
Concept explainers
If myosin V moved more like an “inchworm” (meaning it dragged its rear filament-binding domain forward, but not in a hand-over-hand fashion), how would a graph of spot movement over time differ from the one shown in Figure 14A-2b?
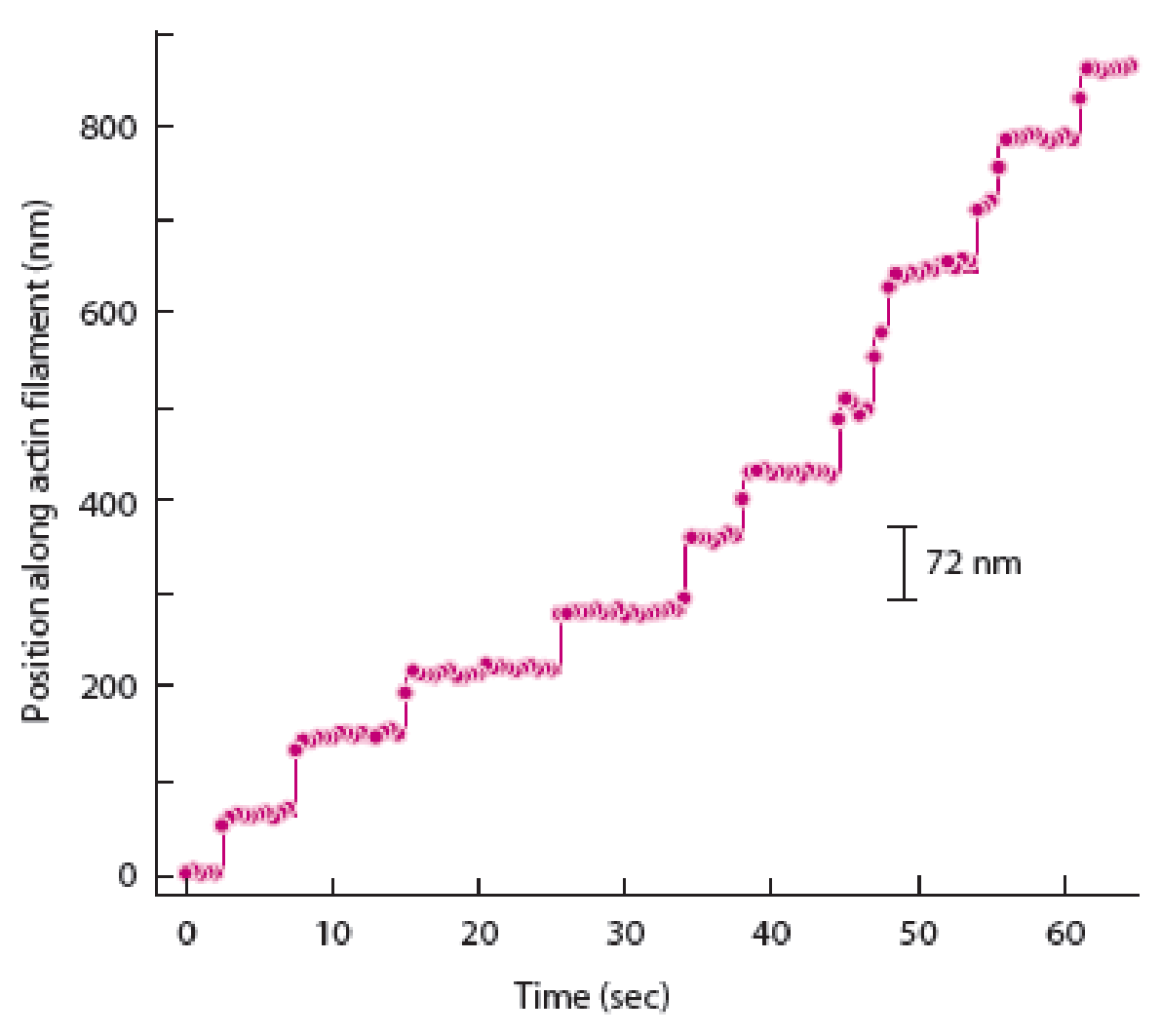
To determine: The way in which graph of spot movement over time differs from the graph shown in figure 14A-2b given in the textbook when myosin V moved like an inchworm.
Introduction: Myosin protein V is an important protein which is involved in the transports of various types of intracellular cargo along with actin proteins. These myosin proteins are involved in the transport of various membrane-bound organelles and show two alternating types of mechanisms for movement. One method is the hand-over-hand method and another method is the inch worm model.
Explanation of Solution
Hand-over-hand mechanism of myosin V movement states that the trailing head would move past the stationary forward head by an approximate distance of 74 nanometers to reach a new actin binding site. In contrast to this, the inch-worm model suggests that both heads would advance or move forward only by 37 nanometers during the step. Both trailing and leading heads do not undergo the process of position exchange during the inch worm model.
The best way to determine the difference between the hand-over-hand model and inch-worm models is to strictly follow the displacement of one of myosin V heads. Therefore, a graph of spot movement over time will differ in case of distance as 74 nanometers (position along actin filament) will be shown in the hand-over-hand model and 35 nanometers will be shown in inchworm model of spot movement.
