
University Physics Volume 2
18th Edition
ISBN: 9781938168161
Author: OpenStax
Publisher: OpenStax
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 27CQ
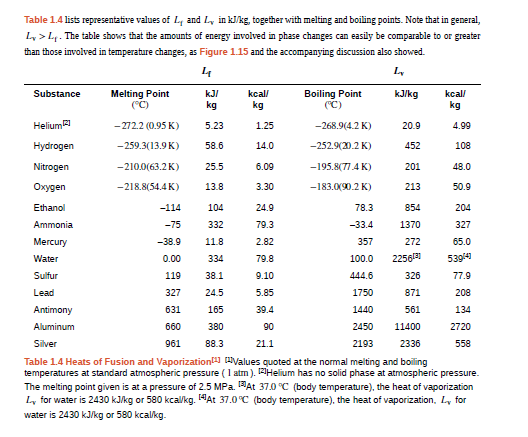
In a physics classroom demonstration, an instructor inflates a balloon by mouth and then cools it in liquid nitrogen. When cold, the shrunken balloon has a small amount of light blue liquid in it, as well as some snow-like crystals. As it warms up, the liquid boils, and part of the crystals sublime, with some crystals lingering for a while and then producing a liquid. Identify the blue liquid and the two solids in the cold balloon. Justify your identifications using data from Table 1.4.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
In a physics classroom demonstration, an instructor inflates a balloon by mouth and then cools it in liquid nitrogen. When cold, the shrunken balloon has a small amount of light blue liquid in it, as well as some snow-like crystals. As it warms up, the liquid boils, and part of the crystals sublimate, with some crystals lingering for awhile and then producing a liquid. Identify the blue liquid and the two solids in the cold balloon. Justify your identifications using data shown..
The combined gas law PV=nRT is a very useful equation. From the equation one can see the relationships between the variables. Scenario: I just climbed to the top of mount Everest and it is a beautiful day. For a celebration I want to eat a dehydrated meal with raw chicken. This is safe to do so?
Questions to think about: What pressure is present on the top of Mt. Everest? Greater or less than the pressure at sea level? How does that relate to temperature? What temperature will water boil at the top of Mt. Everest? Will that temperature be hot enough to cook the raw chicken?
Acetone has a surface tension of 0.0237 N/m at 20°C at a density of 0.792 g/cm3. It rises 1.5 cm in a certain capillary tube.
1. What is the surface tension in dynes/cm?
2. What is the radius of the capillary tube in cm?
Show complete solution.
Chapter 1 Solutions
University Physics Volume 2
Ch. 1 - Check Your Understanding Does a given reading on a...Ch. 1 - Check Your Understanding Two objects A and B have...Ch. 1 - Check Your Understanding If 25 kJ is necessary to...Ch. 1 - Check Your Understanding Explain why a cup of...Ch. 1 - Check Your Understanding Why does snow Often...Ch. 1 - Check Your Understanding Name an example from...Ch. 1 - Check Your Understanding How does the rate of heat...Ch. 1 - Check Your Understanding Explain why using a fan...Ch. 1 - Check Your Understanding How much greater is the...Ch. 1 - What does it mean to say that two systems are in...
