
Concept explainers
(a)
Interpretation:
The given compound is to be identified as
Concept introduction:
Heterocyclic compounds are defined as cyclic compounds which consist of at least one heteroatom, mostly nitrogen, sulfur, or oxygen. Heterocyclic compounds can be aromatic, antiaromatic, or nonaromatic. In some heterocyclic compounds, a noncarbon atom contributes to a p atomic orbital to the aromatic
Huckel’s
1) Aromatic if the number of electrons in that
Answer to Problem 14.20P
The given compound is antiaromatic.
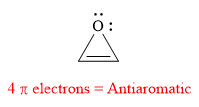
Explanation of Solution
The given compound is:
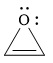
It is a heterocyclic compound with an oxygen atom. The hybridization of the oxygen atom must be

The total number of electrons in this
The given heterocyclic compound is antiaromatic if the total number of electrons in that
(b)
Interpretation:
The given compound is to be identified as aromatic, antiaromatic, or nonaromatic.
Concept introduction:
Heterocyclic compounds are defined as cyclic compounds which consists of at least one heteroatom mostly nitrogen, sulfur, or oxygen. Heterocyclic compounds can be aromatic, antiaromatic, or nonaromatic. In some heterocyclic compounds, a noncarbon atom contributes a p atomic orbital to the aromatic
Huckel’s rule for aromaticity states that if a species possesses a pi system of molecular orbitals constructed from p-orbitals that are fully conjugated around a ring (
1) Aromatic if the number of electrons in that
Answer to Problem 14.20P
The given compound is aromatic.
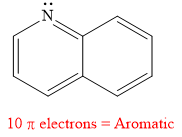
Explanation of Solution
The given compound is:
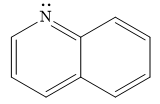
It is a heterocyclic compound with a nitrogen atom. The hybridization of the nitrogen atom must be

Thus, the total number of electrons in this
The given heterocyclic compound is aromatic if the number of electrons in that
(c)
Interpretation:
The given compound is to be identified as aromatic, antiaromatic, or nonaromatic.
Concept introduction:
Heterocyclic compounds are defined as cyclic compounds which consist of at least one heteroatom mostly nitrogen, sulfur, or oxygen. Heterocyclic compounds can be aromatic, antiaromatic, or nonaromatic. In some heterocyclic compounds, a noncarbon atom contributes a p atomic orbital to the aromatic
Huckel’s rule for aromaticity states that if a species possesses a pi system of molecular orbitals constructed from p-orbitals that are fully conjugated around a ring (
1) Aromatic if the number of electrons in that
Answer to Problem 14.20P
The given compound is nonaromatic.
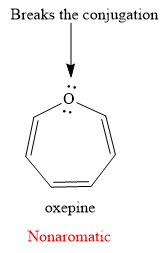
Explanation of Solution
The given compound is:
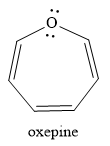
It is an oxygen containing a heterocycle consisting of a seven-membered ring with three double bonds. Due to geometrical constrains, the lone pairs of electrons on the oxygen atom are not in conjugation with the

If the molecule is assumed to be planar (flat), then too it would contain
The rule for aromaticity or antiaromaticity applies only if the system is planar, cyclic and has overlap of p-orbitals.
(d)
Interpretation:
The given compound is to be identified as aromatic, antiaromatic, or nonaromatic.
Concept introduction:
Heterocyclic compounds are defined as cyclic compounds which consists of at least one heteroatom mostly nitrogen, sulfur, or oxygen. Heterocyclic compounds can be aromatic, antiaromatic, or nonaromatic. In some heterocyclic compounds, a noncarbon atom contributes a p atomic orbital to the aromatic
Huckel’s rule for aromaticity states that if a species possesses a pi system of molecular orbitals constructed from p-orbitals that are fully conjugated around a ring (
1) Aromatic if the number of electrons in that
Answer to Problem 14.20P
The given compound is nonaromatic.
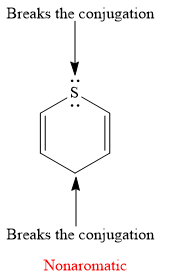
Explanation of Solution
The given compound is:
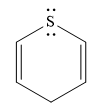
It is a sulfur containing a heterocycle consisting of a six-membered ring with two double bonds. There are two lone pair of electrons on the sulfur atom, but the sulfur atom is

