
Concept explainers
Interpretation:
The structural formula for 2-propanol should be drawn and whether it is primary, secondary, or tertiary alcohol should be indicated.
Concept Introduction:
The formula in which the arrangements of atoms in a molecule are shown is said to be the structural formula.
- An organic compound in which hydroxyl
functional group that is -OH is bonded to the carbon atom is said to be an alcohol. The general formula for alcohol is Cn H2 n + 1 OH. Based on the attachment to the carbon the alcohols are classified as primary, secondary, or tertiary alcohol.
When alcohol (having hydroxyl group) is attached to a primary carbon atom (carbon atom to which one carbon atom is attached) then such alcohol is said to be a primary alcohol.
When alcohol (having hydroxyl group) is attached to a secondary carbon atom (carbon atom to which two carbon atoms are attached) then such alcohol is said to be a secondary alcohol.
When alcohol (having hydroxyl group) is attached to a tertiary carbon atom (carbon atom to which three carbon atoms are attached) then such alcohol is said to be a tertiary alcohol.
Answer to Problem 137AP
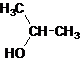
Secondary alcohol.
Explanation of Solution
For number of carbons atoms in chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
The numbering of the carbon chain is done in such a way that the substituents get the lower number.
The given name is 2-propanol that means the parent chain has 3 number of carbons in the chain and suffix -ol represents it is an alcohol group that is R-OH (where R is
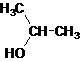
Since, -OH is attached to secondary carbon so, it is a secondary alcohol.
Interpretation:
The structural formula for 2-methyl-2-propanol should be drawn and whether it is primary, secondary, or tertiary alcohol should be indicated.
Concept Introduction:
The formula in which the arrangements of atoms in a molecule are shown is said to be the structural formula.
- An organic compound in which hydroxyl functional group that is -OH is bonded to the carbon atom is said to be an alcohol. The general formula for alcohol is Cn H2 n + 1 OH. Based on the attachment to the carbon the alcohols are classified as primary, secondary, or tertiary alcohol.
When alcohol (having hydroxyl group) is attached to a primary carbon atom (carbon atom to which one carbon atom is attached) then such alcohol is said to be a primary alcohol.
When alcohol (having hydroxyl group) is attached to a secondary carbon atom (carbon atom to which two carbon atoms are attached) then such alcohol is said to be a secondary alcohol.
When alcohol (having hydroxyl group) is attached to a tertiary carbon atom (carbon atom to which three carbon atoms are attached) then such alcohol is said to be a tertiary alcohol.
Answer to Problem 137AP
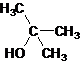
Tertiary alcohol.
Explanation of Solution
For number of carbons atoms in chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
The numbering of the carbon chain is done in such a way that the substituents get the lower number.
The given name is 2-methyl-2-propanol that means the parent chain has 3 number of carbons in the chain and suffix -ol represents it is an alcohol group that is R-OH (where R is aryl/alkyl group) at 2 position, and a methyl substitution at 2 position. So, the structural formula of 2-methyl-2-propanol is:
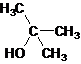
Since, -OH is attached to tertiary carbon so, it is a tertiary alcohol.
Interpretation:
The structural formula for 4-isopropyl-2-heptanol should be drawn and whether the it is primary, secondary, or tertiary alcohol should be indicated.
Concept Introduction:
The formula in which the arrangements of atoms in a molecule are shown is said to be the structural formula.
- An organic compound in which hydroxyl functional group that is -OH is bonded to the carbon atom is said to be an alcohol. The general formula for alcohol is Cn H2 n + 1 OH. Based on the attachment to the carbon the alcohols are classified as primary, secondary, or tertiary alcohol.
When alcohol (having hydroxyl group) is attached to a primary carbon atom (carbon atom to which one carbon atom is attached) then such alcohol is said to be a primary alcohol.
When alcohol (having hydroxyl group) is attached to a secondary carbon atom (carbon atom to which two carbon atoms are attached) then such alcohol is said to be a secondary alcohol.
When alcohol (having hydroxyl group) is attached to a tertiary carbon atom (carbon atom to which three carbon atoms are attached) then such alcohol is said to be a tertiary alcohol.
Answer to Problem 137AP
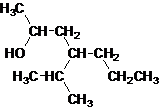
Secondary alcohol.
Explanation of Solution
For number of carbons atoms in chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
The numbering of the carbon chain is done in such a way that the substituents gets the lower number.
The given name is 4-isopropyl-2-heptanol that means the parent chain has 7 number of carbons in the chain and suffix -ol represents it is an alcohol group that is R-OH (where R is aryl/alkyl group) at 2 position, and anisopropyl substitution at 4 position. So, the structural formula of 4-isopropyl-2-heptanol is:
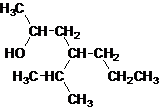
Since, -OH is attached to secondary carbon so, it is a secondary alcohol.
