
Concept explainers
a)
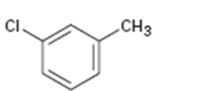
Interpretation:
Whether the compound given is ortho-, meta-, or para-disubstituted is to be stated.
Concept introduction:
Disubstituted benzenes are named using the prefixes ortho (o), meta (m) and para (p). An ortho-disubstituted benzene has its two substituent groups in a 1,2-relationship on the ring. A meta-disubstituted benzene has its two substituent groups in a 1,3-relationship on the ring. A para-disubstituted benzene has its two substituent groups in a 1,4-relationship on the ring.
To state:
Whether the compound given is ortho-, meta-, or para-disubstituted.
b)
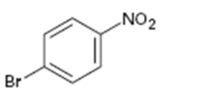
Interpretation:
Whether the compound given is ortho-, meta-, or para-disubstituted is to be stated.
Concept introduction:
Disubstituted benzenes are named using the prefixes ortho (o), meta (m) and para (p). An ortho-disubstituted benzene has its two substituent groups in a 1,2-relationship on the ring. A meta-disubstituted benzene has its two substituent groups in a 1,3-relationship on the ring. A para-disubstituted benzene has its two substituent groups in a 1,4-relationship on the ring.
To state:
Whether the compound given is ortho-, meta-, or para-disubstituted.
c)
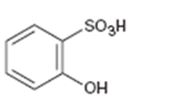
Interpretation:
Whether the compound given is ortho-, meta-, or para-disubstituted is to be stated.
Concept introduction:
Disubstituted benzenes are named using the prefixes ortho (o), meta (m) and para (p). An ortho-disubstituted benzene has its two substituent groups in a 1,2-relationship on the ring. A meta-disubstituted benzene has its two substituent groups in a 1,3-relationship on the ring. A para-disubstituted benzene has its two substituent groups in a 1,4-relationship on the ring.
To state:
Whether the compound given is ortho-, meta-, or para-disubstituted.
Trending nowThis is a popular solution!

Chapter 15 Solutions
Organic Chemistry
- CO7T01Q5547 Give the IUPAC name for the following compound:arrow_forwardMenthene, a compound found in mint plants, has the formula C10H18 and the IUPAC name 1-isopropyl- 4-methylcyclohexene. What is the structure of menthene?arrow_forwardWhat alkane, with molecular formula C7H16, forms seven monochlorinated products (disregarding stereoisomers) when heated with Cl2?arrow_forward
- Which of the following can be a molecular formula of an acyclic (this means no rings) hydrocarbon that contains one alkyne bond? C3H14 C8H16 C8H18 C3H20 C3H12arrow_forwardWhat hydrocarbon with the molecular formula C4H10 forms three monochlorinated products? One is achiral and two are chiral.arrow_forwardDraw the structures for the 8 constitutional isomers withe the molecular formula C9H12 that contain a benzene ring.arrow_forward
- Write structural formulas for all ketones with the molecular formula C6H12O and give each its IUPAC name. Which of these ketones are chiral?arrow_forwardWhen the acyclic portion of the molecule contains more carbon atoms (or an important functional group), name the ring as a cycloalkyl substituent. (g) xe (1) (h)arrow_forwardDraw the five constitutional isomers having molecular formula C6H14.arrow_forward

 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning



