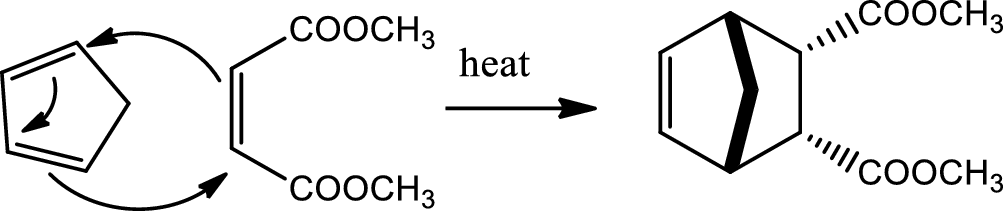
Reaction 20.3:
Claisen rearrangement:
The Claisen rearrangement is a powerful carbon-carbon bond forming chemical reaction. This rearrangement transforms an allyl phenyl ether to an ortho-substituted phenol. This rearrangement is an exothermic, concerted pericyclic reaction. The kinetics are first order and the whole transformation proceeds through a highly ordered cyclic transition state and it is intramolecular.
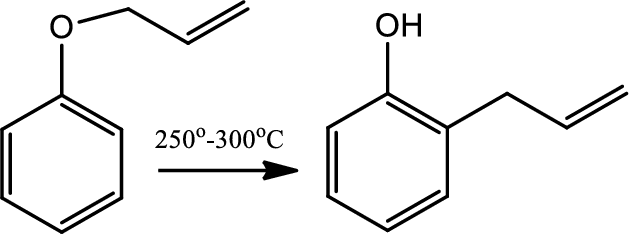
Reaction 20.4:
Cope rearrangement:
Cope rearrangement is the
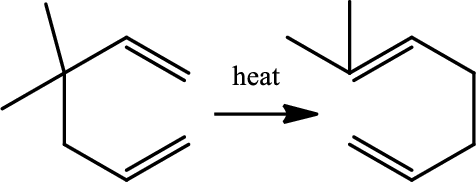
Chapters 21 roadmap reaction legend,
Reaction 21.1:
Acid base reaction of phenol:
Water soluble phenols react with the strong bases to give water soluble salts.
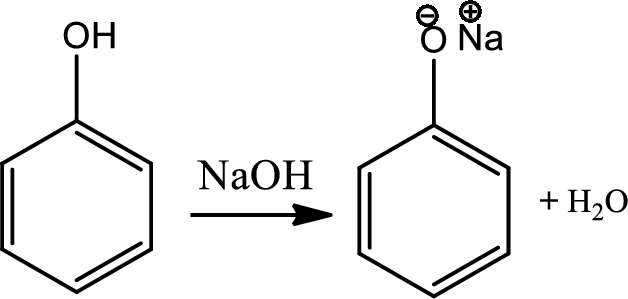
Reaction 21.2:
Kolbe synthesis:
Kolbe synthesis is a carboxylation chemical reaction that involves heating of sodim phenoxide with carbon dioxide and then treating the final product with sulfuric acid. This is basically nucleophilic addition of phenoxide to carbon dioxide that gives substituted cyclohexadienone which undergoes keto-enol tautomerism to regenerate aromatic ring. This gives maily ortho product which is known as salicylic acid.
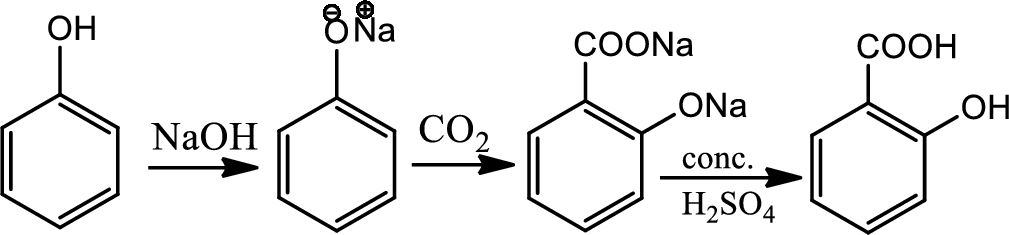
Reaction 21.3:
Oxidation of phenols to quinone:
Phenols can be easily oxidised to quinone. Oxidation of phenols by
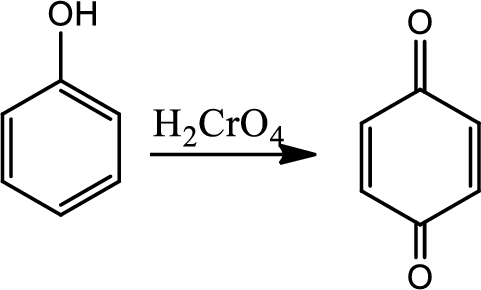
Reaction 21.4:
Oxidation at benzylic position:
The benzylic carbon bonded hydrogen is weak and oxidation at benzylic position is to break the benzylic
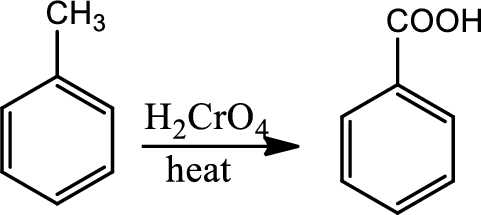
Reaction 21.5:
Halogenation at benzylic position:
Halogenation is regioselective for benzylic position and occurs by a radical chain mechanism. Bromination shows higher regioselectivity for a benzylic position than does chlorination. The reaction occurs via a radical chain mechanism which is initiated when the halohen molecule undergoes homolytic cleavage and gives two halide radical. One of the radical abstracts the benzylic hydrogen to create a resonance stabilised benzylic radical that reacts with another molecule of halogen to give the halogenated product and the new radical continues the chain reaction. NBS is used as the source of bromine.
