
(a)
Interpretation:
The conversion of hexanoic acid to given amine has to be shown.
Concept introduction:
Acid chlorides are most often prepared by treating a
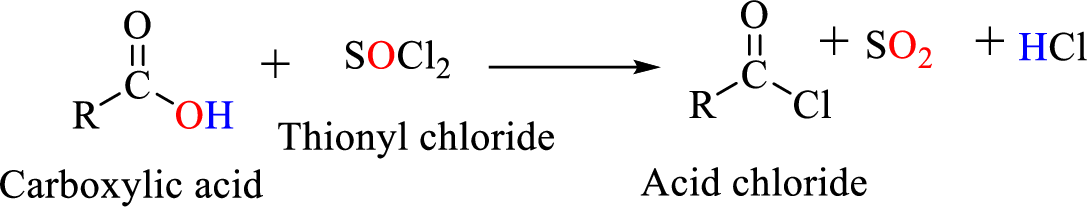
Reaction of acid chloride with ammonia or
The reaction of acid chlorides with ammonia and primary and secondary amines will yield amide.
The nucleophilic addition of ammonia or amine to the carbonyl carbon occurs which is followed by a proton transfer forming an addition intermediate that will eliminate chloride and lose a proton to give the amide compound.
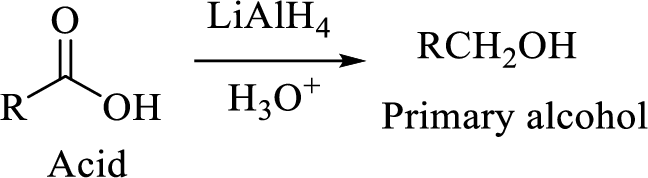
(b)
Interpretation:
The conversion of hexanoic acid to given amine has to be shown.
Concept introduction:
Acid chlorides are most often prepared by treating a carboxylic acid with thionyl chloride.
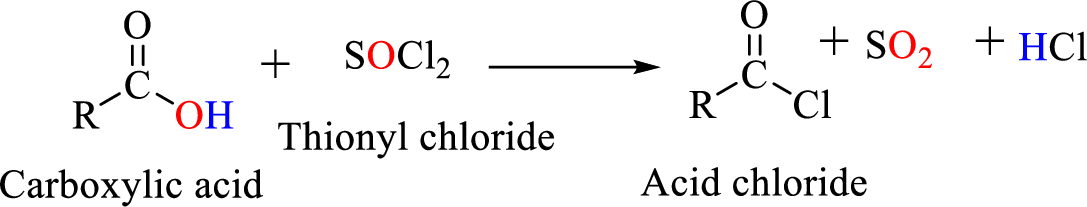
Reaction of acid chloride with ammonia or amines:
The reaction of acid chlorides with ammonia and primary and secondary amines will yield amide.
The nucleophilic addition of ammonia or amine to the carbonyl carbon occurs which is followed by a proton transfer forming an addition intermediate that will eliminate chloride and lose a proton to give the amide compound.
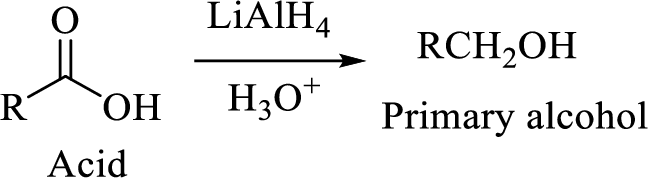
Trending nowThis is a popular solution!

Chapter 18 Solutions
Organic Chemistry
- Give an acceptable name for each amine.arrow_forwardA) Name the following amine. H3C−CH2−CH2−NH−CH2−CH2−CH3 Spell out the full name of the compound. B ) Name the following amine. CH3−CH2−NH−CH2−CH2−CH3 Spell out the full name of the compound.arrow_forward1. Which statement best described the ability of amines to hydrogen bond? A. Primary, secondary and tertiary amines can all hydrogen bond with molecules identical to themselves. B. Primary and secondary amines can hydrogen bond with molecules identical to themselves, but tertiary amines cannot. C. Only primary amines can hydrogen bond with molecules identical to themselves. Secondary and tertiary amines cannot. D. Primary, secondary and tertiary amines cannot hydrogen bond with molecules identical to themselves, but the can hydrogen bond with water.arrow_forward
- The presence of amides in living organisms is beneficial due its stability which results from being the least reactive carboxylic acid derivative. True or Falsearrow_forward4. Draw the following molecules. 3-(N-methylamino)butanal p-N,N-diethylaminophenol o-ethoxyaniline (R)-butan-2-amine N-cyclopentyl-N-tert-butyloctanaminearrow_forwardDraw the major product of this reaction. Ignore inorganic byproducts and the amine side product. 1. NaOH, heat N 1 2. Neutralizing work- uparrow_forward
- chyl-4-dimethylamine chyl-N,N-dimethylpentyl-2-amine chyl-N,N-dimethylpentyl-2-amine chyl-N,N-dimethylpentan-2-aminearrow_forwardWhat steps would be used to synthesize an organic amine? Group of answer choices Ketone + oxidizing agent →carboxylic acid + ammonia →organic amine Alkene + acid → alcohol + ammonia →organic amine Ether + acid →ester + ammonia →organic amine Alkane + acid → alcohol + ammonia →organic amine Carboxylic acid + alcohol →ester + ammonia →organic amine Aldehyde + oxidizing agent →carboxylic acid + ammonia → organic aminearrow_forwardThe hydrolysis of an amide in acidic conditions forms A. a carboxylate salt and an alcohol B. a carboxylate salt and an amine C. an alcohol and an amine salt (an ammonium ion) D. a carboxylic acid and an amine salt (an ammonium ion)arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning




