
Concept explainers
Draw a three-dimensional representation for each molecule. Indicate which ones have a dipole moment and in what direction it is pointing.
- (a) CH3F
- (b) CH2Cl2
- (c) CH2ClBr
- (d) CFCl3
- (e) CCl4
- (f) CH2=CCl2
- (g) CH2=CHCl
- (h) HC≡C—C≡CH
- (i) CH3C≡N
- (j) (CH3)2C=O
- (k) BrCH=CHBr (two answers)
(a)
Interpretation:
The Three-dimensional representation for the molecule has to be drawn and dipole movement, the direction of its dipole moment has to be identified.
Concept introduction:
Polar Molecules:
Polar Molecules contains partial positive and partial negative charge in the molecule due its electronegativity difference between the molecules.
Dipole moment:
The charge separation of the molecule produces dipole moment. Dipole moment arises between two ions in an ionic bond or covalent bond.
Dipole moment depends on the differences in electronegativity of the atom in the molecule. The electronegativity is more, the larger the dipole moment. The dipole moment is calculated from the polarity of the molecule.
Explanation of Solution
The given compound is shown below,
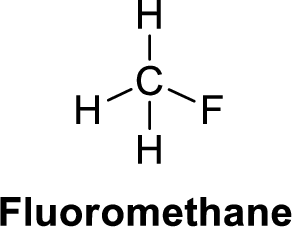
Fluoromethane is polar molecule because it has more electronegativity fluorine atom. The structure of fluoromethane is given below,

Fluorine is more electronegative than carbon and hydrogen. Carbon is more electronegative than hydrogen.
The direction of dipole moment in dichloromethane is given below,

The Three-dimensional representation for the molecule is shown below,
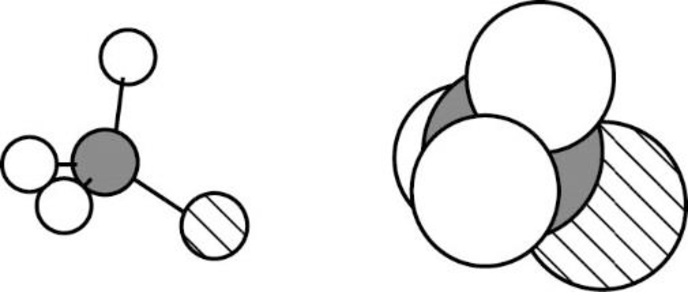
Figure 1
(b)
Interpretation:
Three-dimensional representation for the molecule has to be drawn and dipole movement, the direction of its dipole moment has to be identified.
Concept introduction:
Polar Molecules:
Polar Molecules contains partial positive and partial negative charge in the molecule due its electronegativity difference between the molecules.
Dipole moment:
The charge separation of the molecule produces dipole moment. Dipole moment arises between two ions in an ionic bond or covalent bond.
Dipole moment depends on the differences in electronegativity of the atom in the molecule. The electronegativity is more, the larger the dipole moment. The dipole moment is calculated from the polarity of the molecule.
Explanation of Solution
The given compound is shown below,
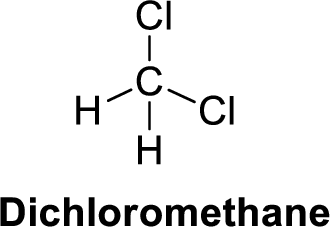
Dichloromethane is polar molecule because it has more electronegativity chlorine atom. The structure of dichloromethane is given below,

Chlorine is more electronegative than carbon and hydrogen. Carbon is more electronegative than hydrogen.
The direction of dipole moment in dichloromethane is given below,
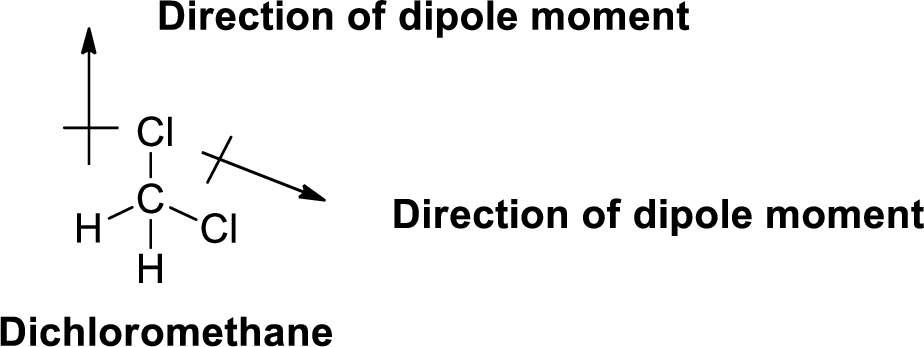
The Three-dimensional representation for the molecule is shown below,
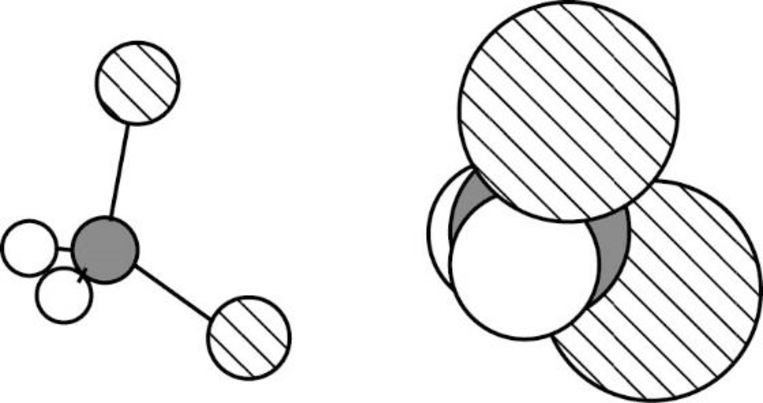
Figure 2
(c)
Interpretation:
Three-dimensional representation for the molecule has to be drawn and dipole movement, the direction of its dipole moment has to be identified.