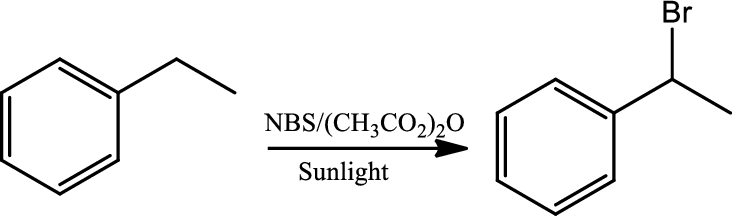
Reaction 21.6:
Hydrogenolysis of benzylic ethers:
Bezylic ethers can be cleaved to give an alcohol or toluene using catalytic hydrogenation of (
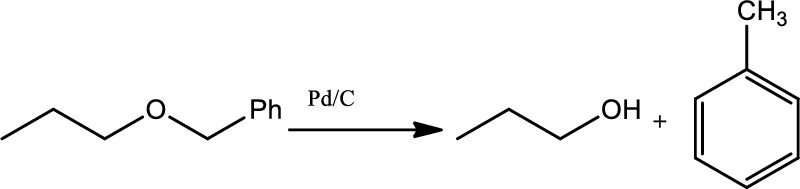
Chapters 22 roadmap reaction legend,
Reaction 22.1:
Halogenation:
In halogenation reaction, aromatic ring reacts with chlorine in the presence of lewis acid catalyst
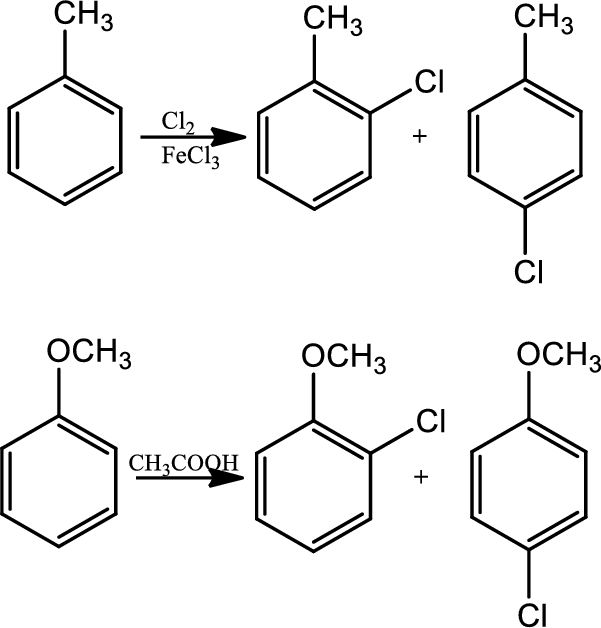
Reaction 22.2:
Nitration:
This is a type of electrophilic substitution on aromatic ring. The electrophile is the nitronium ion
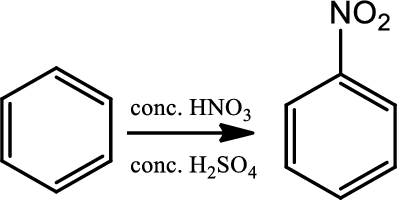
Reaction 22.3:
Sulfonation:
This is a type of electrophilic substitution on aromatic ring. The electrophile is either
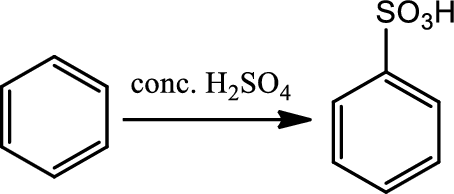
Reaction 22.4:
Fridel-craft’s alkylation:
This is a type of electrophilic substitution on aromatic ring. The electrophile is a carbocation formed as an ion pair interaction of haloalkane with lewis acid. Rearrangements from a less stable carbocation to more stable carbocation are common, the mechanism involves an initial reaction between the haloalkane and lewis acid
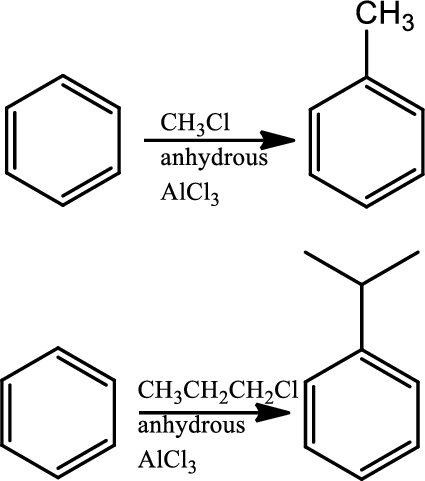
