
(a)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
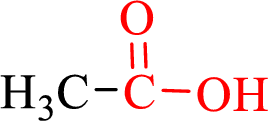
Nomenclature of
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the
alkane name with –oic acid.
Naming of compounds with two
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
Carboxylic salts are the water-soluble ammonium or alkali metal salts of carboxylic acids.
(a)
Explanation of Solution
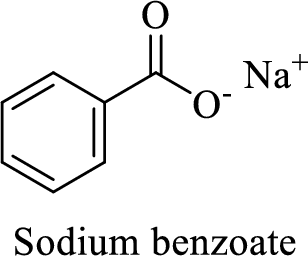
Name of the given salt is sodium benzoate.
From the name, we will get the following fact about the structure of the compound.
- ✓ The given compound is a sodium salt of benzoic acid.
Thus,
The structural formula for this salt can be drawn as shown below,
(b)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
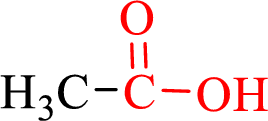
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid. If two or more carboxylic functional groups are present in the same compound then its number should be taken in to consideration and the prefix di, tri, tetra.. must be used.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
Carboxylic salts are the water-soluble ammonium or alkali metal salts of carboxylic acids.
(b)
Explanation of Solution
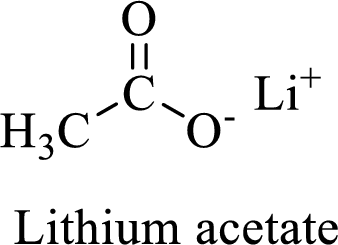
Name of the given salt is Lithium acetate.
From the name, we will get the following fact about the structure of the compound.
- ✓ The given compound is a Lithium salt of acetic acid
Thus,
The structural formula for this compound can be drawn as shown below,
(c)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
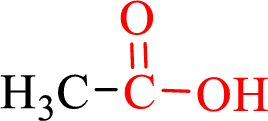
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
Carboxylic salts are the water-soluble ammonium or alkali metal salts of carboxylic acids.
(c)
Explanation of Solution
Name of the given salt is Ammonium acetate.
From the name, we will get the following fact about the structure of the compound.
- ✓ The given compound is an ammonium salt of acetic acid.
Thus,
The structural formula for this compound can be drawn as shown below,
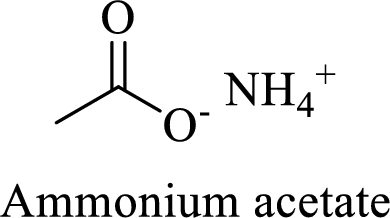
(d)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
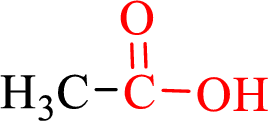
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
Carboxylic salts are the water-soluble ammonium or alkali metal salts of carboxylic acids.
(d)
Explanation of Solution
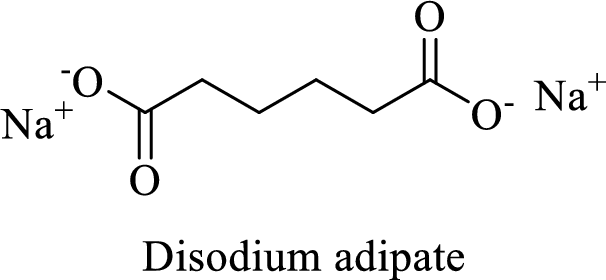
Name of the given salt is Disodium adipate.
From the name, we will get the following facts about the structure of the compound.
- ✓ The given compound is a sodium salt of adipic acid.
- ✓ Two sodium atoms are attached to the both carboxyl group of adipic acid.