As the
The rule for aromaticity or antiaromaticity applies only if the system is planar, cyclic and has overlap of p-orbitals.
(e)
Interpretation:
The given compound is to be identified as aromatic, antiaromatic, or nonaromatic.
Concept introduction:
Heterocyclic compounds are defined as cyclic compounds which consists of at least one heteroatom mostly nitrogen, sulfur, or oxygen. Heterocyclic compounds can be aromatic, antiaromatic, or nonaromatic. In some heterocyclic compounds, a noncarbon atom contributes a p atomic orbital to the aromatic
Huckel’s rule for aromaticity states that if a species possesses a pi system of molecular orbitals constructed from p-orbitals that are fully conjugated around a ring (
1) Aromatic if the number of electrons in that
Answer to Problem 14.20P
The given compound is nonaromatic.
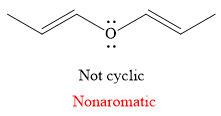
Explanation of Solution
The given compound is:
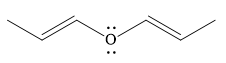
It is a straight chain compound with an oxygen atom and two double bonds. The above compound is not a cyclic compound. The rule for aromaticity or antiaromaticity applies only if the system is planar, cyclic and has overlap of p-orbitals. Thus, the

The rule for aromaticity or antiaromaticity applies only if the system is planar, cyclic and has overlap of p-orbitals.
(f)
Interpretation:
The given compound is to be identified as aromatic, antiaromatic, or nonaromatic.
Concept introduction:
Heterocyclic compounds are defined as cyclic compounds which consists of at least one heteroatom mostly nitrogen, sulfur, or oxygen. Heterocyclic compounds can be aromatic, antiaromatic, or nonaromatic. In some heterocyclic compounds, a noncarbon atom contributes a p atomic orbital to the aromatic
Huckel’s rule for aromaticity states that if a species possesses a pi system of molecular orbitals constructed from p-orbitals that are fully conjugated around a ring (
1) Aromatic if the number of electrons in that
Answer to Problem 14.20P
The given compound is aromatic.
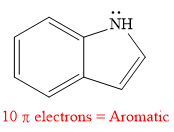
Explanation of Solution
The given compound is:
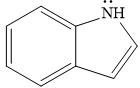
It is a heterocyclic compound with a nitrogen atom in which one six-membered and one five-membered rings are fused. The hybridization of the nitrogen atom is

Thus, the total number of electrons in this
The given heterocyclic compound is aromatic if the number of electrons in that
Want to see more full solutions like this?
Chapter 14 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- These following structures are aromatic, antiaromatic or nonaromatic?arrow_forwardIn the first blank, label the molecule as either aromatic, antiaromatic, or nonaromatic. In the second, for aromatic and antiaromatic compounds provide the number of pi electrons that would be used. Place a zero in the second blank if non aromatic.arrow_forwardDaw the resoance sructures of following compounds. Also, draw the resonance hybrid and indicate the major contributor in the drawn resonance structures.arrow_forward
- Are the following compounds aromatic, nonaromatic, or antiaromatic? Explain your reasoningusing the general rules for aromaticityarrow_forwardIn the first blank, label the molecule as either aromatic, antiaromatic, or nonaromatic. In the second, provide the number of pi electrons that would be used. Place a zero in the second blank if non aromatic.arrow_forwardare these aromatic, antiaromatic, or nonaromatic? Draw in all lonepairs and indicate the orbital each lone pair resides in.arrow_forward
- Hello, can you draw and name two different molecules (with the explicit formula) with a total of 12 carbons, together with the side groups, according to the options given? a)Write and name two cyclic amide molecule. b)Write and name two aromatic amine molecule.arrow_forwardIs this an aromatic, antiaromatic, or non aromatic molecule?arrow_forwardWhich are aromatic species? Please circle correct species. (assume planarity unless otherwise noted)arrow_forward
- Draw the curved arrows to show the resonance structure for the following molecules. ( there are 3 molecules )arrow_forwardwhich molecules should be aromaticarrow_forwardIn HF , neither H nor F holds a full formal charge of +1 or 1 . Organic chemists represent apartial charge using the Greek letter delta () . On the electron density map of the molecule HF above, add a + to one atom and a to the other to indicate which way the bond is polarized.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