Ch. 1 - Give an example in which A has some kind of...Ch. 1 - If a thermometer is allowed to come to equilibrium...Ch. 1 - Give an example of a physical property that varies...Ch. 1 - Pouring cold water into hot glass or ceramic...Ch. 1 - One method of getting a tight fit, say of a metal...Ch. 1 - Does it really help to run hot water over a tight...Ch. 1 - When a cold alcohol thermometer is placed in a hot...Ch. 1 - Calculate the length of a 1-meter rod of a...Ch. 1 - Noting the large stresses that can be caused by...Ch. 1 - How is heat transfer related to temperature?Ch. 1 - Describe a situation in which heat transfer...Ch. 1 - When heat transfers into a system, is the energy...Ch. 1 - The brakes in a car increase in temperature by T...Ch. 1 - A pressure cooker contains water and steam in...Ch. 1 - As shown below, which is the phase diagram for...Ch. 1 - Can carbon dioxide be liquefied at room...Ch. 1 - What is the distinction between gas and vapor?Ch. 1 - Heat transfer can cause temperature and phase...Ch. 1 - How does the latent heat of fusion of water help...Ch. 1 - What is the temperature of ice right after it is...Ch. 1 - If you place 0 ice into 0 water in an insulated...Ch. 1 - What effect does condensation on a glass of ice...Ch. 1 - In Miami, Florida, which has a very humid climate...Ch. 1 - In winter, it is often warmer in San Francisco...Ch. 1 - Freeze-dried foods have been dehydrated in a...Ch. 1 - In a physics classroom demonstration, an...Ch. 1 - Mechanisms of Heat Transfer What are the main...Ch. 1 - When our bodies get too warm, they respond by...Ch. 1 - Shown below is a cut-away drawing of a thermos...Ch. 1 - Some electric stoves have a flat ceramic surface...Ch. 1 - Loose-fitting white clothing covering most of the...Ch. 1 - One way to make a fireplace more energy-efficient...Ch. 1 - On cold, clear nights horses will sleep under the...Ch. 1 - When watching a circus during the day in a large,...Ch. 1 - Satellites designed to observe the radiation from...Ch. 1 - Why are thermometers that ale used in weather...Ch. 1 - Putting a lid on a boiling pot greatly reduces the...Ch. 1 - Your house will be empty for a while in cold...Ch. 1 - You pour coffee into an unlidded cup, intending to...Ch. 1 - Broiling is a method of cooking by radiation,...Ch. 1 - On a cold winter morning, why does the metal of a...Ch. 1 - While traveling outside the United States, you...Ch. 1 - What are the following temperatures on the Kelvin...Ch. 1 - (a) Suppose a cold front blows into your locale...Ch. 1 - An Associated Press article on climate change...Ch. 1 - (a) At what temperature do the Fahrenheit and...Ch. 1 - A person taking a reading of the temperature in a...Ch. 1 - The height of the Washington Monument is measured...Ch. 1 - How much taller does the Eiffel Tower become at...Ch. 1 - What is the change in length of a 3.00-cm-long...Ch. 1 - How large an expansion gap should be left between...Ch. 1 - You are looking to buy a small piece of land in...Ch. 1 - Global warming will produce rising sea levels...Ch. 1 - (a) Suppose a meter stick made of steel and one...Ch. 1 - (a) If a 500-mL glass beaker is filled to the brim...Ch. 1 - Most cars have a coolant reservoir to catch...Ch. 1 - A physicist makes a cup of instant coffee and...Ch. 1 - The density of water at 0 C is very nearly 1000...Ch. 1 - Show that =3a , by calculating the infinitesimal...Ch. 1 - Calorimetry On a hot day, the temperature of an...Ch. 1 - To sterilize a 50.0-g glass baby bottle, we must...Ch. 1 - The same heat transfer into identical masses of...Ch. 1 - Rubbing your hands together warms them by...Ch. 1 - A 0.250-kg block of a pule material is heated from...Ch. 1 - Suppose identical amounts of heat transfer into...Ch. 1 - (a) The number of kilocalories in food is...Ch. 1 - Following vigorous exercise, the body...Ch. 1 - In a study of healthy young men[1], doing 20...Ch. 1 - A 1.28-kg sample of water at 10.0 is in a...Ch. 1 - Repeat the preceding problem, assuming the water...Ch. 1 - How much heat transfer (in kilocalories) is...Ch. 1 - A bag