
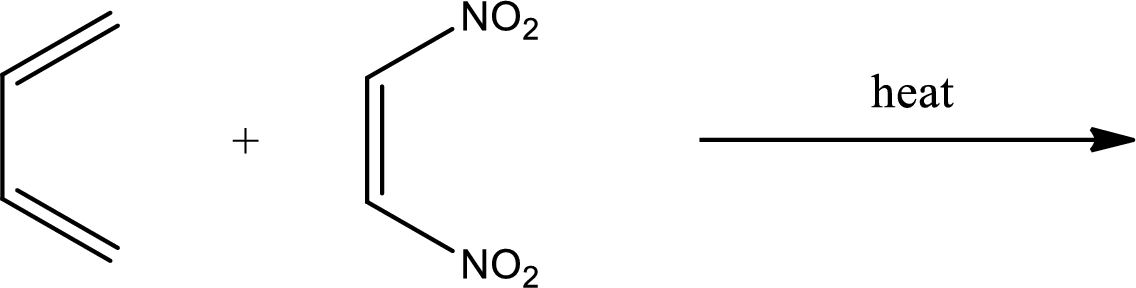
Interpretation:
Major product has to be predicted for the given reaction.
Concept Introduction:
Diels-Alder reaction is the 4+2 addition reaction. Here a total of six pi bonds are involved resulting in formation of one pi bond and two single bonds. Diels-Alder reaction occurs via a single concerted step and not by involvement of ions. If a monosubstituted ethylene is used, the product obtained contains a stereocenter. This can be represented as,
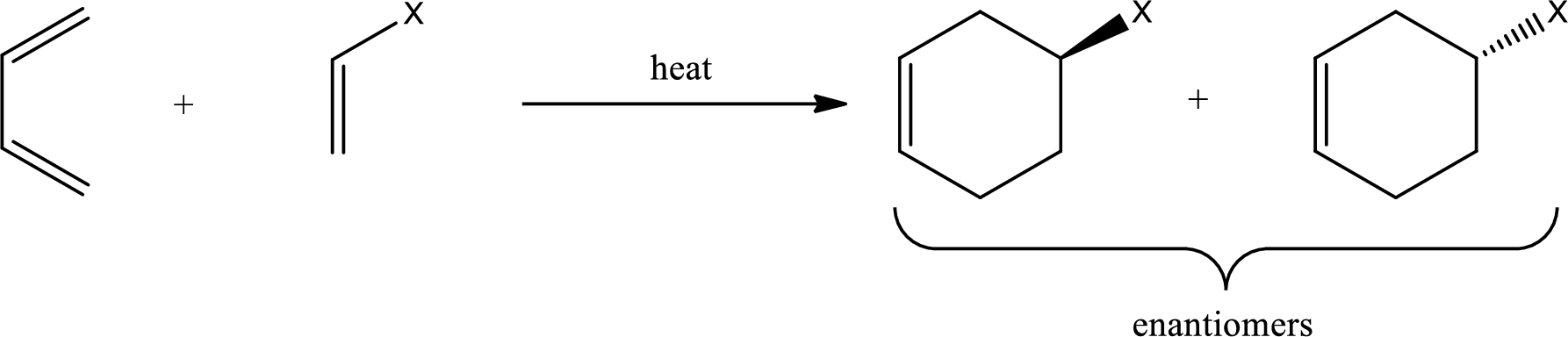
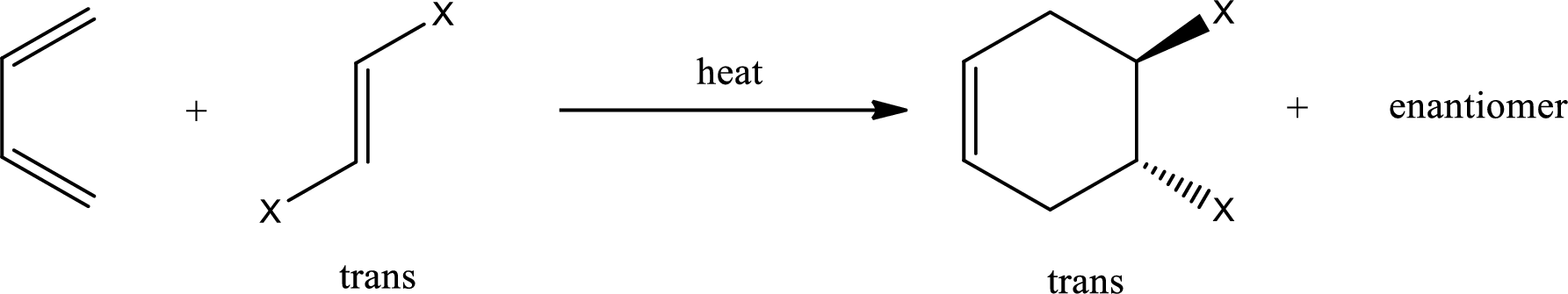
If 1,2-disubstituted ethylene is used as starting material, then the configuration of the dienophile is preserved in the product that is formed also. This means, if the dienophile is in cis-configuration, then the product formed will also have cis-configuration.

If the dienophile is in trans-configuration, then the product formed will also have trans-configuration.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
- Identify the expected major product for the following reaction:arrow_forwardFor each of the following reactions predict the major product(s)arrow_forwardPlease predict the product for each of the following reactions. Make sure to clearly indicate the correct stereochemistry for each compound:arrow_forward