Concept introduction:
Polar Molecules:
Polar Molecules contains partial positive and partial negative charge in the molecule due its electronegativity difference between the molecules.
Dipole moment:
The charge separation of the molecule produces dipole moment. Dipole moment arises between two ions in an ionic bond or covalent bond.
Dipole moment depends on the differences in electronegativity of the atom in the molecule. The electronegativity is more, the larger the dipole moment. The dipole moment is calculated from the polarity of the molecule.
Explanation of Solution
The given compound is shown below,
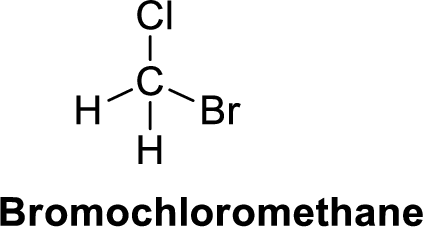
Bromochloromethane is polar molecule because it has more electronegativity chlorine and bromine atom. The structure of Bromochloromethane is given below,

Chlorine is more electronegative than carbon and hydrogen, similarly bromine is more electronegative than carbon and hydrogen. Carbon is more electronegative than hydrogen.
The direction of dipole moment in Bromochloromethane is given below,
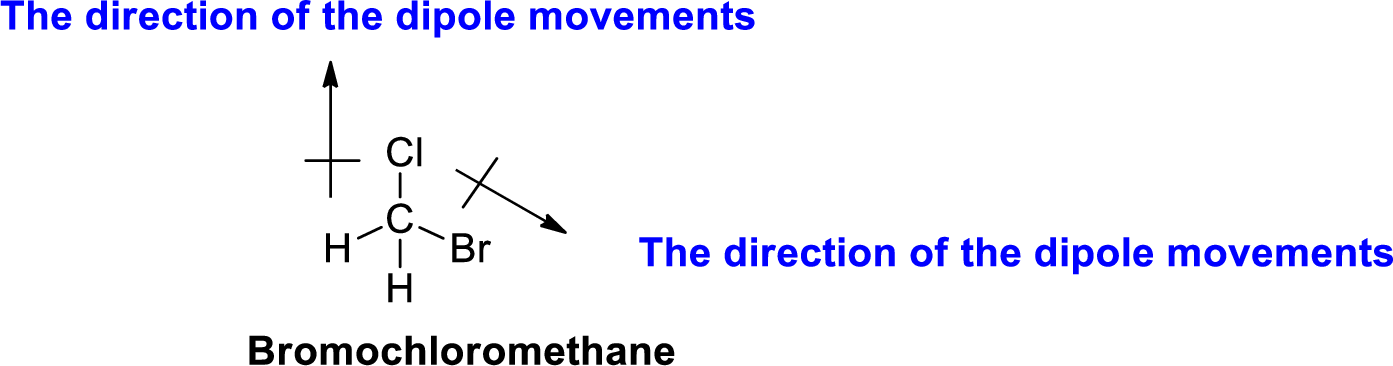
The Three-dimensional representation for the molecule is shown below,
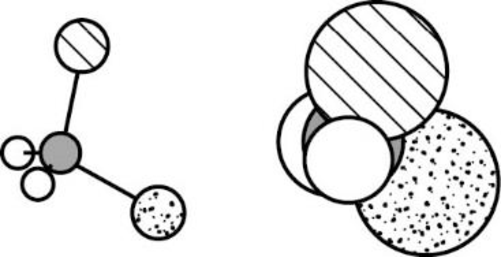
Figure 3
(d)
Interpretation:
Three-dimensional representation for the molecule has to be drawn and dipole movement, the direction of its dipole moment has to be identified.
Concept introduction:
Polar Molecules:
Polar Molecules contains partial positive and partial negative charge in the molecule due its electronegativity difference between the molecules.
Dipole moment:
The charge separation of the molecule produces dipole moment. Dipole moment arises between two ions in an ionic bond or covalent bond.
Dipole moment depends on the differences in electronegativity of the atom in the molecule. The electronegativity is more, the larger the dipole moment. The dipole moment is calculated from the polarity of the molecule.
