
Thinking Like an Engineer: An Active Learning Approach (4th Edition)
4th Edition
ISBN: 9780134639673
Author: Elizabeth A. Stephan, David R. Bowman, William J. Park, Benjamin L. Sill, Matthew W. Ohland
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 12.5, Problem 9CC
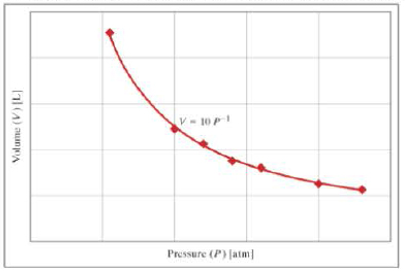 The graph shows the ideal gas law relationship (PV=nRT) between pressure (P) and volume (V). If the tank is at a temperature of 300 kelvins and is filled with nitrogen (formula, N2; molecular weight, 28 grams per mole), what is the mass of gas in the tank in units of grams?
The graph shows the ideal gas law relationship (PV=nRT) between pressure (P) and volume (V). If the tank is at a temperature of 300 kelvins and is filled with nitrogen (formula, N2; molecular weight, 28 grams per mole), what is the mass of gas in the tank in units of grams?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
The equation of state of an ideal gas describes the relationship between its pressure p, volume V,
and temperature T as:
PV = nRT
where n is the amount of substance of gas (number of moles), and R is the ideal gas constant.
What are the dimensions of R?
How much heat is absorbed by a 27.0 g sample of water in going from liquid at 38.2 °C to steam
at 119 °C and a pressure of 1.00 atm? Use the data in the following table. Express your answer
in kilojoules. Pay attention to units!
Some Thermal Properties of Water
Property
Specific heat (J/g °C)
Solid
Liquid
Gas
AHfusion (kJ/mol; at 0 °C)
A Hvaporization (kJ/mol; at 100 °C)
A Hsublimation (kJ/mol)
Melting point (°C)
Boiling point (°C)
Answer:
Value
2.07
4.18
2.01
6.01
40.6
50.9
0
100
The ideal gas law relates the pressure P, volume V, absolute temperature T (Kelvin), and amount of gas n.
The law is P = mAT
where R is the gas constant.
An engineer must design a large natural gas storage tank to be expandable to maintain the pressure constant at
2.2 atmospheres. In December when the temperature is -15°C, the volume of gas in the tank is 28,500 ft°. What
will the volume of the same quantity of gas be in July when the temperature is 31°C?
(Hint: Use the fact that n, R, and P are constant in this problem. Note also that Kelvin = °C+273.2)
Chapter 12 Solutions
Thinking Like an Engineer: An Active Learning Approach (4th Edition)
Ch. 12.3 - Fluid A as a dynamic viscosity of 0.5 centipoise...Ch. 12.3 - Fluid A has a dynamic viscosity of 0.5 centipoise...Ch. 12.3 - Fluid A has a dynamic viscosity of 0.5 centipoise...Ch. 12.4 - You have three springs, with stiffness 1,2 and 3...Ch. 12.4 - You have three resistors with resistance 2,2, and...Ch. 12.4 - You have four 60-nanofarad [nF] capacitors. Using...Ch. 12.4 - You have three 120 millihenry [mH] inductors. Can...Ch. 12.5 - The graph shows the ideal gas law relationship...Ch. 12.5 - The preceding graph shows the ideal gas Jaw...Ch. 12.6 - The decay of a radioactive isotope was tracked...
