
Concept explainers
(a)
Interpretation:
The complete mechanism that proceeds when the given species treated with triphenylphosphine followed by butyllithium is to be drawn.
Concept introduction:
The Wittig reagents can be generated from
Answer to Problem 17.53P
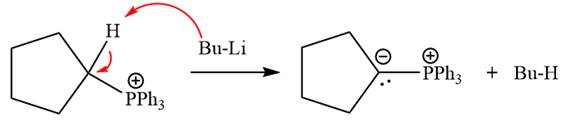
The complete mechanism of the given alkyl halide is treated with triphenylphosphine followed by butyllithium as shown below.
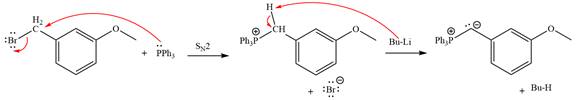
Explanation of Solution
The given alkyl halide is
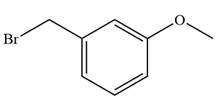
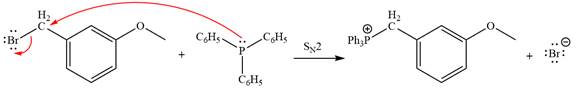
The Wittig reagent is generated from the given alkyl halide and triphenylphosphine followed by butyllithium. Triphenylphosphine,
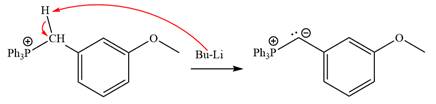
Lastly, the product obtained from the first step is treated with butyllithium to form Wittig reagent.
The complete mechanism of the given alkyl halide is treated with triphenylphosphine followed by butyllithium as shown below.
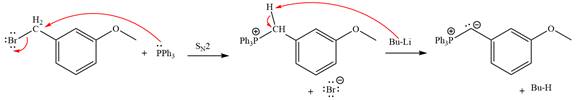
The complete mechanism for the given species treated with triphenylphosphine followed by butyllithium is drawn.
(b)
Interpretation:
The complete mechanism that proceeds when the given species treated with triphenylphosphine followed by butyllithium is to be drawn.
Concept introduction:
The Wittig reagents can be generated from alkyl halides. The alkyl halide is first reacted with triphenylphosphine and the product of that reaction is treated with a strong base. In the first step reaction fovors
Answer to Problem 17.53P
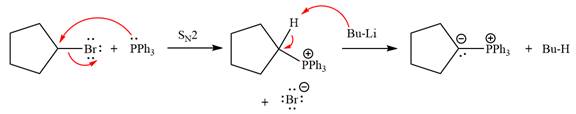
The complete mechanism of the given alkyl halide is treated with triphenylphosphine followed by butyllithium as shown below.
Explanation of Solution
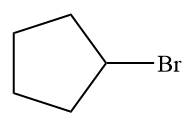
The given alkyl halide is
The Wittig reagent is generated from the given alkyl halide and triphenylphosphine followed by butyllithium. Triphenylphosphine,
Lastly, the product obtained from the first step is treated with butyllithium to form Wittig reagent.
The complete mechanism of the given alkyl halide is treated with triphenylphosphine followed by butyllithium as shown below.
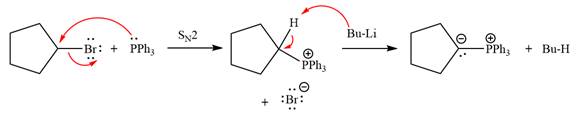
The complete mechanism for the given species treated with triphenylphosphine followed by butyllithium is drawn.
(c)
Interpretation:
The complete mechanism that proceeds when the given species treated with triphenylphosphine followed by butyllithium is to be drawn.
Concept introduction:
The Wittig reagents can be generated from alkyl halides. The alkyl halide is first reacted with triphenylphosphine and the product of that reaction is treated with a strong base. In the first step reaction fovors
Answer to Problem 17.53P
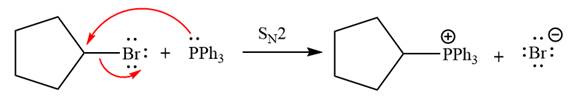
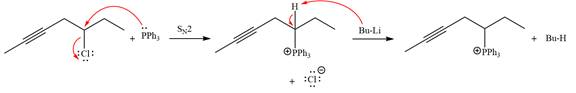
The complete mechanism of the given alkyl halide is treated with triphenylphosphine followed by butyllithium as shown below.
Explanation of Solution
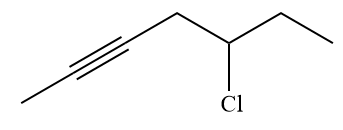
The given alkyl halide is
The Wittig reagent is generated from the given alkyl halide and triphenylphosphine followed by butyllithium. Triphenylphosphine,
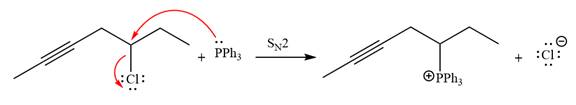
After that, the product obtained from the first step is treated with butyllithium to form Wittig reagent.
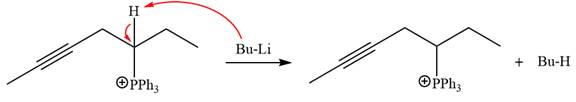
The complete mechanism of the given alkyl halide is treated with triphenylphosphine followed by butyllithium as shown below.
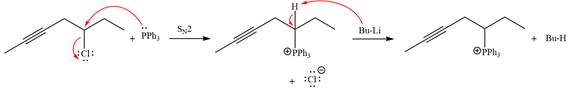
The complete mechanism for the given species treated with triphenylphosphine followed by butyllithium is drawn.
Want to see more full solutions like this?
Chapter 17 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Alkyl diazonium salts decompose to form carbocations, which go on to form products of substitution, elimination, and (sometimes) rearrangement. Keeping this in mind, draw a stepwise mechanism that forms all of the following products.arrow_forwardIdentify compounds A,C,I in the following reaction sequences.arrow_forwardDraw a complete detailed mechanism for this reaction.arrow_forward
- When a single compound contains both a nucleophile and a leavinggroup, an intramolecular reaction may occur. With this in mind, draw theproduct of the following reaction.arrow_forwardDraw a step wise, detailed mechanism for the following reaction.arrow_forwardPlease draw detailed mechanism of this reaction. Picture are attached:arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
