
Concept explainers
Air is expanded in a steady flow process through a turbine. Initial conditions are 1300°C and 2.0 MPa absolute. Final conditions are 500°C and atmospheric pressure. Show this process on a Ts diagram. Evaluate the changes in internal energy, enthalpy, and specific entropy for this process.
The change in internal energy, enthalpy, specific entropy and show this process on a T-s diagram.
Explanation of Solution
Given:
Initial temperature
Initial pressure
Final temperature
Final pressure
Calculation:
Determine the change in internal energy.
Thus, the change in internal energy is
Determine the change in enthalpy.
Thus, the change in enthalpy is
Determine the change in specific entropy.
Thus, the change in specific entropy is
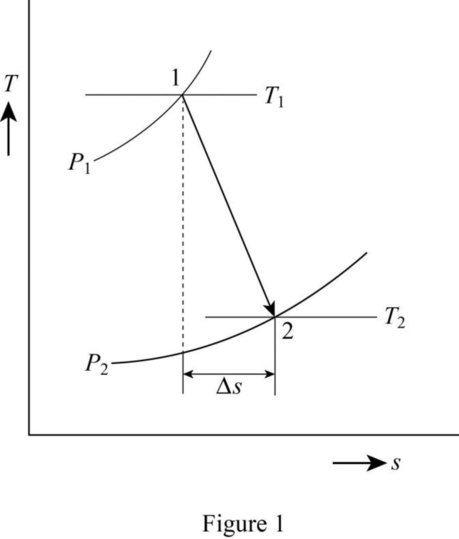
Draw the T-s diagram as in Figure (1).
Want to see more full solutions like this?
Chapter 12 Solutions
Fox and McDonald's Introduction to Fluid Mechanics
Additional Engineering Textbook Solutions
Fundamentals Of Thermodynamics
Applied Fluid Mechanics (7th Edition)
Automotive Technology: Principles, Diagnosis, and Service (5th Edition)
Engineering Mechanics: Statics & Dynamics (14th Edition)
Applied Statics and Strength of Materials (6th Edition)
Statics and Mechanics of Materials (5th Edition)
- Three cubic meter of air at a pressure of 20 kPag and 20 0 C is compressed atconstant volume to a pressure of 425 kPag. determine the following: a. change in internal energy, change in enthalpy and change in entropyarrow_forwardSteam at a temperature of 250 degree C has a specific volume of 0.15436 m3/kg . What are the pressure , specific internal energy, enthalpy, and entropy?arrow_forwardSteam at a temperature of 250°C has a specific volume of 0.15436 m3/kg. What are the pressure, specific internal energy, enthalpy, and entropy?arrow_forward
- Five kilograms of a perfect gas expands polytropically from a pressure of 1 MPa and temperature of 300 °C to a pressure of 0,2 MPa and a temperature 165 °c. If the specific heat capacities are C, = 1,011 and C, = 0,777 kJ/kgK, draw P-V of the process and calculate: 1. (a) Characteristic gas constant Change of entropy Change of internal energy polytropic index of expansion Work done Heat flow during expansion of the gas (f)arrow_forwardCompute the change in specific entropy when helium, initially at a temper-ature of 273 K and a pressure of 100 kPa, undergoes the following processes: a.An isochoric process with final pressure 250 kPa b.By computing the change in specific entropy, determine whether it is possible foran adiabatic process to bring the gas to a final temperature of 200 K and a densityof 0.3 kg/m3. If not, why not? And if so, how could it be done?arrow_forwardA vessel contains 10 kg of water at 200 kPa. Determine pressure, specific and total internal energy, specific and total enthalpy, and specific and total entropy. Consider 5 different cases/states: T = 30 ⁰C T = Tsat and x = 0 T = Tsat and x = 0.5 T = Tsat and x = 1 T = 300 ⁰C Plot these states on the Tv diagram and label magnitudes. Refer property tablesarrow_forward
- For the specific volume of wet steam, SV=(1-Xv)SV (liq) + XvSV (vapor). Entropy is also calculated this way. If a tank initially has 5kg of wet steam with mass of vapor =1 kg at 100 kPa, and it is heated such that saturated vapor remains in the tank. Assuming that the process is in constant volume, what will be the entropy change of the steam (Kj/K)?arrow_forwardIf 10 kgm/min of air are compressed isothermally from P1 = 96 kPa and Ṿ1 = 7.65 m3/min to P2 = 620 kPa, find: Non-flow work in kJ/min Change of entropy in kJ/min-K Heat in kJ/minarrow_forwardsteam at a temperature of 250°C has a specific volume of 0.15436m³/kg.What are the pressure, specific internal energy, entalphy,and entropy?arrow_forward
- If 528 kJ of heat are added to 2.25 kg of helium at constant volume process when the initial temperature is 40 degreeC, find (a) the final temperature, (b) the change in enthalpy, (c) the change in entropyarrow_forwardAir inside a cylinder has increased its pressure from 100 KPa to 200 KPa. Evaluate the change in Entropy if the intial temperature is 23 deg C. The volume of the cylinder is constant at 0.25 m^3.arrow_forwardSteam at a temperature of 250°C has a specific volume of 0.15436 cubic meter/kg. What are the pressure, specific internal energy, enthalpy, and entropyarrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