Ch. 12 - The graph shows the ideal gas law relationship (PV...Ch. 12 - An inductor is an electrical device that can store...Ch. 12 - Solid objects, such as your desk or a rod of...Ch. 12 - Mercury has a dynamic viscosity of 1.55...Ch. 12 - SAE 99W10, a brand new type of motor oil has a...Ch. 12 - You have two springs each of stiffness 2 newton...Ch. 12 - Prob. 7ICACh. 12 - Four springs were tested, with the results shown...Ch. 12 - Four circuits were tested, with the results shown...Ch. 12 - Assume you have an unlimited number of inductors...Ch. 12 - a. The equivalent capacitance of the circuit shown...Ch. 12 - A standard guitar, whether acoustic or electric,...Ch. 12 - The vibrating frequency of a guitar string depends...Ch. 12 - Solid objects, such as your desk or a rod of...Ch. 12 - Eutrophication is a process whereby lakes,...Ch. 12 - The following graph shows the relationship between...Ch. 12 - The total quantity (mass) of a radioactive...Ch. 12 - Match the data series from the options shown on...Ch. 12 - 1. For a simple capacitor with two f lat plates,...Ch. 12 - 2. When we wish to generate hydroelectric power,...Ch. 12 - 3. When rain falls over an area for a sufficiently...Ch. 12 - You are experimenting with several liquid metal...Ch. 12 - 5. The resistance of a wire (R [ohm)) is a...Ch. 12 - 6. Use the figure shown to answer the following...Ch. 12 - 7. You are given four springs, one each of...Ch. 12 - You have three springs. You conduct several tests...Ch. 12 - 9. You are given four resistors, each of 7.5, 10,...Ch. 12 - 10. You have three resistors. You conduct several...Ch. 12 - 11. Use the diagrams shown to answer the following...Ch. 12 - 12. When a buoyant cylinder of height H, such as a...Ch. 12 - 13. It is difficult to bring the Internet to some...Ch. 12 - 14. The data shown in the following graph was...Ch. 12 - 15 A standard guitar, whether acoustic or...Ch. 12 - 16. Your supervisor has assigned you the task of...Ch. 12 - 17. One of the NAE Grand Challenges for...Ch. 12 - 18. When volunteers build a Habitat for Humanity...Ch. 12 - 1. As part of an electronic music synthesizer, you...Ch. 12 - Prob. 20RQ
Additional Engineering Textbook Solutions
Find more solutions based on key concepts
The spring of k and unstretched length 1.5R is attached to the disk at a radial distance of 0.75R from the cent...
Engineering Mechanics: Statics
Determine the length of the cantilevered beam so that the maximum bending stress in the beam is equivalent to t...
Mechanics of Materials (10th Edition)
A nozzle at A discharges water with an initial velocity of 36 ft/s at an angle with the horizontal. Determine ...
Vector Mechanics for Engineers: Dynamics
3.3 It is known that a vertical force of 200 lb is required to remove the nail at C from the board. As the nail...
Vector Mechanics for Engineers: Statics
A windowmounted air conditioner removes 3.5kJ from the inside of a home using 1.75 kJ work input. How much ener...
EBK FUNDAMENTALS OF THERMODYNAMICS, ENH
What is the importance of modeling in engineering? How are the mathematical models for engineering processes pr...
Heat and Mass Transfer: Fundamentals and Applications
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- A hemispherical container, 26 inches in diameter, is filled with a liquid at 20°C and weighed. The liquid weight is found to be 1617 ounces. What is the density of the fluid in kg/m3? ]kg/m3arrow_forwardThe Antoine Equation can be used to estimate vapor pressure at various temperatures for different substances using sets of empirically-derived constants. The equation can be written as: Where for water A = 8.07131, B = 1730.63, C = 233.426, T = temperature [°C], and P = vapor pressure [mmHg]. Note that with these constants the temperature and pressure must be provided in °C and mmHg, respectively. Using the Antoine Equation and the provided constants, what atmospheric pressure (provided in terms of % of standard atmospheric pressure) will permit water to boil at 75 °C?arrow_forwardStudy the table to the right and then answer these questions by filling in the blank columns in the table.1. Convert the miles per gallon figures in the table tokilometers per liter (kpl).2. How many liters (and how many gallons) of gasolinewould each type of car use annually if it were driven19,300 kilometers (12,000 miles) per year?3. How many kilograms (and how many pounds) of carbon dioxide would be released into the atmosphereannually by each car, based on the fuel consumptioncalculated in question 2? Assume that the combustion of gasoline releases 2.3 kilograms of CO2 per liter(19 pounds per gallon).arrow_forward