Reaction 22.5:
Fridel-craft’s acylation:
This is a type of electrophilic substitution on aromatic ring. The electrophile is an acylium cation formed as an ion pair by interaction of an acyl halide with a lewis acid. The mechanism involves an initial reaction between the acid chloride and lewis acid
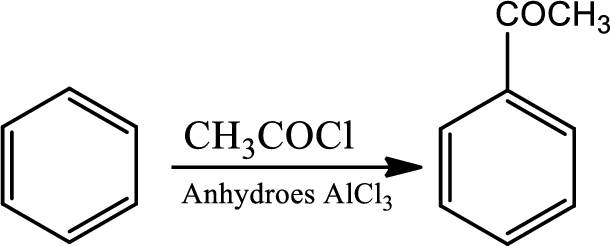
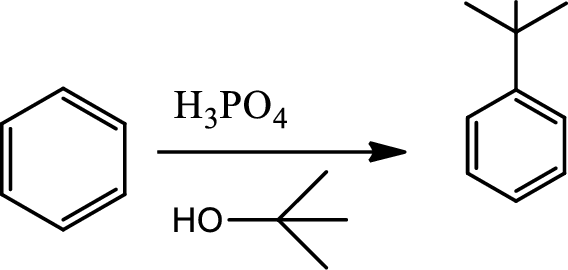
Reaction 22.6:
Alkylation using alkene:
This is a type of electrophilic substitution on aromatic ring. The electrophile is a carbocation fromed by the reaction between the alkene and acids.
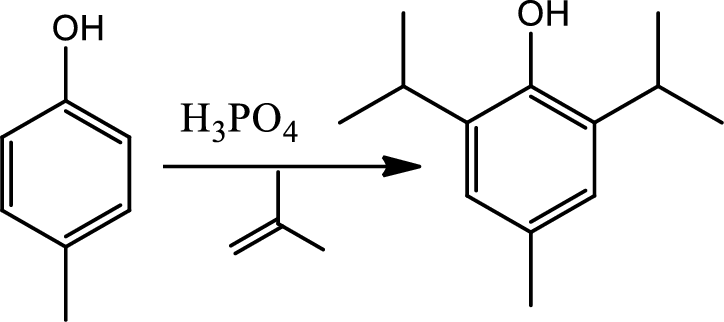
Reaction 22.7:
Alkylation using alcohols:
This is a type of electrophilic substitution on aromatic ring. The electrophile is a carbocation formed from an alcohol reacting with acid.
Chapters 23 roadmap reaction legend,
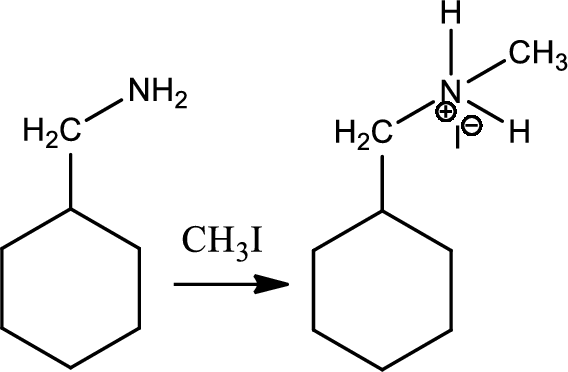
Reaction 23.1:
Alkylation of ammonia and amine:
Alkylation of amine is a type of organic reaction between an alkyl halide and ammonia or amine. This is nucleophilic aliphatic substitution and the reaction product is higher substitued amine. But the problem with the reaction is the overalkylation.