Explanation of Solution
The given compound is shown below,
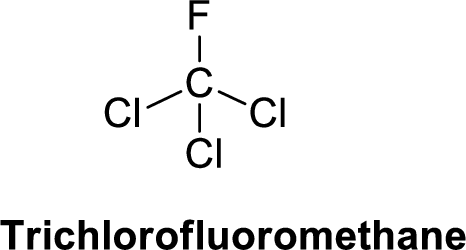
Trichlorofluoromethane is polar molecule because it has more electronegativity fluorine atom and chlorine atom. The structure of Trichlorofluoromethane is given below,
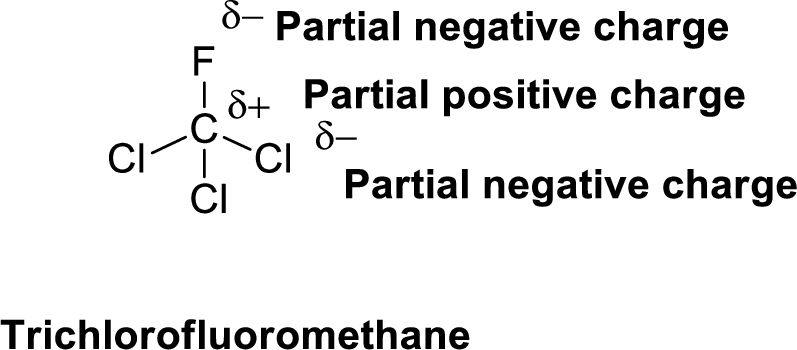
Fluorine is more electronegative than chlorine and carbon, similarly chlorine is more electronegative than carbon.
The direction of dipole moment in Trichlorofluoromethane is given below,
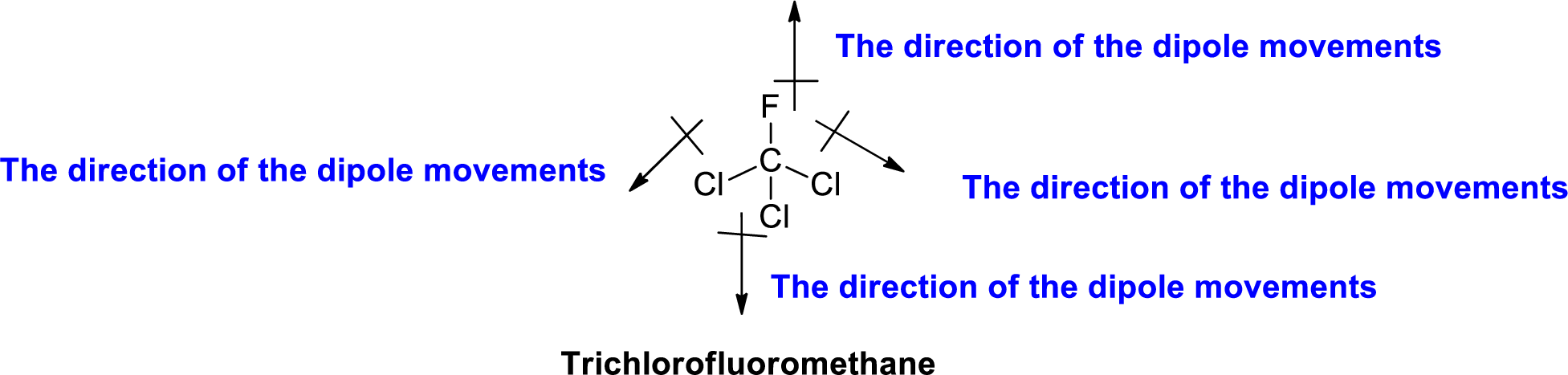
The Three-dimensional representation for the molecule is shown below,
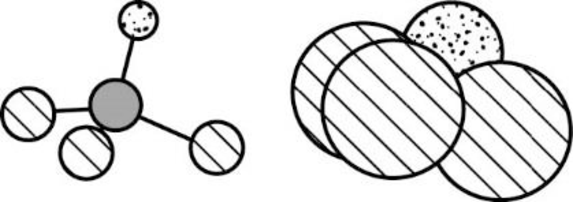
Figure 4
(e)
Interpretation:
Three-dimensional representation for the molecule has to be drawn and dipole movement, the direction of its dipole moment has to be identified.
Concept introduction:
Polar Molecules:
Polar Molecules contains partial positive and partial negative charge in the molecule due its electronegativity difference between the molecules.
Dipole moment:
The charge separation of the molecule produces dipole moment. Dipole moment arises between two ions in an ionic bond or covalent bond.
Dipole moment depends on the differences in electronegativity of the atom in the molecule. The electronegativity is more, the larger the dipole moment. The dipole moment is calculated from the polarity of the molecule.
