
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.SE, Problem 35AP
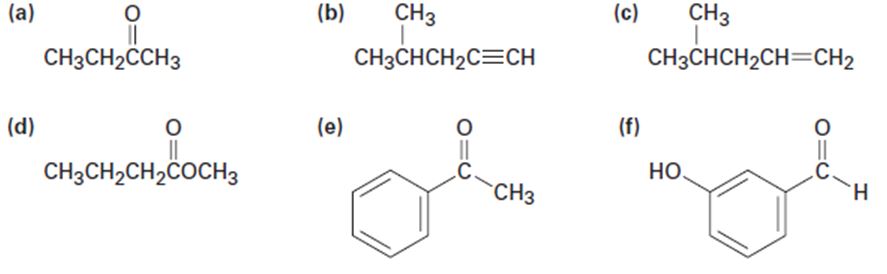
At what approximate positions might the following compounds show IR absorptions?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
What is the compound of this IR Spectrum?
Each of the IR spectra shown below is accompanied by a set of four compounds. In each case, indicate which of the four compounds is responsible for the spectrum.
Identify the important absorption peaks in the following IR spectra and describe the appearance of the important absorption peaks in the IR Spectra.
No cursive writing please. Thank you
Chapter 12 Solutions
Organic Chemistry
Ch. 12.2 - Prob. 1PCh. 12.2 - Two mass spectra are shown in FIGURE 12-8. One...Ch. 12.3 - What are the masses of the charged fragments...Ch. 12.3 - Prob. 4PCh. 12.5 - Prob. 5PCh. 12.5 - Prob. 6PCh. 12.7 - What functional groups might the following...Ch. 12.7 - How might you use IR spectroscopy to distinguish...Ch. 12.8 - Prob. 9PCh. 12.8 - Where might the following compounds have IR...
Ch. 12.8 - Where might the following compound have IR...Ch. 12.SE - Prob. 12VCCh. 12.SE - Show the structures of the fragments you would...Ch. 12.SE - Propose structures for compounds that fit the...Ch. 12.SE - Write molecular formulas for compounds that show...Ch. 12.SE - Camphor, a saturated monoketone from the Asian...Ch. 12.SE - The nitrogen rule of mass spectrometry says that a...Ch. 12.SE - In light of the nitrogen rule mentioned in Problem...Ch. 12.SE - Nicotine is a diamino compound isolated from dried...Ch. 12.SE - The hormone cortisone contains C, H, and O, and...Ch. 12.SE - Halogenated compounds are particularly easy to...Ch. 12.SE - Prob. 22APCh. 12.SE - Propose structures for compounds that fit the...Ch. 12.SE - 2-Methylpentane (C6H14) has the mass spectrum...Ch. 12.SE - Assume that you are in a laboratory carrying out...Ch. 12.SE - What fragments might you expect in the mass...Ch. 12.SE - How might you use IR spectroscopy to distinguish...Ch. 12.SE - Would you expect two enantiomers such as...Ch. 12.SE - Would you expect two diastereomers such as meso-2,...Ch. 12.SE - Propose structures for compounds that meet the...Ch. 12.SE - How could you use infrared spectroscopy to...Ch. 12.SE - Prob. 32APCh. 12.SE - At what approximate positions might the following...Ch. 12.SE - How would you use infrared spectroscopy to...Ch. 12.SE - At what approximate positions might the following...Ch. 12.SE - Assume that you are carrying out the dehydration...Ch. 12.SE - Assume that you are carrying out the base-induced...Ch. 12.SE - Prob. 38APCh. 12.SE - Carvone is an unsaturated ketone responsible for...Ch. 12.SE - Prob. 40APCh. 12.SE - The mass spectrum (a) and the infrared spectrum...Ch. 12.SE - The mass spectrum (a) and the infrared spectrum...Ch. 12.SE - Propose structures for compounds that meet the...Ch. 12.SE - 4-Methyl-2-pentanone and 3-methylpentanal are...Ch. 12.SE - Grignard reagents undergo a general and very...Ch. 12.SE - Ketones undergo a reduction when treated with...Ch. 12.SE - Nitriles, R–=C≡N, undergo a hydrolysis...Ch. 12.SE - The infrared spectrum of the compound with the...Ch. 12.SE - The infrared spectrum of the compound with the...Ch. 12.SE - Prob. 50AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Calculate the IHD of C7H6XNO and identify the important peaks in the following MS spectral data and draw the structure of the important peaks in the following MS spectral data.arrow_forwardIf you know the energies of the HOMO and LUMO of a particular compound, how could you calculate the lowest energy optic absorption?arrow_forwardndicate which among the following compounds corresponds to each of the IR spectra shown below byplacing the letter of the chosen compound inside the box. Indicate the important absorptions (bylabeling the bands in the spectra) that allowed you to make the assignmentarrow_forward
- This is the 13C spectrum for an X compound with molecular formula C7H12O4. The substance is not soluble in NaHCO3 and has a stretch at 1740cm-1 on the IR spectrum. What is the structure of X?arrow_forwardEach of the IR spectra shown below is the spectrum of one of the following compounds. Identify the compound that produced each spectrumarrow_forwardMatch the peaks in this spectrum with hydrogens on the structure below. A) H3C I H₂ 3.0 H₂ CH3 IV 2.0 ppm 1.0 1.68 ppm II: 0.99 ppm III: 2.32 ppm IV: 3.61 ppm 1: B) I: 0.99 ppm II: 1.68 ppm III: 2.32 ppm IV: 3.61 ppm C) I: II: 1.68 ppm 0.99 ppm +arrow_forward
- How does the operating frequency in NMR spectroscopy compare with the operating frequency in IR and UV/Vis spectroscopy?arrow_forwardDetermine which of the following compounds should display its Imax at 288 nm and justify the reason for your selection.arrow_forwardDeduce which hydrogens are responsible for each peak of the spectrum and note any anomaliesarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY