
Concept explainers
Explain the following phenomenon: You have a test tube with an aqueous solution of silver nitrate as shown in test tube l below. A few drops of aqueous sodium chromate solution was added with the end result shown in test tube 2. A few drops of aqueous sodium chloride solution was then added with the end result shown in test tube 3.
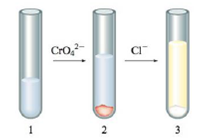
Use the Ksp values in the book to support your explanation, and include the balanced equations. Also, list the ions that are present in solution in each test tube.
Trending nowThis is a popular solution!

Chapter 16 Solutions
Chemistry
Additional Science Textbook Solutions
Introductory Chemistry (5th Edition) (Standalone Book)
Basic Chemistry (5th Edition)
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
General, Organic, and Biological Chemistry (3rd Edition)
Principles of Chemistry: A Molecular Approach (3rd Edition)
Introductory Chemistry (6th Edition)
- The common ion effect for ionic solids (salts) is to significantly decrease the solubility of the ionic compound in water. Explain the common ion effect.arrow_forwardA solution contains 0.00740 M calcium ion. A concentrated sodium fluoride solution is added dropwise to precipitate calcium fluoride (assume no volume change). a At what concentration of F does precipitate start to form? b When [F] = 9.5 104 M, what is the calcium-ion concentration? What percentage of the calcium ion has precipitated?arrow_forwardThe addition of hydrolochloric acid to the solution containing the silver group of cations is to selectively precipitate out the lead (II) ions. -True -Falsearrow_forward
- The value of the Solubility Product Constant for zinc sulfide is Write the reaction that corresponds to this Ksp value. + + Be sure to specify the state of each reactant or product.If a box is not needed leave it blank.arrow_forwardSolid Silver chromate is added to pure water at 25 degrees C. After the mixture is stirred to ensure that equilibrium is established, some of the solid remains undissolved at the bottom of the flask. Analysis of the equilibrium solution shows that its silver ion concentration is 1.0 x10^-4. Calculate ksp for this compound. The ksp for CaF2 is 2.5 x 10-11 at 25 degrees Celsius. Assuming that CaF2 dissociates complete upon dissolving and that there are no other important equilibria affecting the solubility, calculate the solubility of CaF2 in grams per liter.arrow_forwardCan we compare only the solubility product of barium chromate (BaCrO 4) which is 8.5 * 10 -11 with that of silver chromate (Ag 2 CrO 4) which is 9.0 * 10 -12 for to determine the solubility of these substances? Explain.arrow_forward
- 2. The concentration of a perchloric acid solution was established by titration against primary standard sodium carbonate. A 0.2068-g sample of sodium carbonate required 36.31 to reach the end point. Write the over-all reaction for the titration and calculate for the molarity of the acid.arrow_forwardWhat additional information do we need to answer the following question: How is the equilibrium of solid silver bromide with a saturated solution of its ions affected when the temperature is raised?arrow_forwardA student found two bottles of compounds with missing labels. The compounds are either Benzyl chloride or Bromobenzene. To figure out the contents of each bottle, the student put 2 drops of each compound into separate test tubes labeled X and Y and added 1 mL of a mixture of NaI and acetone to each tube. After 45 seconds, the student observed a white color precipitate in tube X, while there was no precipitate formed in tube Y. What compound (Benzyl chloride or Bromobenzene) does tube X contain? Whyarrow_forward
- 1. At a certain temperature, the solubility product constant* of copper (II) iodate, Cu(IO3)2, is 7.17x10-8 M3. Calculate the solubility of this compound for this temperature.*Unless stated otherwise all solubility product constants are for water. 2. Calculate the solubility of copper (II) iodate in 0.21 M copper (II) nitrate. Ksp* is 7.4x10-8M3.*You should know that the Ksp must refer to the copper iodate because all nitrate compounds are soluble and strong electrolytes! 3. What is the initial (before any reaction takes place) lead nitrate concentration when 6.0 mL of 0.171 M lead nitrate is added to 11 mL of 0.0086 M sodium chloride?arrow_forwardHint: Write the balanced chemical equation for the dissolution of magnesium bromide first. Then answer this question: What is the coefficient in front of the solvated anion? Choose one of the following: 0 1 2 3 4arrow_forwardA solution contains 0.14 M potassium sulfite and 0.14 M potassium carbonate. Solid silver acetate is added slowly to this mixture. What ionic compound precipitates first from the solution? Solubility product constant data is found in the Chemistry References. Formula of first precipitate = Hint: It is not necessary to do a calculation here.arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