containing 0 ice is much more effective in...Ch. 1 - (a) How much heat transfer is required to raise...Ch. 1 - Condensation on a glass of ice water causes the...Ch. 1 - On a trip, you notice that a 3.50-kg bag of ice...Ch. 1 - On a certain dry sunny day, a swimming pool 's...Ch. 1 - (a) How much heat transfer is necessary to raise...Ch. 1 - In 1986, an enormous iceberg broke away from the...Ch. 1 - How many grams of coffee must evaporate from 350 g...Ch. 1 - (a) It is difficult to extinguish a fire on a...Ch. 1 - The energy released from condensation in...Ch. 1 - To help prevent frost damage, 4.00 kg of water at...Ch. 1 - A 0.250-kg aluminum bowl holding 0.800 kg of soup...Ch. 1 - A 0.0500-kg ice cube at 30.0 is placed in 0.400...Ch. 1 - If you pour 0.0100 kg of 20.0 water onto a...Ch. 1 - Indigenous people sometimes cook in watertight...Ch. 1 - What would the final temperature of the pan and...Ch. 1 - (a) Calculate the rate of heat conduction through...Ch. 1 - The rate of heat conduction out of a window on a...Ch. 1 - Calculate the rate of heat conduction out of the...Ch. 1 - Suppose you stand with one foot on ceramic...Ch. 1 - A man consumes 3000 kcal of food in one day,...Ch. 1 - A firewalker runs across a bed of hot coals...Ch. 1 - (a) What is the rate of heat conduction through...Ch. 1 - A warms transfers energy by conduction through its...Ch. 1 - Compare the rate of heat conduction through a...Ch. 1 - Suppose a person is covered head to foot by wool...Ch. 1 - Some stove tops are smooth ceramic for easy...Ch. 1 - One easy way to reduce heating (and cooling) costs...Ch. 1 - Many decisions are made on the basis of the...Ch. 1 - In 1701, the Danish astronomer Ole Rømer proposed...Ch. 1 - What is the percent error of thinking the melting...Ch. 1 - An engineer wants to design a structure in which...Ch. 1 - How much stress is cleated in a steel beam if its...Ch. 1 - A brass rod (Y=90109N/m2), with a diameter of...Ch. 1 - A mercury thermometer still in use for meteorology...Ch. 1 - Even when shut down after a period of normal use,...Ch. 1 - You leave a pastry in the refrigerator on a plate...Ch. 1 - Two solid spheres, A and B, made of the same...Ch. 1 - In some countries, liquid nitrogen is used on...Ch. 1 - Some gun fanciers make their own bullets, which...Ch. 1 - A 0.800-kg iron cylinder at a temperature of...Ch. 1 - Repeat the preceding problem with 2.00 kg of ice...Ch. 1 - Repeat the preceding problem with 0.500 kg of ice,...Ch. 1 - A 30.0-g ice cube at its melting point is dropped...Ch. 1 - (a) Calculate the rate of heat conduction through...Ch. 1 - (a) An exterior wall of a house is 3 m tall and 10...Ch. 1 - For the human body, what is the rate of heat...Ch. 1 - You have a Dewar flask (a laboratory vacuum flask)...Ch. 1 - An infrared heater for a sauna has a surface area...Ch. 1 - (a) Deter-nine the power of radiation from the Sun...Ch. 1 - A pendulum is made of a rod of length L and...Ch. 1 - At temperatures of a few hundred kelvins the...Ch. 1 - In a calorimeter of negligible heat capacity, 200...Ch. 1 - An astronaut performing an extra-vehicular...Ch. 1 - The goal in this problem is to find the growth of...Ch. 1 - As the very first rudiment of climatology,...Ch. 1 - Let's stop ignoring the greenhouse effect and...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Draw separate free-body diagrams for each block and for the spring immediately after release. Indicate separate...
Tutorials in Introductory Physics
What is synchronous rotation, and why is it so common among the jovian moons?
Life in the Universe (4th Edition)
Choose the best answer to each of the following. Explain your reasoning. What is the current temperature of the...
Cosmic Perspective Fundamentals
3. Example 8.3 and some of the problems in this chapter describe how, when an object explodes into two fragment...
College Physics (10th Edition)
Give the metric symbol, or abbreviation, for each prefix.
13. mega
Applied Physics (11th Edition)
Can the voltage across a batterys terminals differ from the batterys rated voltage? Explain.