Want to see more full solutions like this?
Chapter 14 Solutions
Becker's World of the Cell (9th Edition)
- How do the different structures and properties of myosin II and myosin V reflect their different functions in cells?arrow_forwardMYK-461 is also hypothesized to a) stabilize a head-head interaction in two-headed cardiac myosin. b) inhibit skeletal muscle myosin shortening. c) prevent myosin binding protein C from regulating actin-activation of cardiac myosin. d) accelerate ADP release, but only in the absence of actin. e) disrupt the OFF state of the thick-filament. Don't copy from Googlearrow_forwardThe virus SARS-CoV-2 which causes covid-19 has quite flexible nail protein. Make a simplified biomechanical equilibrium model based on the black and white card (electron microscopy). Assume that C is a pin joint. Joint B is described as a torsion spring with force pair moment M = κα, where α is the angle in radians. Suppose further that force from above can be modeled as gravity in A, and that the medium acts as a vertical roller support at R. Dimensions: LBC = 100 nm, LAB = 2LBC. The angle θ = 10◦ . a) If m = 10 milligrams, what is the “stiffness” κ, in fNm / row? (milligram = 10−6 kg, n = nano = 10−9 , f = femto = 10−15.) (5p) b) If m = 10 milligrams, what is the amount of force from the medium (The "roller support")?arrow_forward
- Which of the following is NỘT true of the mechanism by whldh kiesin motor proteins walk along microtubules? Kinesin molecules bind to other kinesin molecules as their cargo One foot, or head, domain of kinesin is always attachhed to the miarotubule Kinesin functions as a dimer. Kinesin dimers function autonomously (l.e. each molecule is bound to one molecule of cargo) Kinesin molecules are plus end-directed motor proteins A Moving to the next question prevents changes to this answer.arrow_forwardTo design a drug that inhibits metastasis, which of the following strategies is the WORST idea? to disrupt cadherin complexes to inhibit Rho family proteins to inhibit ARP complex O to block myosin-based contractile structurearrow_forwardThe tetanus toxin tetanospasmin blocks release of the neurotransmitter (GABA) that inhibits motor neurons and decreases muscle contraction. This causes persistence of the signal to contract and the resulting violent muscle contraction known as tetanic spasm. Compare the sustained contraction of muscle cells in rigor mortis with muscle contraction caused by the action of tetanospasmin on motor neurons. Describe experiments using marker molecules that have been done to elucidate important properties of (1) tight junctions and (2) gap junctions, and propose one additional experiment you could do with each junction to gain a greater understanding of that junction's properties.arrow_forward
- What ion is necessary for movement of the troponintropomyosin complex? What is the role of thesarcoplasmic reticulum in this process?arrow_forwardThe Arp2/3 complex makes branched actin filaments by associating with a ‘mother’ actin filament and initiating assembly of a ‘daughter’ actin filament. Although it has been thought that Arp2/3 complex associates with and makes branches from the side of the ‘mother’ actin filament, it is alternatively possible that Arp2/3 complex associates with and makes branches from the barbed end of the ‘mother’ actin filament. Design an in vitro experiment (with purified proteins, such as actin and Arp2/3 complex) to determine which of these possibilities is correct by taking advantage of capping protein (binds and blocks barbed ends), and direct visualization of actin filaments. (1) Briefly describe your experiment in no more than three sentences (2) Draw diagrams of what the arrangement of actin filaments (simple lines are okay) are expected to look like in your proposed experimental and control conditions that indicate the possible outcomes of your experiment (3) Briefly describe your…arrow_forwardHelp me pleasearrow_forward
- You are growing up myoblasts, C2C12 cells, to use in a myogenic study. You are using T-150 flasks with a culture area of 150 cm2 and when confluent contains 2 x 107 cells.You are growing up myoblasts 3)You seed another culture of a different end cell line, john12 cells at 8am on Friday with 104 cells. By Thursday at noon (12:00pm) they reach confluence. Assume the same confluency as stated above for T150 flask. What is the generation time (doubling time)arrow_forward"In most animal cells, minus end-directed microtubule motors deliver their cargo to the periphery of the cell, whereas plus end-directed microtubule motors deliver their cargo to the interior of the cell", is true or false.arrow_forwardSelect the most accurate explanation for how vesicular transport occurs when a receptor is moving from the plasma memberane to the late endosome/lysosome. A. Endosomes containing the receptor are transported by myosin II walking towards the minus end of microtubules. Endosomes are targeted to the late endosome/lysosome by Rab and SNARES facilitate membrane fusion. B. Endosomes containing the receptor are transported by myosin V walking towards the minus end of microtubules. Endosomes are targeted to the late endosome/lysosome by Rab and SNARES facilitate membrane fusion. OC. Endosomes containing the receptor are transported by dyenin walking towards the minus end of microtubules. Endosomes are targeted to the late endosome/lysosome by Rab and SNARES facilitate membrane fusion. D. Endosomes containing the receptor are transported by kinesin-13 walking towards the plus end of microtubules. Endosomes are targeted to the late endosome/lysosome by clathrin and SNARES facilitate membrane…arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





