
Concept explainers
(a)
Interpretation:
The increasing order of acidity of the alcohols is to be stated.
Concept introduction:
Acidity of a molecule is dependent on the value of
Answer to Problem 10.46AP
The increasing order of acidity of the alcohols is given below.
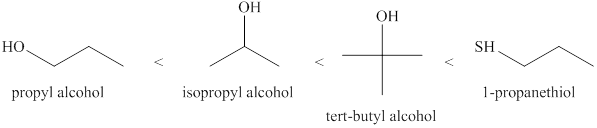
Explanation of Solution
Acidity of the molecules is dependent upon the stability of the ion formed after releasing hydrogen ion.
Sulfur atom present in
Order of the acidity is shown below in Figure 1.

Figure 1
The increasing order of acidity of the molecules is
(b)
Interpretation:
The increasing order of acidity of the molecules is to be stated.
Concept introduction:
Acidity of a molecule is dependent on the value of
Answer to Problem 10.46AP
The increasing order of acidity of the molecules is given below.
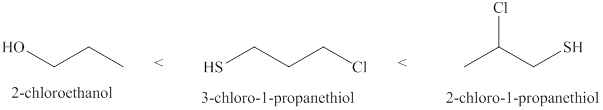
Explanation of Solution
Acidity of the molecules is dependent upon the stability of the ion formed after releasing hydrogen ion. Sulfur atom present in
Therefore, increasing order of the given molecules is shown below in Figure 2.
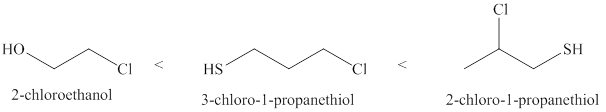
Figure 2
The increasing order of acidity of the given molecules is
(c)
Interpretation:
The increasing order of acidity of the molecules is to be stated.
Concept introduction:
Acidity of a molecule is dependent on the value of
Answer to Problem 10.46AP
The increasing order of acidity of the molecules is stated below.
Explanation of Solution
Acidity of the molecules is dependent upon the stability of the ion formed after releasing hydrogen ion. Nitrogen atom which is positively charged, stabilizes the negative charge which is generated after releasing the hydrogen ion. Therefore, structure 3 is the most stable structure with most acidic character. Position of the electronegative atom also determines the strength of an acid. Closer the electronegative atom to the generated negative charge after releasing the hydrogen ion, more is the strength of the acid.
Therefore, increasing order of the given molecules is stated below.
The increasing order of acidity of the given molecules is
(d)
Interpretation:
The increasing order of acidity of the molecules is to be stated.
Concept introduction:
Acidity of a molecule is dependent on the value of
Answer to Problem 10.46AP
The increasing order of acidity of the molecules is stated below.
Explanation of Solution
Acidity of the molecules is dependent upon the stability of the ion formed after releasing the hydrogen ion. In structure
Therefore, increasing order of acidity of the given molecules is stated below.
The increasing order of acidity of the given molecules is
(e)
Interpretation:
The increasing order of acidity of the molecules is to be stated.
Concept introduction:
Acidity of a molecule is dependent on the value of
Answer to Problem 10.46AP
The increasing order of acidity of the molecules is stated below.
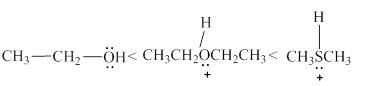
Explanation of Solution
Acidity of the molecules is dependent upon the stability of the ion formed after releasing the hydrogen ion. In structure
Therefore, increasing order of the given molecules is stated below in Figure 3.
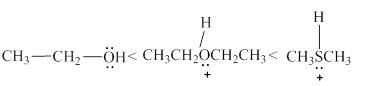
Figure 3
The increasing order of acidity of the given molecules is shown above in Figure 3.
Want to see more full solutions like this?
Chapter 10 Solutions
Organic Chemistry
- 18-28 Arrange these compounds in order of increasing acidity: benzoic acid, benzyl alcohol, phenol.arrow_forwardWill acetylene react with sodium hydride according to the following equation to form a salt and hydrogen, H2? Using pKa values given in Table 4.1, calculate Keq for this equilibrium.arrow_forwardLabel each reactant and product in this reaction as a Brønsted acid or base. HCN+NH−2↽−−⇀CN−+NH3arrow_forward
- 8) Rank the following structures in order of increasing Bronsted acidity. b A O=C OH least acidic B 1 OH most acidic с OH 21 YO!arrow_forwardList the following organic bases in order of increasing basicity, starting with the least basic one and ending with the most basic one. Please just write down the letters! Please don't use notation, just use commas between the letters! CH3 A NaOH B CH3 C CH3 N D Jarrow_forwardIndicate whether the phenol on the left of each pair is more or less acidic than the one on the right. HO N=C HO The phenol on the left is: но F3C HO The phenol on the left is: H2N >arrow_forward
- Rank the following species in order of increasing acidity. The least acidic should be on top, most acidic should be on the bottom. CFH2COOH CF2HCOOH CF3COOH CH3COOHarrow_forwardhydrogens on a carbon adjacent to a carbonyl group are far more acidic than those not adjacent to a carbonyl group. The anion derived from acetone, for example, is more stable than is the anion derived from ethane. Account for the greater stability of the anion from acetone. CH,ČCH, H CH,CH, H Acetone Ethane pK, 20.2 pK, 51arrow_forwardMethyllithium (CH3Li) is often used as a base in organic reactions. Predict the products of the following acid-base reaction.CH3CH2¬OH + CH3¬Li ¡arrow_forward
- Arrange the following molecules in the order of increasing acidity. LEAST ACIDIC < increasing acidity N < d MOST ACIDICarrow_forwardWhat is the conjugate acid of each of the following? What is the conjugate base of each? HNO2 H2Se HSe− H2C6H5O7− H3O2+arrow_forwardWhich base, CH3NH2 or (CH3)2NH, is the stronger base? Which conjugate acid, (CH3)2 NH2+ or(CH3)2 NH3+ , is the stronger acid?arrow_forward

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