Explanation of Solution
The given compound is shown below,
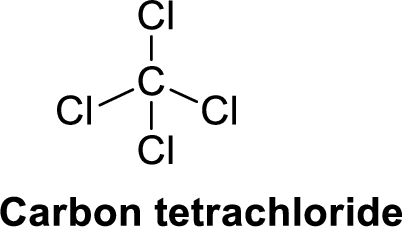
Carbon tetrachloride is polar molecule because it has more electronegativity chlorine. The structure of Carbon tetrachloride is given below,

Chlorine is more electronegative than carbon. The direction of dipole moment in Carbon tetrachloride is given below,
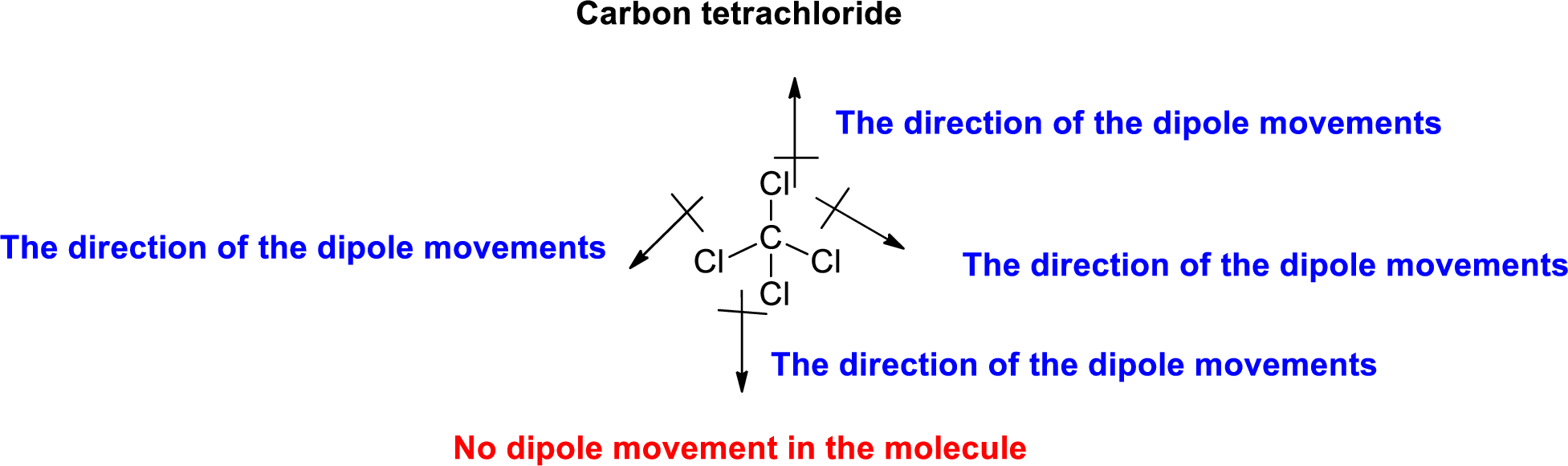
The Three-dimensional representation for the molecule is shown below,
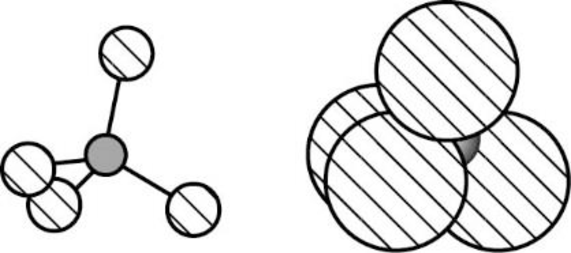
Figure 5
(f)
Interpretation:
Three-dimensional representation for the molecule has to be drawn and dipole movement, the direction of its dipole moment has to be identified.
Concept introduction:
Polar Molecules:
Polar Molecules contains partial positive and partial negative charge in the molecule due its electronegativity difference between the molecules.
Dipole moment:
The charge separation of the molecule produces dipole moment. Dipole moment arises between two ions in an ionic bond or covalent bond.
Dipole moment depends on the differences in electronegativity of the atom in the molecule. The electronegativity is more, the larger the dipole moment. The dipole moment is calculated from the polarity of the molecule.
Explanation of Solution
The given compound is shown below,
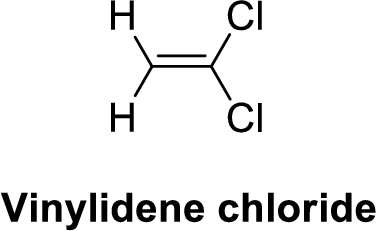
Vinylidene chloride is polar molecule because it has more electronegativity chlorine. The structure of Vinylidene chloride is given below,

Chlorine is more electronegative than carbon and hydrogen, similarly Carbon is more electronegative than hydrogen.
The direction of dipole moment in Vinylidene chloride is given below,

The Three-dimensional representation for the molecule is shown below,
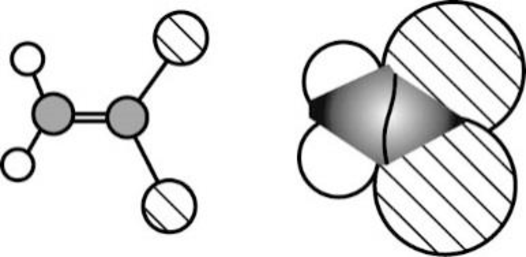
Figure 6
(g)
Interpretation:
Three-dimensional representation for the molecule has to be drawn and dipole movement, the direction of its dipole moment has to be identified.
Concept introduction:
Polar Molecules:
Polar Molecules contains partial positive and partial negative charge in the molecule due its electronegativity difference between the molecules.
Dipole moment:
The charge separation of the molecule produces dipole moment. Dipole moment arises between two ions in an ionic bond or covalent bond.
Dipole moment depends on the differences in electronegativity of the atom in the molecule. The electronegativity is more, the larger the dipole moment. The dipole moment is calculated from the polarity of the molecule.
Explanation of Solution
The given compound is shown below,
Vinyl chloride is polar molecule because it has more electronegativity chlorine. The structure of Vinyl chloride is given below,

Chlorine is more electronegative than carbon and hydrogen, similarly Carbon is more electronegative than hydrogen.