Interpretation:
The structural formula for 2, 3-dichloro-1-pentanol should be drawn and whether the it is primary, secondary, or tertiary alcohol should be indicated.
Concept Introduction:
The formula in which the arrangements of atoms in a molecule are shown is said to be the structural formula.
- An organic compound in which hydroxyl functional group that is -OH is bonded to the carbon atom is said to be an alcohol. The general formula for alcohol is Cn H2 n + 1 OH. Based on the attachment to the carbon the alcohols are classified as primary, secondary, or tertiary alcohol.
When alcohol (having hydroxyl group) is attached to a primary carbon atom (carbon atom to which one carbon atom is attached) then such alcohol is said to be a primary alcohol.
When alcohol (having hydroxyl group) is attached to a secondary carbon atom (carbon atom to which two carbon atoms are attached) then such alcohol is said to be a secondary alcohol.
When alcohol (having hydroxyl group) is attached to a tertiary carbon atom (carbon atom to which three carbon atoms are attached) then such alcohol is said to be a tertiary alcohol.
Answer to Problem 137AP
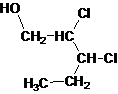
Primary alcohol.
Explanation of Solution
For number of carbons atoms in chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
The numbering of the carbon chain is done in such a way that the substituents get the lower number.
The given name is 2, 3-dichloro-1-pentanol that means the parent chain has 5 number of carbons in the chain and suffix -ol represents it is an alcohol group that is R-OH (where R is aryl/alkyl group) at 1 position, and 2 chlorine substitution at 2 and 3 position. So, the structural formula of 2, 3-dichloro-1-pentanol is:
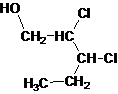
Since, -OH is attached to primary carbon so, it is a primary alcohol.
Want to see more full solutions like this?
Chapter 20 Solutions
Introductory Chemistry: A Foundation
- The general formula of an alkane is CnH2n+2 . What is the general formula of an (a) alkene? (b) alkyne? (c) alcohol derived from an alkane?arrow_forwardWhat functional group distinguishes each of the following hydrocarbon derivatives? a. halohydrocarbons b. alcohols c. ethers d. aldehydes e. ketones f. carboxylic acids g. esters h. amines Give examples of each functional group. What prefix or suffix is used to name each functional group? What are the bond angles in each? Describe the bonding in each functional group. What is the difference between a primary, secondary, and tertiary alcohol? For the functional groups in ah, when is a number required to indicate the position of the functional group? Carboxylic acids are often written as RCOOH. What does COOH indicate and what does R indicate? Aldehydes are sometimes written as RCHO. What does CHO indicate?arrow_forward4. Draw structures for the following alcohols. a. butane-1,2-diol 2,4-dimethylcyclopentanol C. b. d. 3-ethyl-4-methyloctan-1-ol propane-1,2,3-triolarrow_forward
- Which of the following alcohols can be oxidized to a carboxylic acid? Name the carboxylic acid produced. Forthose alcohols that cannot be oxidized to a carboxylic acid,name the final product.a. Ethanol c. 1-Propanolb. 2-Propanol d. 3-Pentanolarrow_forwardWrite the condensed structural formula of the following alcohols and their classification. e. 3-ethyl-2-pentanol d. 2,2-dimethyl-1-hexanol f. 3,5-dimethylcylohexanolarrow_forwardWhat is the correct IUPAC name of neopentyl alcohol O A. pentan-1-ol B. methyl pentan-2-ol O C. 2,2-dimethyl propanol O D. tert- butyl methanolarrow_forward
- A. Draw the structure of each of the following alcohols. Then draw and name the product you would expect to produce by the oxidation of each. a. 4-Methyl-2-heptanol b. 3,4-Dimethyl-1-pentanol c. 4-Ethyl-2-heptanol d. 5,7-Dichloro-3-heptanolarrow_forward1. Draw a simple organic molecule that contain the following functional groups. a. Cycloalkane d. Ester b. Amine c. Ether e. Aldehyde f. Ketonearrow_forwardDraw the structural formulas for each of the following organic compounds. Circle the non- alkyl functional groups a. hexan-2-one b. 2- methylpentanal c. Pentane-1,3-diol d. buta-1,3-diene e. 1-propoxybutane f. ethyl ethanoatearrow_forward
- Draw the condensed structure of each of the following alcohols. Then draw and name the product you would expect to produce by the oxidation of each. a. 4-Methyl-2-heptanol b. 3,4-Dimethyl-1-pentanol c. 4-Ethyl-2-heptanol d. 5,7-Dichloro-3-heptanolarrow_forwardConsider the following starting material and choose all of the functional groups that are likely to oxidize in it. a. aldehyde b. secondary alcohol c. alkane d. hemiacetal e. carboxylic acid f. alkenearrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