Essential University Physics: Volume 2 (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Dry air is primarily composed of nitrogen. In a classroom demonstration, a physics instructor pours 2.00 L of liquid nitrogen into a beaker. After the nitrogen evaporates, how much volume does it occupy if its density is equal to that of the dry air at sea level? Liquid nitrogen has a density of 808 kg/m3.arrow_forwardThe average human has a density of 945 kg/m3 after in haling and 1 020 kg/m3 after exhaling. (a) Without making any swimming movements, what percentage of the human body would be above the surface in the Dead Sea (a body of water with a density of about 1 230 kg/m3) in each of these cases? (b) Given that bone and muscle are denser than fat, what physical characteristics differentiate sinkers (those who tend to sink in water) from floaters (those who readily float)?arrow_forwardWhat mass of silver has a volume of 150 cm3? A certain gas has a volume of 600 cm3. What is its volume when the pressure is tripled? Consider the gas in # 5 above. What is its volume if the pressure is changed to one-third its original value?arrow_forward
- Gas flows through a gray pipe with various inner diameters from left to right. You attach a clear “E” shape pipe with fluid inside and observe the liquid levels rise as depicted in the figure. Using your knowledge of fluids in motion, rank in order from largest to smallest the fluid speeds Va ,Vb ,and Vc at points a,b, and c respectively. Explain your answer.arrow_forwardI asked this under chemistry but directed me to physics...thanks. Suppose you have two 100-mL graduated cylinders. In each cylinder there is 40.0 mL of water. You also have two cubes: one is lead (density = 11.3 g/mL), and the other is aluminum (density = 2.70 g/mL). Each cube measures 1.9 cm on each side. After you carefully lower each cube into the water of its own cylinder, what will the new water level be in each of the cylinders? I know that I will need to add the 40.0 mL water with the lead cube and then 40.0 mL water with the aluminum cube, but I can't figure out how to find the mass of the cubes since they are in cm. Thanks!arrow_forwardA sphygmomanometer is a device used to measure blood pressure, typically consisting of an inflatable cuff and a manometer used to measure air pressure in the cuff. In a mercury sphygmomanometer, blood pressure is related to the difference in heights between two columns of mercury. The mercury sphygmomanometer shown in the figure below contains air at the cuff pressure P. Po The difference in mercury heights between the left tube and the right tube is 102 mmHg systolic blood pressure Pgauge = 0.102 m, a normal systolic reading. What is the gauge in pascals? The density of mercury is h p = 13.6 x 103 kg/m³ and the ambient pressure is Po 1.01 x 105 Pa. %3Darrow_forward
- Calculate the gauge pressures inside the given spherical Liquid Surface tension (N/m) bubbles. Use the surface tension values given in the table. 0.0728 water A 1.65 cm radius bubble of glycerol. glycerol 0.0631 toluene 0.0277 acetone 0.0237 ethanol 0.0223 glycerol bubble gauge pressure: N/m? isopropanol 0.0217 A 2.45 cm radius bubble of acetone. acetone bubble gauge pressure: N/m2 A 2.45 cm radius bubble of ethanol. ethanol bubble gauge pressure: N/m2arrow_forwardA scientist collected the following data, which show how the pressure of water vapor changes with temperature. Temperature (°C) Pressure (mm Hg) 0 4.5 10 9.6 20 19.1 30 33.2 40 58.7 50 95.6 What would a scientist do to best communicate these data to other scientists in a presentation? Replace the table with text highlighting pressures. Change the unit of pressure to atm. Make a graph for better visual representation. Display only rows 1, 3, and 6 of the data to simplify results.arrow_forward22. The diagram shows a closed container partially filled with liquid. Point Y is 1 m below the lid, and Point Z is 3 m below the lid. If the absolute pressure at Point Y is 13,000 Pa, and the absolute pressure at Point Z is 29,000 Pa, what is the pressure at the liquid/gas interface, Point X?: * -lid gas- "Z liquid OA. 3,000 Pa OB. 4,000 Pa OC. 5,000 Pa OD. 7,000 Pa OE. 8,000 Paarrow_forward
- Mercury and gas are in equilibrium in the closed pill with a vertical cross section as in the figure, and the total pressures at the K, L, M points are Pk, Pl and Pm. Which ones are correct accordingly? 1.Pk=Pl 2.Pl=Pm 3.Pk=Pmarrow_forwardThe density of a ring that may be silver, platinum, or a white gold alloy is being measured. Its mass under dry conditions is 2.09 g. In water at 20 °C having a density of 0.998 g/cm³, the mass is found to be 1.89 g. Think & Prepare 1. To find the density of an object you need its mass and volume. How can you accurately determine the volume of the ring? 2. How can you use the weightloss of the ring in water to determine the volume of the ring? What is the density of the ring? g/cm³ For comparison with the final answer (no need to enter an answer for this comparison), = density of gold 19.3 g/cm³ density of platinum = 21.45 g/cm³ density of silver = 10.49 g/cm³ density range of alloy = 14.7 g/cm³ to 16.9 g/cm³arrow_forwardQuestion: Lets think about the pressure difference for bubbles in soda. If the bubble has a radius of 1 um and a surface tension of 0.072 N/m what is the pressure difference between the bubble and the soda? Give your answer in 3 significant figures. Solution: The pressure difference is Paarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

Glencoe Physics: Principles and Problems, Student...
Physics
ISBN:9780078807213
Author:Paul W. Zitzewitz
Publisher:Glencoe/McGraw-Hill
A Level Physics – Ideal Gas Equation; Author: Atomi;https://www.youtube.com/watch?v=k0EFrmah7h0;License: Standard YouTube License, CC-BY