
Interpretation:
The structure of an alcohol of given molecular formula C4H10O to be predicted using 13CNMR spectra.
Concept introduction:
The 13CNMR spectrum gives information on the different electronic environments of carbon. As like 1HNMR, the number of signals generated in 13CNMR are predicted by performing symmetry operations (rotation or reflection symmetry). Only chemical shift values are reported in the spectrum but not the multiplicity and integration values because the coupling between two neighboring 13C-13 C nuclei are weakly involved due to the low abundance of 13C isotopes of carbon atom.
To Identify:
The structure of an alcohol of given molecular formula C8H16O.
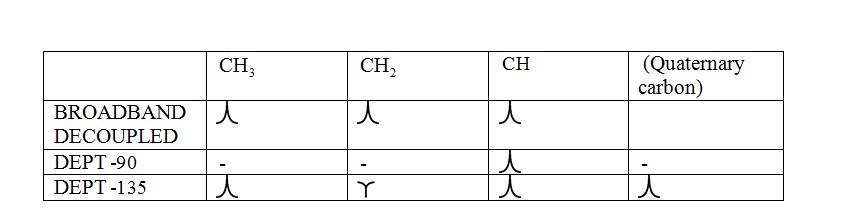
Broadband-decoupled 13CNMR spectrum:
The spectra provide information regarding the total number of carbon environments.
DEPT (Distortionless enhancement by polarization transfer):
a) DEPT-90: The spectrum exhibits signal only from CH group and no signals from CH3, CH2, CH and quaternary carbon (carbon with no protons).
b) DEPT-135: The spectrum exhibits CH3 groups and CH groups as positive signals (pointing up); CH2 groups appear as negative signals (pointing down) and quaternary carbon does not appear.
The signals appear in each type of spectrum:
Trending nowThis is a popular solution!

Chapter 13 Solutions
Organic Chemistry
- Question 6 of 25 Provide the correct systematic name for the compound shown here. || CH3-CH₂-C-O-CH3 iso sec- di cyclo tri tert- acetate pent but eth prop meth oic acid yl an one ester oate K Submit +arrow_forwardProvide the correct IUPAC name for the compound shown here. CH3 H-C-CH2-CH-CH2-CH-CH3 || CH3 Iarrow_forwardWhat is relationship between the two structures shown below? CI H CH3 H-NH2 CI- -H- H NH2 ČH;CH3arrow_forward
- Write the IUPAC name for each of the following: Part A Br Spell out the IUPAC name of the compound. Submit Request Answerarrow_forwardExplain why HC = CH is more acidic than CH3CH3, even though the C - H bond in HC = CH has a higher bond dissociation energy than the C - H bond in CH3CH3arrow_forwardHere is the chemical structure of 2-butanone: H H H. H-C-C -C-C-H H. H. Decide whether each molecule in the table below is another mnolecule of 2-butanone, a molecule of an isomer of 2-butanione, or a molecule of an entirely different compound. molecule relationship to 2-butanone CH, (Choose one) CH,-CH,-ċ=O another molecule of 2-butanone a molecule of an Isomer of 2-butanone a molecule of a different compound H OHHarrow_forward
- Part D Spell out the IUPAC name of the compound. 4-ethyl-hex-2-ene Submit Previous Answers Request Answer X Incorrect; Try Again; 7 attempts remaining Provide Feedbackarrow_forwardHow many asymmetric centers are present in a molecule of 2,4,6-trimethylheptane? please answer mearrow_forwardcompound CH3 CH₂0 CH3 — OH C- CH3 1 CH3-N - CH3 CH₂ ||| -C-H 01C- | NH₂ CH3 family esterarrow_forward
- Explain why an alkyne often has a slightly higher boiling point than an alkene of similar molecular weight. For example, the bp of pent-1-yne is 39°C, and the bp of pent-1-ene is 30°C.arrow_forwardLabel the following compound as R or S. OH CCH2CH3 (CH3)2CH H.arrow_forwardWhat is the IUPAC name of the following compound? * CEN H2 | H2 H3C-CEC -С—с—с—ОН H3C 2-Methyl-2-hydroxymethylhex-4-yne nitrile 2-Cyano-2-methylhex-4-yne-ol 2-Hydroxymethyl-2-methylhex-4-yne nitrile 1-Cyano-2-hydroxymethyl-2-methylhex-4- ynearrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
