
(a)
Interpretation:
The following
CH3CH2CH2CH2CH2Br
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with the preparation, reactions, and properties of organic compounds are said to be
Primary alkyl halide has one carbon bonded to the carbon atom that is bonded with halogen atom. Secondary alkyl halide has two carbons bonded to the same carbon atom bonded with the halogen. A tertiary alkyl halide has three carbons bonded to the same carbon atom bonded with the halogen atom.
(b)
Interpretation:
The following alkyl halide should be classified as primary, secondary and tertiary:
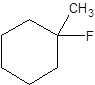
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with the preparation, reactions, and properties of organic compounds are said to be organic chemistry. The molecular formula of an organic compound represents the number of bonded atoms with their atomic symbols. The structural formula represents all the bonded atoms with chemical bonds and the arrangement of atoms in the molecule.
Primary alkyl halide has one carbon bonded to the carbon atom that is bonded with a halogen atom. Secondary alkyl halide has two carbons bonded to the same carbon atom bonded with the halogen. A tertiary alkyl halide has three carbons bonded to the same carbon atom bonded with the halogen atom.
(c)
Interpretation:
The following alkyl halide should be classified as primary, secondary and tertiary:
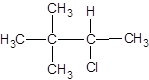
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with the preparation, reactions, and properties of organic compounds are said to be organic chemistry. The molecular formula of an organic compound represents the number of bonded atoms with their atomic symbols. The structural formula represents all the bonded atoms with chemical bonds and the arrangement of atoms in the molecule.
Primary alkyl halide has one carbon bonded to the carbon atom that is bonded with the halogen atom. Secondary alkyl halide has two carbons bonded to the same carbon atom bonded with the halogen. A tertiary alkyl halide has three carbons bonded to the same carbon atom bonded with the halogen atom.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Classify each alkyl halide as 1°, 2°, or 3°. CH3 c. CHg-C-CHCH3 ČH3 ČI CH;CH2CH,CH,CH2-Br b. d. a.arrow_forward18. Ketone reduction Dicyclohexyl ketone Reduce the ketone. 1. NaBH4, ethanol 2. H3O+ H OH Dicyclohexylmethanol (88%) (a 2° alcohol)arrow_forwardGive the IMPAC name for each compound. 1. COOH COOH 2. Br 3. CH3 CHz COOHarrow_forward
- 2-Cyclohexyl-2-methyl-butanol contains carbons. 07 0 8 10 O 11 OOO 0arrow_forwardQuestion 18.c of 20 Aldehydes and ketones are common carbonyl functional groups. Identify the structures and provide names of these molecules. Provide a systematic name for this structure. سکتے 5- H 1- 4- cyclo tert- 6- di n- tri 3- 2- sec- hex eth but Submit +arrow_forwardWhich of the following is phenol? CH2OH он I. IV. CH IL. V. OCH3 I. IV V.arrow_forward
- Draw the structure of a dihalide that could be used to prepare each alkyne. There may be more than one possible dihalide. CH3 b. CH₂-C-C=CH CH₂ a. CH₂C=CCH₂ C. -C=C-arrow_forwardCollege Chem II CHE 132 Saturated & Unsaturated Hydrocarbons 1. The following compounds are named incorrectly. Draw the structure of each compound and give it a correct IUPAC name. a) 2-ethyl-4-methylbutane b) 2-isopropyl-3-ethylpentane Hs C - CH-CH3 ansiba C H3- CH - CH-CH2-CH3 A14-ethyl-2,3-dimethyinexane) CH z opanthine CH3 c) 1-propyl-2-ethylbutane CH3 -CH2 - CH- CH2-,CH-CH>-CH; 13-0thylpetane) CHa d) 2,2-diethylpropane e) 3-sec-butylpentane f) 2,4-diethylhexanearrow_forwardCollege Chem II CHE 132 Alcohols & Ethers - Naming & Reactions 1. Name the following compounds. For a-d, indicate whether the compound is 1,2, or 3. b) CH a) CH-CH-CH-CHy CH-al-CH-CHy d) OH c) CHy f) CHy-CH- CH-CHy e) CHICHS 8) CHy-CH-CHy h) CHy-CH-0-CH-CHICHY CH2 CHy-CH-CH3arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