The direction of dipole moment in Vinyl chloride is given below,

The Three-dimensional representation for the molecule is shown below,
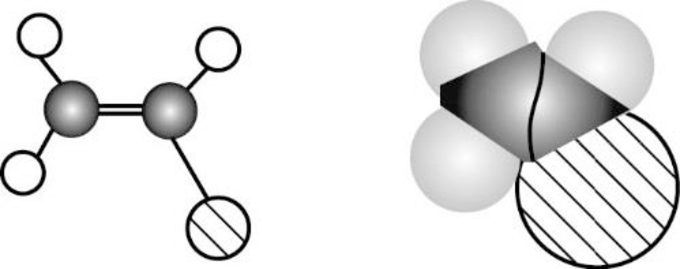
Figure 7
(h)
Interpretation:
Three-dimensional representation for the molecule has to be drawn and dipole movement, the direction of its dipole moment has to be identified.
Concept introduction:
Polar Molecules:
Polar Molecules contains partial positive and partial negative charge in the molecule due its electronegativity difference between the molecules.
Dipole moment:
The charge separation of the molecule produces dipole moment. Dipole moment arises between two ions in an ionic bond or covalent bond.
Dipole moment depends on the differences in electronegativity of the atom in the molecule. The electronegativity is more, the larger the dipole moment. The dipole moment is calculated from the polarity of the molecule.
Explanation of Solution
The given compound is non-polar molecule. The structure of compound is given below,
Carbon is more electronegative than hydrogen.
The direction of dipole moment in of the given compound is given below,
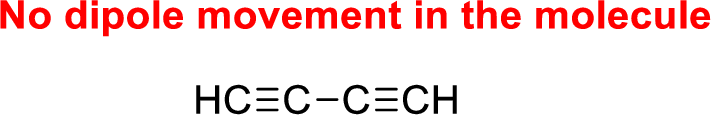
The Three-dimensional representation for the molecule is shown below,
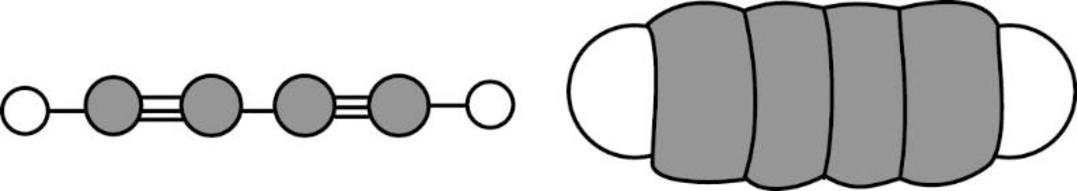
Figure 8
(i)
Interpretation:
Three-dimensional representation for the molecule has to be drawn and dipole movement, the direction of its dipole moment has to be identified.
Concept introduction:
Polar Molecules:
Polar Molecules contains partial positive and partial negative charge in the molecule due its electronegativity difference between the molecules.
Dipole moment:
The charge separation of the molecule produces dipole moment. Dipole moment arises between two ions in an ionic bond or covalent bond.
Dipole moment depends on the differences in electronegativity of the atom in the molecule. The electronegativity is more, the larger the dipole moment. The dipole moment is calculated from the polarity of the molecule.
Explanation of Solution
The given compound is shown below,
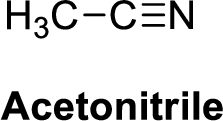
Acetonitrile is polar molecule because it has more electronegativity nitrogen atom. The structure of Acetonitrile is given below,

Nitrogen is more electronegative than carbon and hydrogen, similarly Carbon is more electronegative than hydrogen.
The direction of dipole moment in acetonitrile is given below,

The Three-dimensional representation for the molecule is shown below,
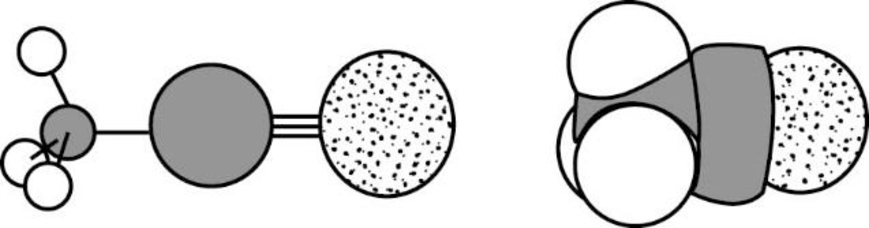
Figure 9
(j)
Interpretation:
Three-dimensional representation for the molecule has to be drawn and dipole movement, the direction of its dipole moment has to be identified.
Concept introduction:
Polar Molecules:
Polar Molecules contains partial positive and partial negative charge in the molecule due its electronegativity difference between the molecules.
Dipole moment:
The charge separation of the molecule produces dipole moment. Dipole moment arises between two ions in an ionic bond or covalent bond.
Dipole moment depends on the differences in electronegativity of the atom in the molecule. The electronegativity is more, the larger the dipole moment. The dipole moment is calculated from the polarity of the molecule.