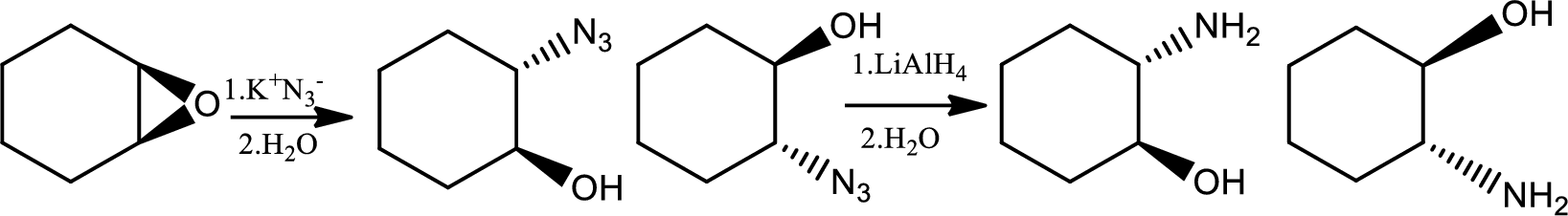
Reaction 23.2:
Alkylation of azide ion followed by reduction:
Azides are prepared by the treatment of a primary or secondary haloakane or an epoxide with
Reaction 23.3:
Nitrosation of tertiary aromatic amines:
This is an electrophilic aromatic substitution. The electrophile here is the nitrosyl cation which is very weak and participates in electrophilic aromatic substitution only with highly activated aromatic rings.
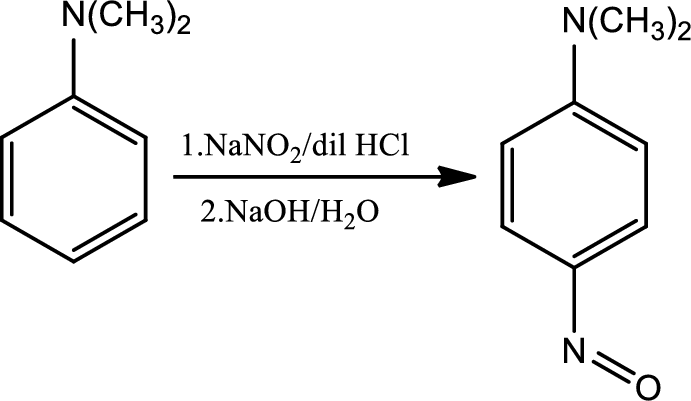
Reaction 23.4:
Formation of N-nitrosamine from secondary amines:
This is an electrophilic aromatic substitution. The electrophile here is the nitrosyl cation which reacts with secondary amine to give N-nitrsoamine.
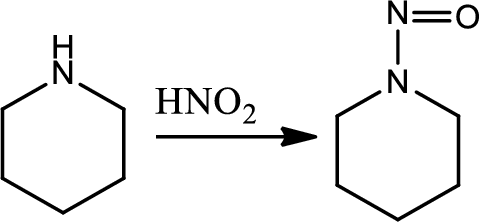
Reaction 23.5:
Diazotisation reaction:
Diazonium salta are organic compounds having
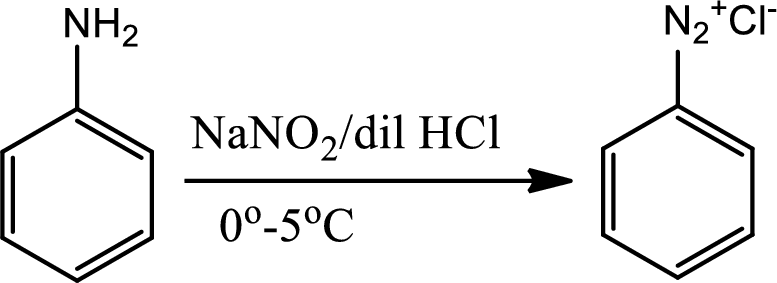
Reaction 23.6:
Conversion of primary aryl amine to phenol:
Formation of an arenediazonium salt followed by loss of nitrogen gives an aryl cation intermediate which then reacts with water to give a phenol.