The structure of adipic acid is shown below,
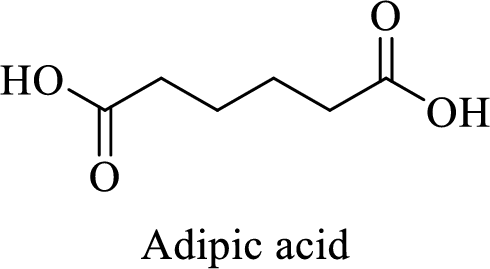
Thus,
The structural formula for this compound can be drawn as shown below,
(e)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
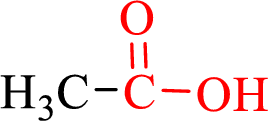
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
Carboxylic salts are the water-soluble ammonium or alkali metal salts of carboxylic acids.
(e)
Explanation of Solution
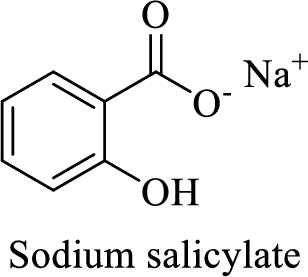
Name of the given salt is sodium salicylate.
From the name, we will get the following fact about the structure of the compound.
- ✓ The compound is a sodium salt of salicylic acid.
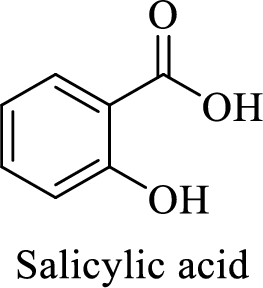
Structure of salicylic acid is shown below,
Thus,
The structural formula for this compound can be drawn as shown below,
(f)
Interpretation: The structural formula for the given compound has to be drawn.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
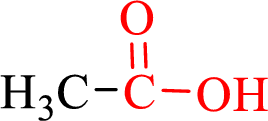
Nomenclature of carboxylic acid:
- • Find the Parent hydrocarbon chain.
- • Carboxyl carbon must be numbered first.
- • Replace the –e in the alkane name with –oic acid. If two or more carboxylic functional groups are present in the same compound then its number should be taken in to consideration and the prefix di, tri, tetra.. must be used.
Naming of compounds with two functional groups;
If a compound has two functional groups, the one with lower priority is indicated by a prefix and another with the higher priority by a suffix.
Carboxylic salts are the water-soluble ammonium or alkali metal salts of carboxylic acids.
(f)
Explanation of Solution
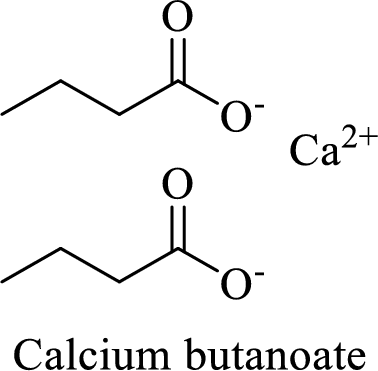
Name of the given salt is calcium butanoate.
From the name, we will get the following fact about the structure of the compound.
- ✓ The given compound is a calcium salt of butanoic acid.
Thus,
The structural formula for this compound can be drawn as shown below,
Want to see more full solutions like this?