Explanation of Solution
The given compound is shown below,
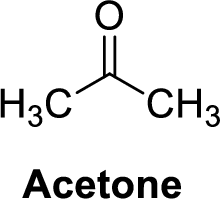
Acetone is polar molecule because it has more electronegativity oxygen atom. The structure of acetone is given below,
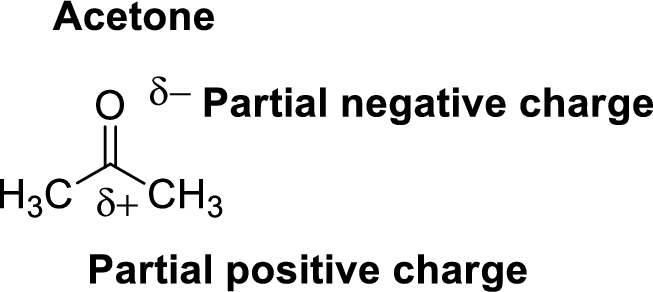
Oxygen is more electronegative than carbon and hydrogen, similarly Carbon is more electronegative than hydrogen.
The direction of dipole moment in acetone is given below,
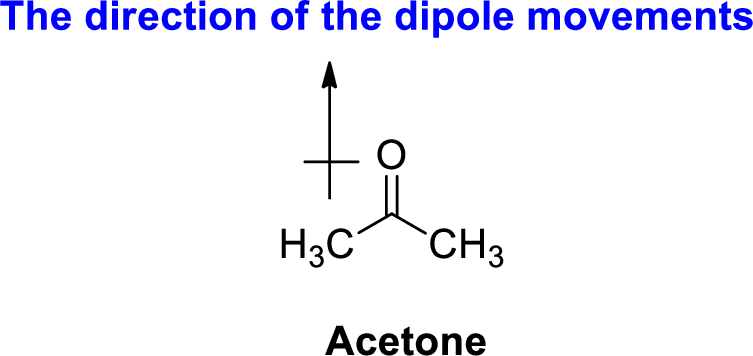
The Three-dimensional representation for the molecule is shown below,
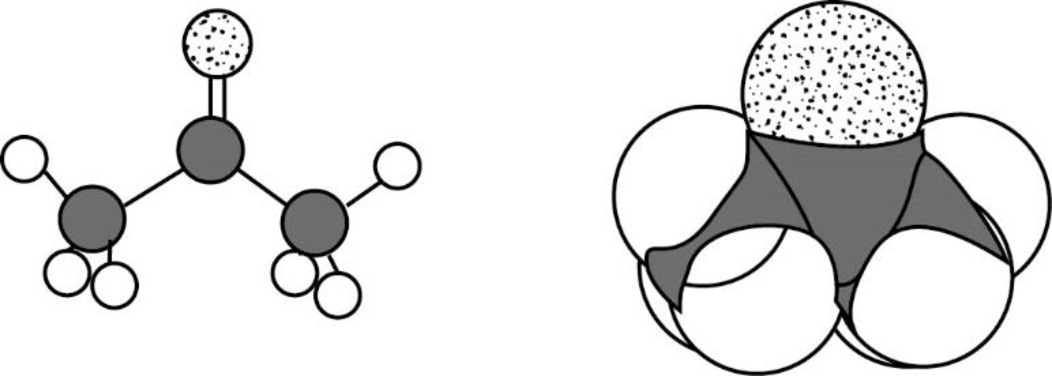
Figure 10
(k)
Interpretation:
Three-dimensional representation for the molecule has to be drawn and dipole movement, the direction of its dipole moment has to be identified.
Concept introduction:
Polar Molecules:
Polar Molecules contains partial positive and partial negative charge in the molecule due its electronegativity difference between the molecules.
Dipole moment:
The charge separation of the molecule produces dipole moment. Dipole moment arises between two ions in an ionic bond or covalent bond.
Dipole moment depends on the differences in electronegativity of the atom in the molecule. The electronegativity is more, the larger the dipole moment. The dipole moment is calculated from the polarity of the molecule.
Explanation of Solution
The given compound 1,2-Dibromoethylene, therefore it has two form
- (i) Cis 1,2-Dibromoethylene
- (ii) Trans- 1,2-Dibromoethylene

1,2-Dibromoethylene is polar molecule because it has more electronegativity bromine atom. The structure of 1,2-Dibromoethylene is given below,
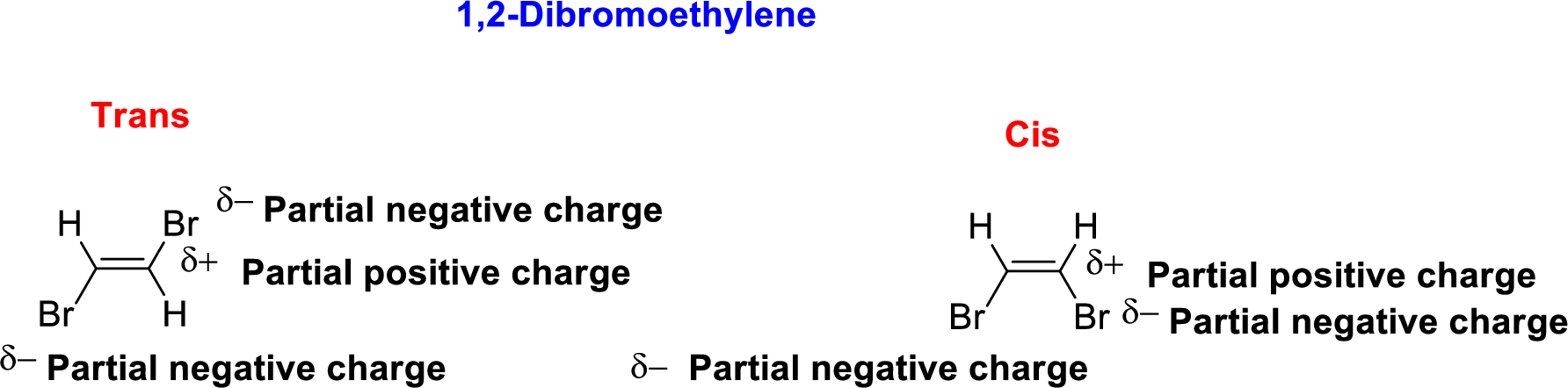
Bromine is more electronegative than carbon and hydrogen, similarly Carbon is more electronegative than hydrogen.
