
Concept explainers
(a)
Interpretation:
The products of hydrolysis of given triacylglycerol needs to be determined.
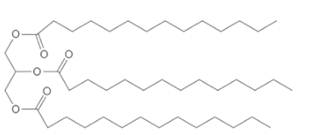
Concept Introduction:
Lipids are
Triacylglycerols are most abundant lipids which are mainly found in animal fat and vegetable oils. They are triesters of glycerol, therefore in the formation of one molecule of triacylglycerol, three molecules of fatty acids react with one molecule of glycerol as given below;
(b)
Interpretation:
The products of hydrolysis of given triacylglycerol needs to be determined.
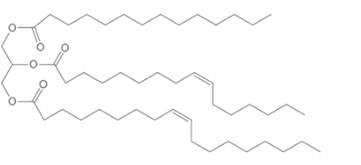
Concept Introduction:
Lipids are biomolecules which are involved in different biochemical reactions. They are special types of organic molecules which can only identify with the help of their physical properties, not by the presence of any certain functional group. In general, lipids contain a large number of C-C and C-H bonds with few polar functional groups such as
Triacylglycerols are most abundant lipids which are mainly found in animal fat and vegetable oils. They are triesters of glycerol, therefore in the formation of one molecule of triacylglycerol, three molecules of fatty acids react with one molecule of glycerol as given below;
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Draw the structure of each type of compound. a. a D-aldotriose b. an L-ketohexose c. a four-carbon aldonic acidarrow_forwardDraw the products formed when triacylglycerol A is treated with each reagent. Rank compounds A, B, and C in order of increasing melting point.a. H2O, H+b. H2 (excess), Pd-C → Bc. H2(1 equiv), Pd-C → Carrow_forwardChemical structure lactams are: A. Phenobarbital B. Ampicillin C. Piracetam D. Ascorbic acidarrow_forward
- 1. Draw the structure of a triacylglycerol formed from glycerol, one molecule of stearic acid, and two molecules of oleic acid. The stearic acid should form an ester bond with the 2° OH group of glycerolarrow_forwardIdentify the major class of each lipid from its components. a. fatty acids + glycerol b. fatty acids + sphingosine + a carbohydrate c. fatty acids + high-molecular-weight alcohols other than glycerol d. glycerol + fatty acids + phosphate group + choline e. fatty acids + sphingosine + phosphate group + a nitrogen compoundarrow_forwardWhy metals are deleterious to the stability of unsaturated fatty acids? I. They initiate free radicals which lead to autoxidation of the fatty acids II. They bind to the double bonds and create an undesirable metallic taste III. They cause the formation of dimers of two or more fatty acids which changes the melting point IV. They will solidify the unsaturated fatty acid а. I and III b. I, II, III and IV c. I only d. Il and IVarrow_forward
- Fatty acids are distributed throughout the body, via a time-release/transport molecule created by: a. an esterification reaction with glycerol b. a hydrolytic reaction with methylamine c. an alkylation reaction with butanol d. a carboxylation reaction with lactic acid e. an ether formation reaction with ethylene glycolarrow_forwardwhat products are formed when cholesterol is reacted with bromine in organic solventarrow_forwardWhat glycosidic bond is present?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


