
The mole fraction of each component in polycrylonitrile material.
The mole fraction of each component in polystrene material.
The mole fraction of each component in polybutadlene material.
Answer to Problem 72AAP
The mole fraction of each component in polycrylonitrile material is
The mole fraction of each component in polystrene material is
The mole fraction of each component in polybutadlene material is
Explanation of Solution
Show the bonding structure of the polyacryonitrile, polybutadlene, and polystyrene as:
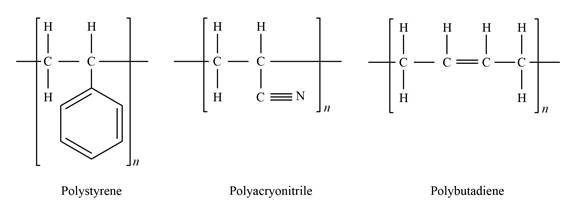
Express the mole fraction of polyacrylonitrile.
Here the number of moles of polystyrene is
Express the mole fraction of polybutadlene.
Express the mole fraction of polystyrene.
Conclusion:
The average molecular mass of the polyacrylonitrile is
The average molecular mass of the polybutadlene is
The average molecular mass of the polystyrene is
Calculate the number of moles of polyacrylonitrile of 100 g of copolymer.
Calculate the number of moles of polybutadlene of 100 g of copolymer.
Calculate the number of moles of polystyrene of 100 g of copolymer.
Substitute
Thus, the mole fraction of each component in polycrylonitrile material is
Substitute
Thus, the mole fraction of each component in polybutalene material is
Substitute
Thus, the mole fraction of each component in polystrene material is
Want to see more full solutions like this?
Chapter 10 Solutions
Foundations of Materials Science and Engineering
- For a given polymer, the activation energy for stress relaxation was measured to be 10 kJ/mol. If the stress relaxation time for this polymer at room temperature is 3,600 s, what would be the relaxation time at 100 ◦C?arrow_forwardConnecting rod (as shown below) is a car part that connects engine piston to crankshaft and converts reciprocating motion into rotating motion. It performs under load at high temperature around 150°C. Long carbon fiber thermoplastic composite (LFT) with 30% carbon fiber has been considered to replace metal (steel or aluminum) rod to improve efficiency. Based on the data for different polymers listed in the table above, which polymer(s) would you choose as the polymer matrix for the connecting rod? What is your rationale?arrow_forwardWhat monomers and polymers and give some examples of eacharrow_forward
- ( B ) A stress of 6.7 MPa is employed to one type of a polymer under constant strain . After 40 days at 20 ° C the stress is reduced to only 8.4 MPa . When the same polymer is heated to 40 ° C the relaxation time is 40 days . 1- Calculate the relaxation time constant for this material at 25 ° C ? 2- Estimate the stress after 50 days at 25 ° C ? 3- Estimate the stress relaxation activation energy for the same polymer .arrow_forwardEthylene, C2H4, and tetrafluoroethylene, C2F4, are used to make the polymers polyethylene and polytetrafluoroethylene (Teflon), respectively.(a) Draw the Lewis structures for C2H4 and C2F4, and give theideal H-C-H and F-C-F bond angles.(b) The actual H-C-H and F-C-F bond angles are 117.4° and112.4°, respectively. Explain these deviations.arrow_forwardBakelite is an example of polymer. Select one: O Thermoset O Thermoplastic O Composite O Elastomersarrow_forward
- What types of additives have to be added into a plastic coater made up PP- PVC blend, will be used to manufacture plastic tablecloth ? (They will work both in manufacturing and in use). Write them according to their order of importancearrow_forwardcis-1, 2-Dimethylcyclobutane is less stable than its trans isomer, but cis-1, 3-dimethylcyclobutane is more stable than its trans isomer.Draw the most stable conformations of both, and explain.arrow_forwardAn unknown polymer sample was found to have the following molecular weight distribution: Molecular weight range 8,000 - 20,000 20,000 - 32,000 32,000 - 44,000 44,000 - 56,000 56,000 - 68,000 68,000 -80,000 80,000 - 92,000 0.03 Knowing that the monomer is methyl methacrylate, which of the below is the number average degree of polymerization for this polymer sample? Select one: O a. O b. DP 476 O C. DP = 338 DP = 664 O d. DP 544 O e. DP = 801 Xi 0.05 0.15 0.21 0.28 0.18 0.10 Wi 0.02 0.08 0.17 0.29 0.23 0.16 0.05arrow_forward
- Calculate the number of grams in one mole of ZnC12.6H20 (calculate molecular weight)? O=16.00 H=1.01 Zn=65.39 Cl=35.45arrow_forwardAnswer the following questions. i) Write down the important properties of polymers. ii) What do you mean by a highly elastic material and provide an example for that material. . iii) Which type of polymer is used to manufacture plastic pipes and explain the characteristics of that polymer. (Minimum 2 points) -arrow_forwardThe repeat unit MW of an unknown polymeric material is 230 g/mol and has the following composition. With the information provided below calculate: -mers 80 % O The number-average MW O The weight-average MW 0 Dispersity 5 90 25 100 30 120 25 150 15arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





