
Classify the carbon atoms in each compound as 1°, 2°, 3°, or 4°.
a.
b.
(a)
Interpretation:
The carbon in the following compound should be classified as
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
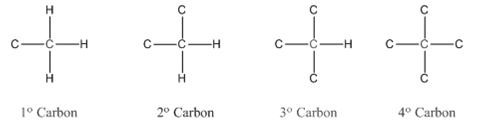
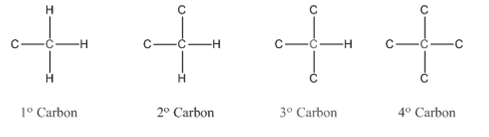
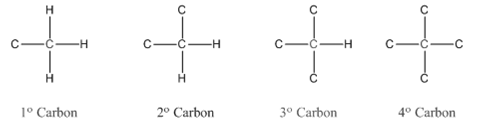
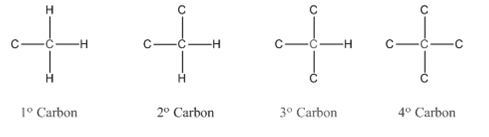
In organic chemistry, carbons are classified as primary, secondary, tertiary and quaternary carbons.
Primary carbon (
Secondary carbon (
Tertiary carbon (
Quaternary carbon (
Answer to Problem 12.3PP
Explanation of Solution
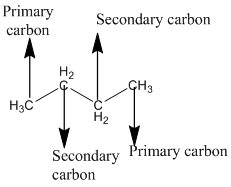
The given compound is
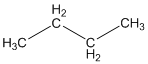
According to the above structure of compound, the terminal carbons present on both sides of the chain is classified as primary carbon as they are linked with one other carbon atom whereas carbon atoms which are present in between the terminal carbonsof the chain are linked with two other carbon atoms thus, classified as secondary carbon.
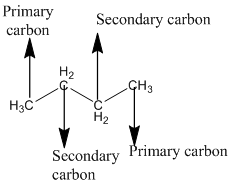
Thus, two primary and two secondary carbons are present in the structure.
(b)
Interpretation:
The carbon in the following compound should be classified as
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
In organic chemistry, carbons are classified as primary, secondary, tertiary and quaternary carbons.
Primary carbon (
Secondary carbon (
Tertiary carbon (
Quaternary carbon (
Answer to Problem 12.3PP
Explanation of Solution
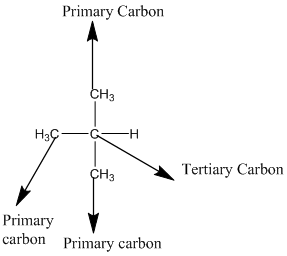
The given compound is
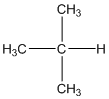
According to the above structure of compound, the terminal carbons (3 carbons) present is classified as primary carbon as they are linked with one other carbon atom whereas carbon atom which is present in the middle linked with three other carbon atoms thus, classified as tertiary carbon.
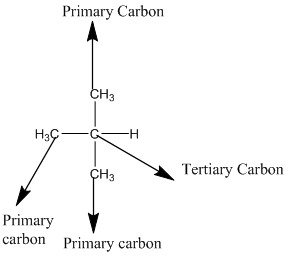
Thus, three primary carbons and onetertiary carbon atom are present in the structure.
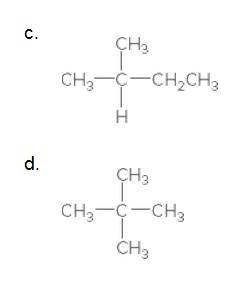
(c)
Interpretation:
The carbon in the following compound should be classified as
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
In organic chemistry, carbons are classified as primary, secondary, tertiary and quaternary carbons.
Primary carbon (
Secondary carbon (
Tertiary carbon (
Quaternary carbon (
Answer to Problem 12.3PP
Explanation of Solution
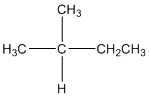
The given compound is
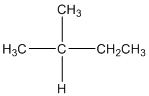
According to the above structure of compound, the terminal carbons (3 carbons) present is classified as primary carbon as they are linked with one other carbon atom whereas the carbon which is linked with two other carbon atoms is classified as secondary carbon and the carbon which is linked with three other carbon atoms is classified as tertiary carbon.
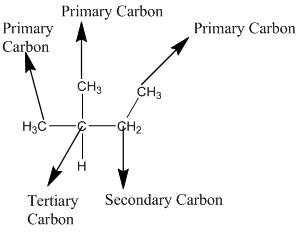
Thus, three primary carbons, one secondary carbon atom and one tertiary carbon atom are present in the structure.