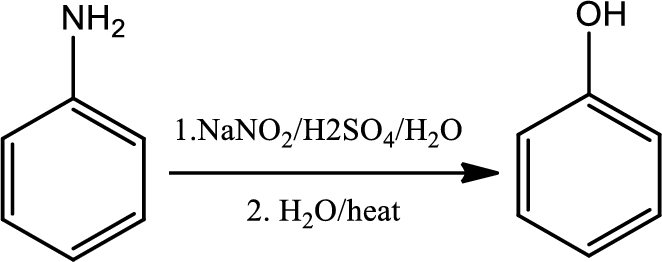
Reaction 23.7:
Schiemann reaction:
The Schiemann reaction is a chemical reaction in which a primary amine is transformed to an aryl fluoride via diazonium tetrafluoroborate intermediate.
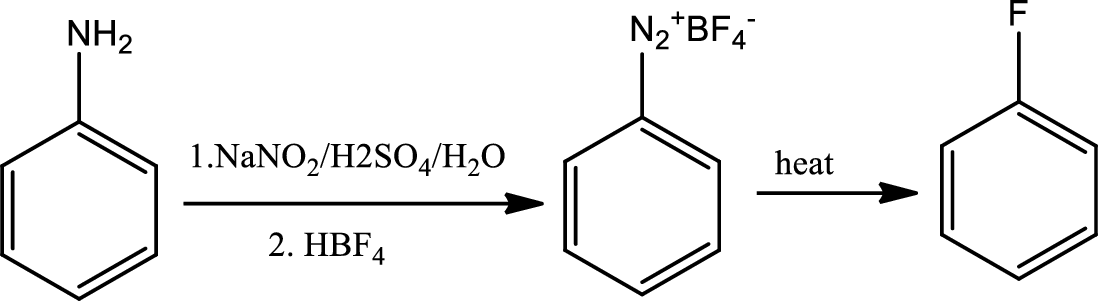
Reaction 23.8:
Sandmeyer reaction:
The Sandmeyer reaction is a chemical reaction used to synthesize aryl halides from aryl diazonium salts using copper salts as reagents or catalysts. It is an example of a radical nucleophilic aromatic substitution. The Sandmeyer reaction provides a method through which the unique transformation on benzene such as halogenation, cyanation, trifluoromethylation and hydroxylations can be done.
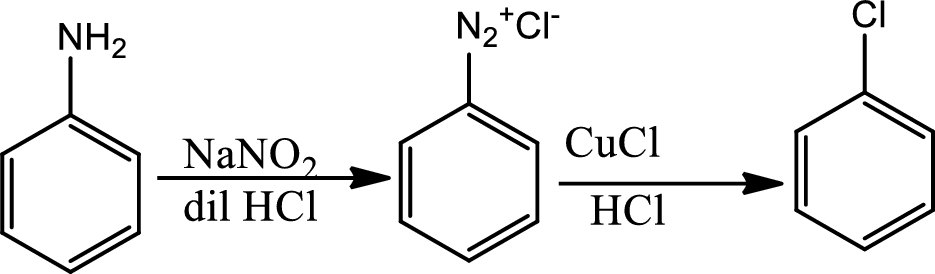
Reaction 23.9:
Reaction of arenediazonium salt with KI:
Reaction of arenediazonium salt with KI is the most convinient way of introducing iodide to benzene ring.
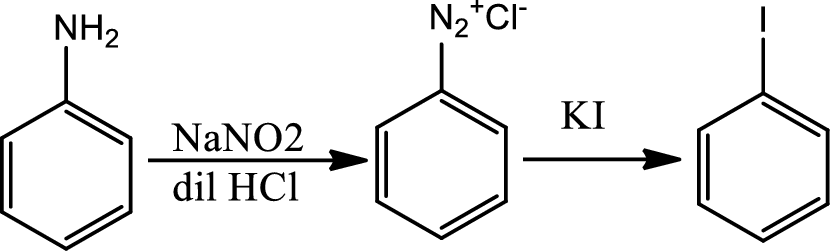
Reaction 23.10:
Reduction of arenediazonium salt with hypophosphorus acid:
By this pathway amine or nitro group can be removed from benzene ring.
