
Interpretation: The overall charge of a nonpolar amino acid at a pH below its pI.
Concept introduction:
➢ Amino acid: Any molecule contains both amino and acid group
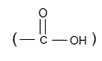
Ex:
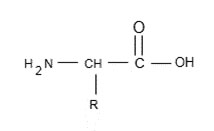
➢ The group can be simple as hydrogen atom (glycine) to more complicated groups such as alcohols,
➢ If is non polar
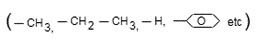
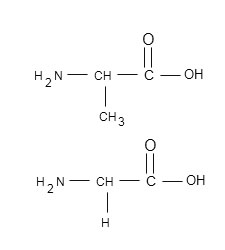
Then it is called non polar amino acid.
Ex:
The amino acid has two group,
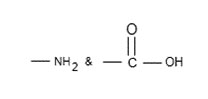
➢ One acts as base and other acts as acid. So net charge or from of amino acid depends upon value as follows:
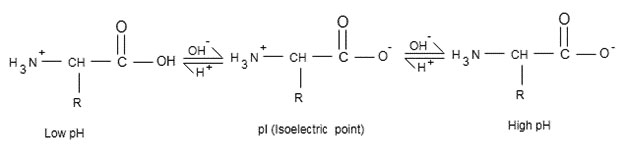
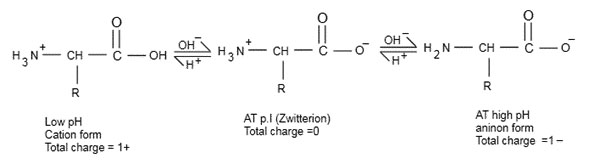
➢ Amino acid is any molecule that contains both amino and acid group. If then net charge on amino acid is zero called zwitterion from contains equal no of charge.
➢ If , then at this acts as acid and donates one proton to for
And it has total charge
➢ If , then
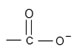 Acts as base and accept one proton form solution and convert to
Acts as base and accept one proton form solution and convert to
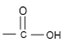 So at such it has total charge
So at such it has total charge
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