Chapter 17 Solutions
Organic Chemistry
- (c) NH2 Draw Your Solution HA (cat.)arrow_forwardHow could you distinguish NH4Cl and AgCL? (a) By smell (b) By color (c) By solubility in water (d) By tastearrow_forwardCalculate the equilibrium constant for the acid–base reaction between the reactants in each of the following pairs: (a) HCl + H2O (b) CH3COOH + H2O (c) CH3NH2 + H2O (d) CH3N+H3 + H2Oarrow_forward
- Identify each of the following oxides as an acid or baseanhydride. Write the chemical formula and give the nameof the acid or base formed upon reaction with water.(a) MgO (b) Cl2O (c) SO3 (d) Cs2Oarrow_forwardPhenylamine is an aromatic amine that is used in the manufacture of dyes. When absorbed through the skin itcauses the Fe+2 in hemoglobin to become oxidized into Fe+3, resulting in the formation of methemoglobin whichcannot bind to or transport oxygen. Phenylamine is soluble in water and acts as a weak base.C6H5NH2 (aq) + H2O (ℓ) ⇋ C6H5NH3+ (aq) + OH- (aq)a. When you measure the concentrations of the ionized substances you find them to be:[C6H5NH2] = 0.234 mol/L [C6H5NH3+] = 2.8 x 10-5 mol/L [OH-]= 2.8 x 10-5 mol/LIf the Kb is 4.27 x 10-10, is the reaction at equilibrium? If not, which direction does it need to move (rightor left) to reach equilibrium? Explain. b. At equilibrium the concentrations of the ionized substances are:[C6H5NH2] = 0.0537 mol/L [C6H5NH3+] = 4.79 x 10-6 mol/L [OH-]= 4.79 x 10-6 mol/LIf this reaction is taking place in a 2.0L container, and 1.5 moles of phenylamine were added to thereaction, what will the new concentrations of the three ionic species be when…arrow_forward(a) When acetone dissolves in water, what major species are present? Write structures, cations or anions separated by comma. (b) When potassium bromide dissolves in water, what major species are present? Write structures, cations or anions separated by comma. (c) When lithium hydroxide dissolves in water, what major species are present? Write structures, cations or anions separated by comma. (d) When sulphuric acid dissolves in water, what major species are present? Write structures, cations or anions separated by comma.arrow_forward
- (a) Hydrogen peroxide, H2O2, is a Brønsted Lowry acid. It is used as an antiseptic and bleaching agent. Write the formula for the conjugate base of hydrogen peroxide. (b) Hydrazine, N2H4, is a Brønsted-Lowry base used as a rocket fuel. Write the formula for the conjugate acid of hydrazine. (c) Phenol, HOC6H5, is a Brønsted-Lowry acid used to make plastics, nylon, and slimicides. Write the formula for its conjugate base. (d) Aniline, C6H5NH2, is a Brønsted-Lowry base used to make polyurethane. Write the formula for its conjugate acid.arrow_forwardBenzoic acid (C6H5COOH) and aniline (C6H5NH2) areboth derivatives of benzene. Benzoic acid is an acid withKa = 6.3 x10-5 and aniline is a base with Ka = 4.3 x10-10. (a) What are the conjugate base of benzoic acid andthe conjugate acid of aniline? (b) Anilinium chloride(C6H5NH3Cl) is a strong electrolyte that dissociates intoanilinium ions (C6H5NH3+) and chloride ions. Which willbe more acidic, a 0.10 M solution of benzoic acid or a 0.10M solution of anilinium chloride? (c) What is the value ofthe equilibrium constant for the following equilibrium?C6H5COOH(aq) + C6H5NH2(aq) ⇌ C6H5COO-(aq) + C6H5NH3+(aq)arrow_forwardSelect the correct answers: “Ocean acidification is a process that occurs when atmospheric carbon dioxide dissolves in the ocean and is subsequently converted into (methane; carbonic acid; carbonate ions; sodium carbonite) . This then causes the pH of the ocean to (increase; decrease; fractionate; multiply) and become more acidic.”arrow_forward
- Provide explanation of correct and incorrect option. please do not provide solution based on AI.arrow_forwardWhat is the basis for referring to the hydrogen ion as a proton? (a) The Arrhenius definition of an acid. (b) The Brønsted-Lowry definition of an acid x (c) The Lewis definition of an acid. (d) H* exists as a bare proton. (e) None of these.arrow_forward(a) Predict the products of the following acid-base reactions using curved-arrow mechanisms to indicate electron flow. (b) Indicate the acid, base, conjugate acid, and conjugate base of each reaction. (c) Indicate whether the reactants or products are favored at equilibrium a) CH,COOH CH3O b) CH,CH,OH H2Narrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