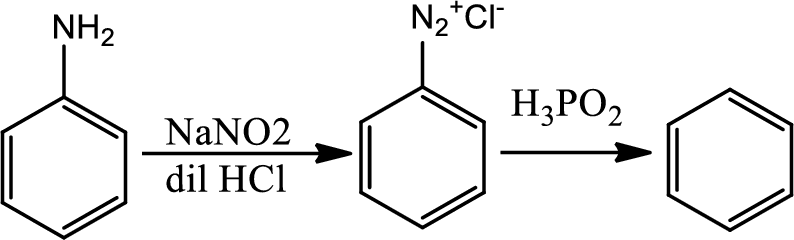
Reaction 23.11:
Hoffmann elimination:
Reaction of a quarternary ammonium halide with moist silver oxide to produce a quarternary ammonium hydroxide followed by heating to give an alkene is a reaction known as Hoffmann elimination. The mechanism involves the simultenous deprotonation of a beta hydrogen by base and los of the amino group in an anti geometry. In this process less substituted alkene is formed.
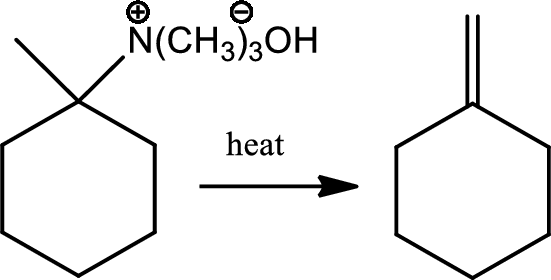
Reaction 23.12:
Cope elimination:
Treatment of the tertiary amine with hydrogen peroxide gives an amine oxide which on heating gives an alkene and an N,N-dialkylhydroxyamine in the reaction is known as Cope elimination. The elimination is syn stereoselective and involves a cyclic flow of six electrons in planar transition state.
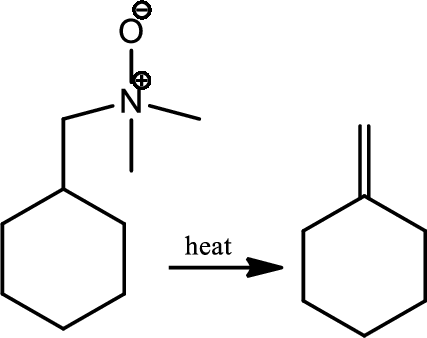
Reaction 23.12:
Reaction of cyclic beta amino alcohol with nitrous acid:
Treating a cyclic beta amino alcohol with nitrous acid leads to rearrangement and a ring expanded ketone. The mechanism involves an initial formation of diazonium ion followed by simulteneous loss of nitrogen and rearrangement by a 1,2-shift to give a resonance stabilised cation that loses a proton to give the product cyclic ketone.
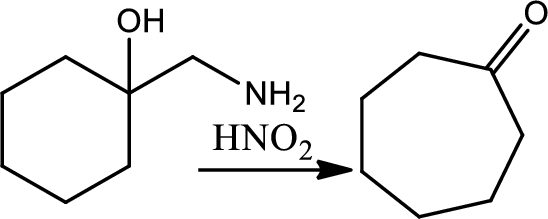
Thus, the reaction roadmap for the reactions in the study Guide section of chapters can be made as shown below for the chapters question 20-23,
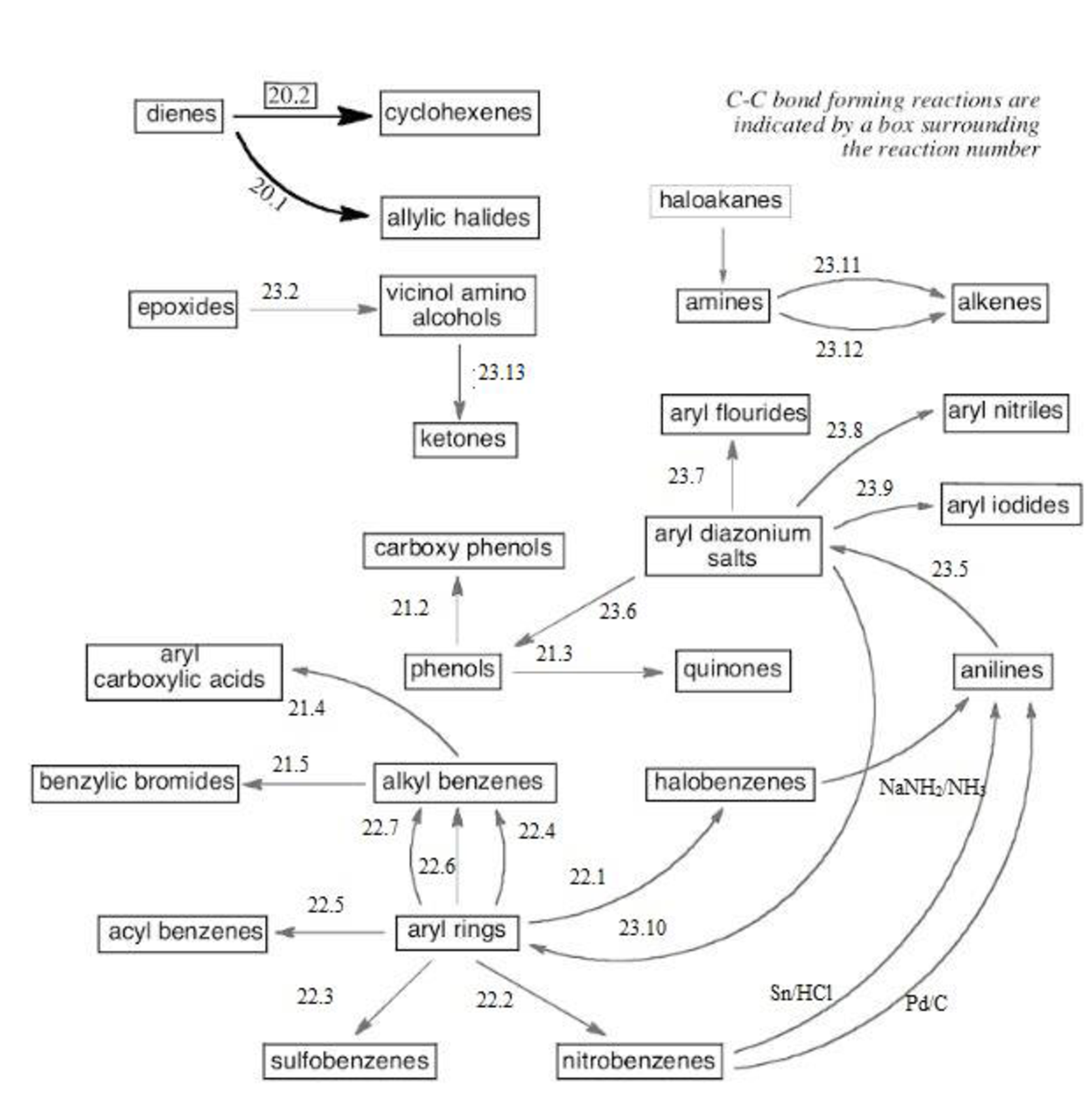
Figure-1
Want to see more full solutions like this?
Chapter 20 Solutions
Organic Chemistry
- 3) A common practice that organic chemists work on is a process known as retrosynthesis. In this a chemist will take known reactants and devise a synthetic pathway to produce the desired products using reactions that they know. As you proceed through organic chemistry you will be tasked with thinking about this process. How would you make the desired final product starting from the alkyne. It will take a couple of reactions. Please show the reagents necessary. And for your last step please show the arrow pushing. Starting material Br Product Br ...arrow_forwardIn this lab you are performing your first organic synthesis and safety information on all the materials you are using must be obtained. Companies are required to provide material safety data sheets (MSDS) for chemical compounds they sell. Please examine the MSDS for acetic anhydride, available here. You will find GHS pictograms within the sheet that summarize the main hazards of this compound. The pictograms are essentially the same as the symbols on WHMIS labels, available here for reference. Based on the pictograms, whatare the main hazards for acetic anhydride? Select as many answers as appropriate however points will be deducted for incorrect guesses. Select one or more: Corrosive Flammable material Acute oral or inhalation toxicity Oxidizing material Carcinogen Self-reactive substance Gas under pressurearrow_forwardWrite the products of the following sequences of reactions. Refer to your reaction roadmaps to see how the combined reactions allow you to navigate between the different functional groups. Note that you will need your old Chapters 611, Chapters 1518, and Chapter 19 roadmaps along with your new Chapter 20 reaction roadmap for these.arrow_forward
