
Concept explainers
(a)
Interpretation:
Whether CH3(CH2)6CH3 is optically active or not needs to be determined.
Concept introduction:
The ability of substances to rotate the plane of polarized light passes through it is defined as an optical activity of the substance. The molecules with chiral center or asymmetric in nature can show optical activity. Thus, the presence of the chiral center in the molecule is important in this case.
(b)
Interpretation:
Whether the given molecule is optically active or not needs to be determined.
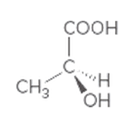
Concept introduction:
The ability of substances to rotate the plane of polarized light passes through it is defined as the optical activity of the substance. The molecules with chiral center or asymmetric in nature can show optical activity. Thus, the presence of the chiral center in the molecule is important in this case.
(c)
Interpretation:
Whether the given molecule is optically active or not needs to be determined.
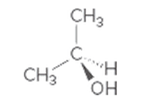
Concept introduction:
The ability of substances to rotate the plane of polarized light passes through it is defined as the optical activity of the substance. The molecules with chiral center or asymmetric in nature can show optical activity. Thus, the presence of the chiral center in the molecule is important in this case.
(d)
Interpretation:
Whether the given molecule is optically active or not needs to be determined.
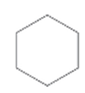
Concept introduction:
The ability of substances to rotate the plane of polarized light passes through it is defined as the optical activity of the substance. The molecules with chiral center or asymmetric in nature can show optical activity. Thus, the presence of the chiral center in the molecule is important in this case.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Draw the structure for a compound with molecular formula C2H2I2F2 a. that is optically inactive because it does not have an asymmetric center. b. that is optically inactive because it is a meso compound. c. that is optically active.arrow_forwardClassify each pair of compounds as constitutional isomers or stereoisomers.arrow_forwardWhat is the relationship between the two molecules shown below? OH H CH3 ОН H H H H H Н. H. H. -CH3 H H H H H H A) enantiomers B) diastereomers C) conformational isomers D) constitutional isomers E) identicalarrow_forward
- Label each stereogenic center as R or S.arrow_forward1. Give the number of stereogenic centers in the pain reliever Met-enkephalin, mark the corresponding stereogenic centers with an asterisk. H₂N. OH HO S I CH3arrow_forwardCaptopril is a drug used to treat high blood pressure and congestiveheart failure.Designate each stereogenic center as R or S.arrow_forward
- Identify the relationship between the following two compounds. h A) identical B) constitutional isomers C) enantiomers D) diastereomersarrow_forwardLocate the stereogenic centers in each compound. A molecule may have one or more stereogenic centers. Gabapentin enacarbil [part (d)] is used to treat seizures and certain types of chronic pain.arrow_forwardIdentify the relationship between the following two compounds. A) identical B) constitutional isomers C) enantiomers D) diastereomersarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