- 1. Refrigerant R-22 with the mass of 2 kg is located inside the sealed container, initially saturated vapor at 6.8 bar. Heat was added to refrigerant and final pressure reached 20 bar according to pv=const. Neglecting the kinetic and potential energy changes determine: Internal energy change in [J] Heat added to the refrigerant in [J] and work done by fluid in [J]. 3.draw the Temperature-volume diagram showing all stages. 4.What do you think about the final stage condition? Is it gas, liquid-gas, or liquid stage? Use energy balance equation and find all unknowns using the NIST webbook.arrow_forwardIn order to fnd the specifc heat c of an unknown substance, you place 75 g of the substance in a 30 g copper calorimeter that contains 65 g of water, all initially at 20 °C. You then add 100 g of water at 80 °C, and measure the fnal temperature of the system to be 49 °C. Assuming no heat is lost to the environment, what is c? [Answer: c = 2200 J/kg. K] 즈arrow_forwardThe elevations of the ski jumping hill shown below are as follows: hA = 37 mhB = 2.4 mhC = 5.5 m If the velocity of a ski jumper is measured to be 25.3 m/s at position B, what will be the launch velocity (in m/s) at position C [round your final answer to one decimal place]?arrow_forward
- In medical literatures, local blood perfusion rate is typically presented as xx ml/(min 100g tissue), in another word, it represents xx ml of blood supplied to a tissue mass of 100 g per minute to satisfy its nutritional needs. As we learned from the course lectures, the local blood perfusion rate appearing in the Pennes bioheat equation is in a unit of 1/s, or can be interpreted as xx ml of blood supplied to a tissue volume of 1 ml per second. The following lists the blood perfusion rates in various organs or structures in a human body from medical textbooks: brain (50 ml/(min 100g tissue)), kidney (35 ml/(min 100g tissue)), and muscle at rest (3 ml/(min 100g tissue)). Please convert the above local blood perfusion rates into values with the unit of 1/s, therefore, they can be used in the Pennes bioheat equation. The tissue density in a human body is 1050 kg/m³.arrow_forwardA vertical piston-cylinder arrangement with a diameter of 120 mm contains water with a quality of 60%. The pressure is 3 bar. Initially the piston rests 40 mm above the bottom of the cylinder. Heat is then added until the volume is 3 times the original volume. (a) Determine the mass of the water in the cylinder [ Select ] O kg (b) Determine the work done. [ Select ] O (J) (c) The final temperature [ Select ] O °C (d) The heat transferred to the system [ Select ]arrow_forwardWhat are the gauge pressures at point A, B and C in each figure below? Water density is 1000 kg/m³. The pressure po is the absolute pressure. Ambient pressure 101 325 Pa. Round off the answer to an integer in kPa, but enter the answer without the unit. (a) P=1 bar (b) B. C• (c) 3m 2marrow_forward
- On an alien planet, the law that expresses the flow of blood through a blood vessel, F, as a function of the radius r of the vessel is f=kr5/2where k is a constant. Certain geological features of this planet produce a gas that changes the radius of blood vessels by 5% at the time of inhalation. What would the corresponding increase in blood flow through the vessel at this time be? Write 1-2 sentences explaining how you found your answer.arrow_forwardThe following processes occurs in a reversible thermodynamic cycle: 1-2: Reversible polytropic compression at pressure [X] bar at volume [Y] m3 to a pressure [Z] bar and specific volume [A] m3/kg. The index of compression may be taken as n. 2-3: Reversibly expansion with expansion index of 2 to pressure [B] bar. 3-1: Reversible cooling at constant volume to the initial state. What are the final attempt values of X, Y, Z, A and B from the Part A of the eThuto Assessment. calculated formula similar question? X= ; Y= ; Z= ; A= ; B=arrow_forwardIn a hydraulics laboratory, a certain test liquid flows incompressible. For the flow of this liquid, it is correct to state that: Answers: a) The pressure must be the same at all points within the liquid. b) the velocity must be the same at all points within the liquid. c) the density must have the same value at all points within the liquid and cannot change as it flows. d) the pressure at a certain point within the liquid cannot change as it flows.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY

Elements Of Electromagnetics
Mechanical Engineering
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:9780134319650
Author:Russell C. Hibbeler
Publisher:PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:9781259822674
Author:Yunus A. Cengel Dr., Michael A. Boles
Publisher:McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:9781118170519
Author:Norman S. Nise
Publisher:WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:9781337093347
Author:Barry J. Goodno, James M. Gere
Publisher:Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:9781118807330
Author:James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:WILEY
Dimensional Analysis - in physics; Author: Jennifer Cash;https://www.youtube.com/watch?v=c_ZUnEUlTbM;License: Standard youtube license