- A newer generation of antipsychotics, among them clozapine, are now used to treat the symptoms of schizophrenia. These drugs are more effective than earlier drugs in improving patient response in the areas of social withdrawal, apathy, memory, comprehension, and judgment. They also produce fewer side effects such as seizures and tardive dyskinesia (involuntary body movements). In the following synthesis of clozapine, Step 1 is an Ullmann coupling, a type of nucleophilic aromatic substitution that uses a copper catalyst. (a) Show how you might bring about formation of the amide in Step 2. (b) Propose a reagent for Step 3. (c) Propose a mechanism for Step 4. (d) Is clozapine chiral? If so, how many of the possible stereoisomers are formed in this synthesis?arrow_forwardWe return to classes in person on campus and you enter the organic chemistry lab and find a bottle labelled “Compound X”. Your task is to identify the structure of “Compound X” based on the following reactions that you performed. Be sure to include full justification.arrow_forwardciple Attempts Not allowed. This test can only be taken once. ce Completion Once started, this test must be completed in one sitting. Do not leave the test before clicking Save and Submit This test does not allow backtracking. Changes to the answer after submission are prohibited. emaining Time: 52 minutes, 44 seconds. Question Completion Status: A Moving to the next question prevents changes to this answer. Question 2 Reaction of arenediazonium salts with aryl benzene give.... o Aryl halied Benzonitrile Phenole Azo dayes A Moving to the next question prevents changes to this answer. P Type here to search 1HUAWE F1 F2 F3 F4 F5 F6 F7 %23 24 % & 2 5 6. 7 8. Q W E T Y. us A H. 1S C, Z C VI B U DIarrow_forward
- 2. Show a reaction sequence to convert oil of wintergreen (methyl salicylate) into Aspirin (acetyl salicylic acid). This is a two-step sequence. You must show the structures for the intermediate product and the final product, along with the reagents/reaction conditions. OHarrow_forwardAnswer all three parts plz. You are starting with three aryl bromides. They undergo a typical grignard reaction with magnesium metal and anhydrous ether (diethyl ether). Dry ice is then added to it and it is then cooled. Draw and label the names of the aryl carboxylic acids that will form for each. Part a) bromobenzene Part b) para-bromoanisole Part c) meta-bromoanisolearrow_forwardAlcohols are acidic in nature. Therefore, a strong base can abstract the acidic hydrogen atom of the alcohol in a process known as deprotonation. The alcohol forms an alkoxide ion