(d)
Interpretation:
The carbon in the following compound should be classified as
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
In organic chemistry, carbons are classified as primary, secondary, tertiary and quaternary carbons.
Primary carbon (
Secondary carbon (
Tertiary carbon (
Quaternary carbon (
Answer to Problem 12.3PP
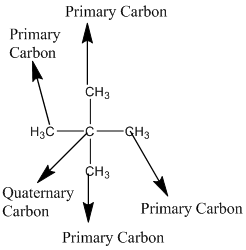
Explanation of Solution
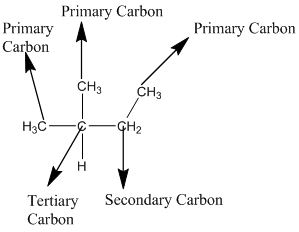
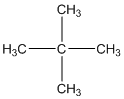
The given compound is
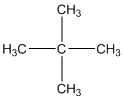
According to the above structure of compound, the terminal carbons (four carbons) present is classified as primary carbon as they are linked with one other carbon atom whereas the carbon which is linked with four other carbon atoms is classified as quaternary carbon.
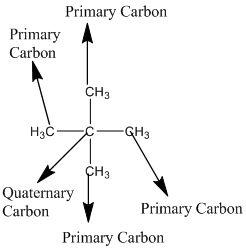
Thus, four primary carbons and one quaternary carbon atom are present in the structure.
Want to see more full solutions like this?
Chapter 12 Solutions
General, Organic, and Biological Chemistry - 4th edition
- The chemical structure of ethanol (CH, CH, OH) is shown below. Highlight each atom that is in a methyl group. H H H –C -C О — Н H H :0 :arrow_forward4. The full structural formula of three organic compounds, P, Q and R, are shown below. H H HHH H HHHH H- C-C- H H C C-C = C- H H C C C C-H H H H H H. a. State one similarity between P, Q and R in terms of their molecular formula. b. Name the homologous series that compounds P, Q and R belong to. c. State one similarity between Q and R in terms of chemical bonding. d. Which of these compounds are isomers? Explain your answer.arrow_forwardConvert each condensed formula to a Lewis structure. a.CH3(CH2)4CH(CH3)2 b. (CH3)3CCH(OH)CH2CH3 c. (CH3)2CHCHO d.(HOCH2)2CH(CH2)3C(CH3)2CH2CH3arrow_forward
- Below you will find five reaction products (labeled product 1, product 2, etc.), but you will only find four reaction schemes. Your job is to match the reaction scheme with the correct product. Simply fill in the blank with the product number (i.e. 1, 2, 3, 4, or 5). Then, state the regio-control that governs the reaction (i.e. Markovnikov or anti-Markovnikov). If the reaction does not have any regio-control then enter none. OH OH OH - OH d OH Product 2 Product 3 Product 4 Product 5 1. Hg(OAc)2, H₂O 2. NaBH4 H₂O Product 1 a. regiocontrol: Br b. regiocontrol: C. H₂O+arrow_forwardQ#3: Identify A-I and A-D. C,H&O3 1. CH3O 2. CH3Br HO HCI, H2O E Socl, CH3OH 1. CH30 excess H 2. HCI B F excess 1. CH3O 2. CH3B. HCI, H2Oarrow_forwardChoose the correct structure for each of the following compounds. a. an alcohol with molecular formula C₂H6O O CH3 CH₂-O-CH3 O CH3-C-H OCH3-CH₂-OH O CH3-O-CH3 b. an aldehyde with molecular formula C3H$O ů OCH, CCH, O CH3-CH₂-O-CH3 OCH3 CH₂-CH OH CH3-CH-CH3arrow_forward
- The chemical structure of lactic acid (C₂H₂O₂) is shown below. Highlight each atom that is in a hydroxy group. H:0: H-O-C-C-0-H HIC-H 1 H Xarrow_forwardWhat is the correct Lewis structure for acetaldehyde, an aldehyde which has the molecular formula CH,O? H :0 :0 H- H :0: H :0: H -č: FC-H H- -H H- -C-H H- C -ċ-H H- H H H a b d e a. Ob. Oc. Od. Oe.arrow_forwardWhich structure is the most stable NH₂ A. Br O NH₂ E. D. B. NH₂ NH₂ Y by x Br Rr Br C. Br NH₂arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





