
(a)
Interpretation:
The synthesis of acetoacetic ester can be used to prepare 5-phenyl-2,5-pentanedione has to be showed.
Concept Introduction:
Acetoacetic ester is a versatile starting material for the formation of new carbon-carbon bonds. It is because of the presence of acidic α-hydrogens between two carbonyl groups, the nucleophilicity of the enolate anion formed by the loss of α-hydrogen and the ability of the product to undergo decarboxylation after hydrolysis of ester.
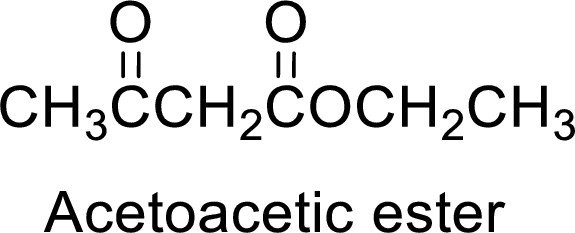
(b)
Interpretation:
The synthesis of acetoacetic ester can be used to prepare 1-cyclopentyl-1-ethanone has to be showed.
Concept Introduction:
Acetoacetic ester is a versatile starting material for the formation of new carbon-carbon bonds. It is because of the presence of acidic α-hydrogens between two carbonyl groups, the nucleophilicity of the enolate anion formed by the loss of α-hydrogen and the ability of the product to undergo decarboxylation after hydrolysis of ester.
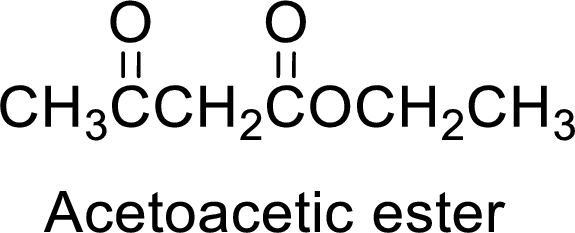
(c)
Interpretation:
The synthesis of acetoacetic ester can be used to prepare 3-ethyl-2-pentanone has to be showed.
Concept Introduction:
Acetoacetic ester is a versatile starting material for the formation of new carbon-carbon bonds. It is because of the presence of acidic α-hydrogens between two carbonyl groups, the nucleophilicity of the enolate anion formed by the loss of α-hydrogen and the ability of the product to undergo decarboxylation after hydrolysis of ester.
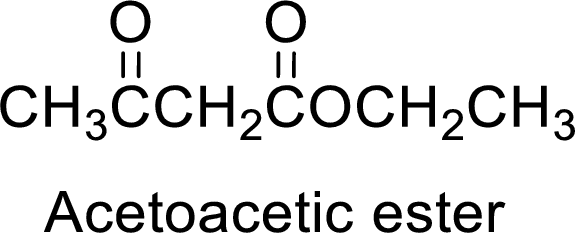
Trending nowThis is a popular solution!

Chapter 19 Solutions
Organic Chemistry
- Predict the products formed when cyclohexanone reacts with the following reagents.phenylhydrazine and weak acidarrow_forwardHow imines and enamines are converted back to carbonyl compounds by hydrolysis with mild acid ?arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Part A Given 7.30 gg of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%% yield? Express your answer in grams to three significant figures. Part B A chemist ran the reaction and obtained 5.95 gg of ethyl butyrate. What was the percent yield? Express your answer as a percent to three significant figures. Part C The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0%% yield. How many grams would be produced from 7.30 gg of…arrow_forward
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.45 gg of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%% yield? Express your answer in grams to three significant figures. A chemist ran the reaction and obtained 5.50 gg of ethyl butyrate. What was the percent yield? Express your answer as a percent to three significant figures. The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0%% yield. How many grams would be produced from 8.45 gg of butanoic acid and excess…arrow_forwardPrepare the following compounds starting from benzaldehyde and the appropriate ketone. Provide reactions for preparing the ketones starting from aromatic hydrocarbon compounds.arrow_forwardIndicate the reagents and draw the intermediate products necessary to accomplish the following transformations.arrow_forward
- Name the carbonyl compound that would be formed by the complete acidic hydrolysis of the following hemiacetal/hemiketal or acetal/ketal: OH OCH₂CH₂CH₂CH₂CH3arrow_forwardPredict the products formed when cyclohexanone reacts with the following reagents.ethylene glycol and p-toluenesulfonic acidarrow_forwardComplete the following reaction pathways with phenolarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