by losing the proton attached to the oxygen atom of the hydroxyl ( -OH) group. The alkoxide formed can act as a base or a nucleophile depending on the substrate and reaction conditions. However, not all bases can abstract the acidic proton of alcohols and not all alcohols easily lose the proton. Deprotonation depends on the strength of the base and the acidity of the alcohol. Strong bases, such as NaNH2, can easily abstract a proton from almost all alcohols. Likewise, more acidic alcohols lose a proton more easily. Determine which of the following reactions would undergo deprotonation based on the strength of the base and the acidity of the alcohol. Check all that apply. ► View Available Hint(s) CH3CH,OH + NH3 →CH,CH,O-NH CH3 CH3 H3C-C-H+NH3 → H3 C-C-H OH O-NH CH3CH2OH + NaNH, → CH3CH,O-Na* + NH3 CHC12 Cl₂…arrow_forward
- Show a possible stepwise synthesis of 2, 3-dichloropentane starting with ethanol and propanol as the only sources of carbon atoms and any other inorganic reagents, solvents and laboratory equipment. Name the type of reaction that is occurring in each step of the synthesis.arrow_forward1. What are the general instructions before, during, and after experiment, especially in organic chemistry? What are the dos and don’ts in the laboratory? 2. A BSES student, Christian, is working in an organic laboratory for the experiment on the organic synthesis without the guidance of his superlatively handsome instructor, Arwil Nathaniel Alfonso ©. He prepared five organic solvents: 1-pentanol, 2-pentanol, 2-pentanone, benzaldehyde, and m-cresol but he didn't put any label in any of these. Meanwhile, Ryan used the solvents she prepared and placed back in different order. Knowing Christian would punch Ryan in his digestive tract because there is a possible explosion. Prepare a systematic flow chart of qualitative tests to help Ryan to label the solvents before Christian used the organic solvents. 3. Draw the systematic reaction mechanism for between the reaction of isoamyl alcohol and acetic acid. What are the products? What is the functional group of the major product? Does it have…arrow_forwardComplete the following reaction schemes by choosing the correct product, starting material, and/or reagent from the selection below. Your answer should simply be the number that represents your choice. For example, if you believe that the correct answer is the structure represented by the 3, then enter 3 into the correct answer box. Also, answer any additional questions that might be included with a reaction scheme(s). H₂C 0 1. NaNH, 1. NaNH, 2. CH₂CH₂CH₂Br 2. CH₂CH₂Br OH 1 4 3 2 5 O 6 Naº NH3 (1) 11 OH 7 7 XXXXXXXXX E RUTINE H H₂ Pd/C, CaCO3 8 1. NaNHz 2. CH₂(CH₂)₂CH₂Br 13 MCPBA 12 A B a. What reagent(s) is/are required for reaction A to proceed as drawn? What reagent(s) is/are required for reaction B to proceed as drawn? H₂C 9 H₂ Pd/C 14 -0 CH3 Br 10 1. BH-THF 2. H,O2, HỌ, H,O 15arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