The direction of dipole moment in 1,2-Dibromoethylene is given below,
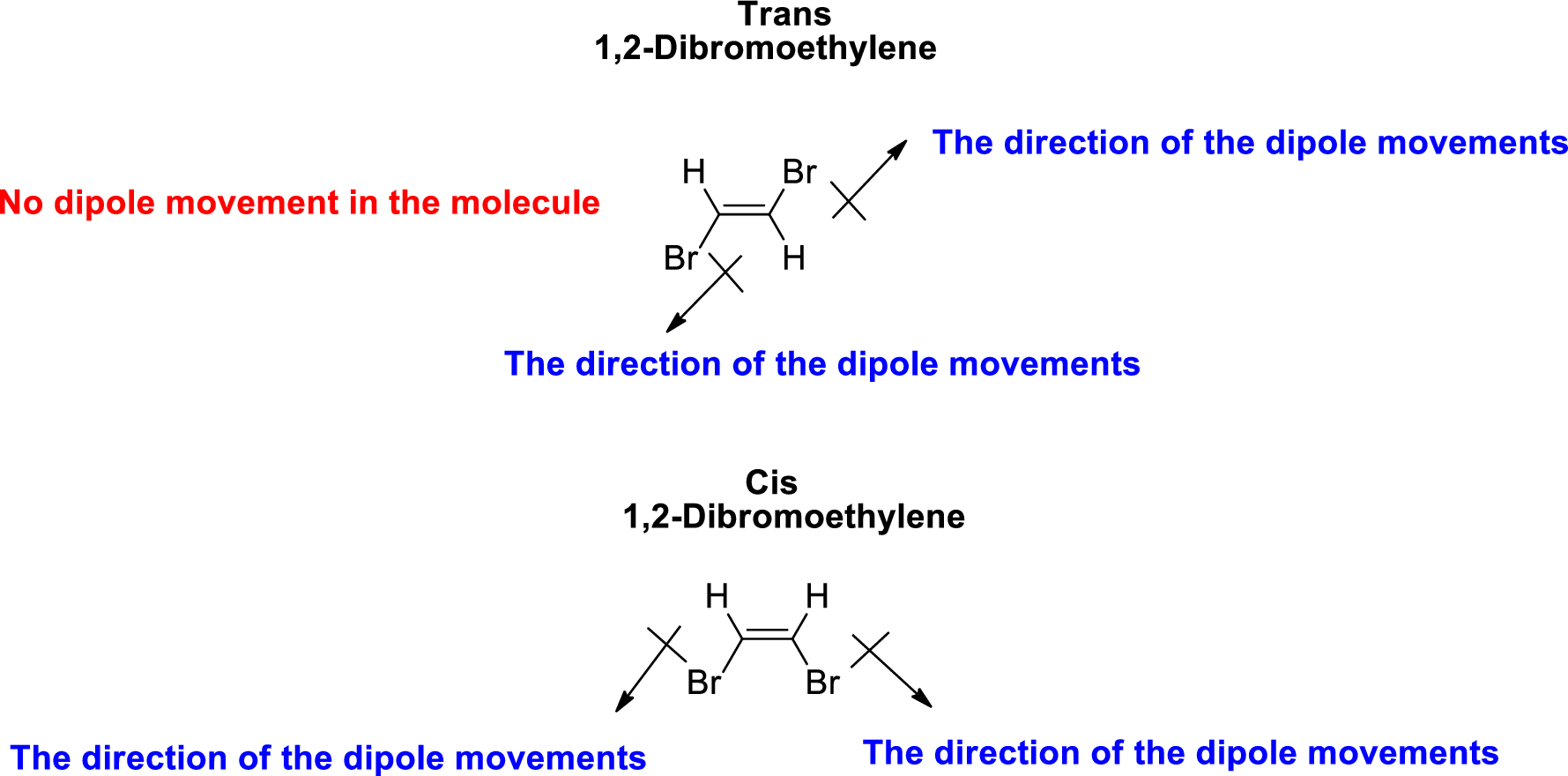
The Three-dimensional representation for the molecule is shown below,
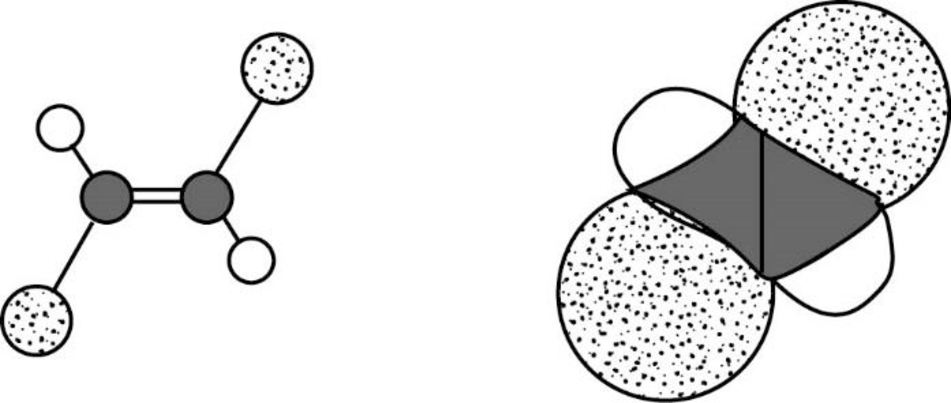
Figure 11
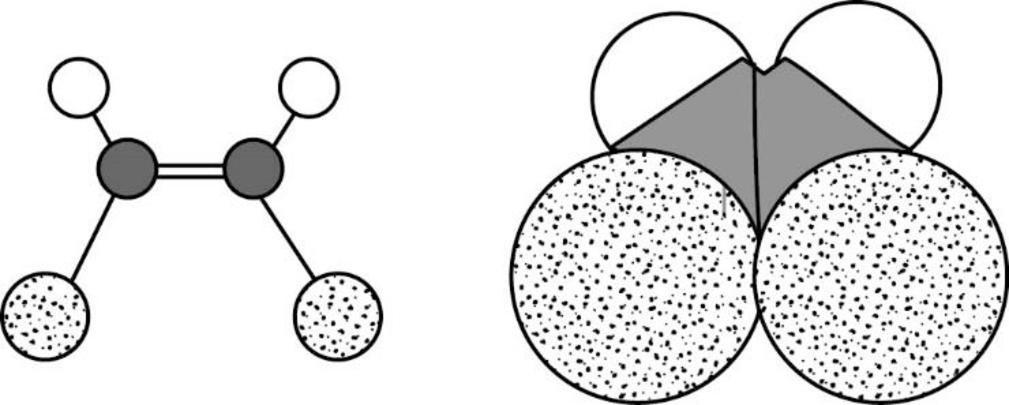
Figure 12
Want to see more full solutions like this?
Chapter 1 Solutions
Organic Chemistry
- (a) Triazine, C3 H3 N3, is like benzene except that in triazineevery other C¬H group is replaced by a nitrogen atom.Draw the Lewis structure(s) for the triazine molecule. (b) Estimatethe carbon–nitrogen bond distances in the ring.arrow_forward(a) Triazine, C3 H3 N3, is like benzene except that in triazineevery other C¬H group is replaced by a nitrogen atom. Draw the Lewis structure(s) for the triazine molecule. (b) Estimatethe carbon–nitrogen bond distances in the ring.arrow_forwardFor each of the following covalent bonds: (a) use the symbols δ+ and δ- to indicate the direction of polarity (if any).(a) C-F; (b) N-Br; (c) B-C; (d) Si-H(b) Rank the following covalent bonds in order of increasing polarity. (i) C-H, O-H, N-H; (ii) C-N, C-O, B-O; (iii) C-P, C-S, C-Narrow_forward
- Which of the following molecules does not have a dipole moment? (a) CH3Cl (b) CH2Cl2 (c) CFCl3 (d) CHCl3arrow_forwardChloral (Cl₃C—CH=O) forms a monohydrate, chloralhydrate, the sleep-inducing depressant called “knockout drops”in old movies. (a) Write two possible structures for chloral hy-drate, one involving hydrogen bonding and one that is a Lewis adduct. (b) What spectroscopic method could be used to identify the real structure? Explain.arrow_forwardThe structural formula for ethane is (a) What is the molecular formula for ethane?(b) What is its empirical formula?(c) Can we infer from this drawing that the H—C—H bond angles are 90 degrees?arrow_forward
- Make three-dimensional drawings of the following molecules, and predict whether eachhas a dipole moment. If you expect a dipole moment, show its direction.(a) H2C꓿CH2(b) CHCl3(c) CH2Cl2(d) H2C꓿CCl2arrow_forward(a) Which dipole pair shown below has the lower energy? (b) If you double the distance between the dipole centers, by what factor does pair interaction change? Does it increase or decrease? (a) Parallel (b) Antiparallel Dipoles separated by a distance r, (a) parallel, and (b) antiparallel.arrow_forwardindicate the approximate values for the indicated bond angles in the following molecules. 1 (a) H 0-N=ö 2. Н 4 H. (b) H C-c=0 H H H (c) H-N-O–H . H. (d) H-C-C=N: H.arrow_forward
- Specify the local electron geometries about the atoms labelled a-d. Unshared electron pairs affect local geometry and are included in the structural formula. For each atom enter one of the following: tetrahedral, square planar, trigonal planar, trigonal pyramidal, or linear. (a) (b) (c) (d) H- H H-C H H H H H 05 > -Harrow_forwardPropylene, C3H6, is a gas that is used to form the importantpolymer called polypropylene. Its Lewis structure is given. (a) What is the total number of valence electrons in the propylenemolecule? (b) How many valence electrons are usedto make σ bonds in the molecule? (c) How many valenceelectrons are used to make π bonds in the molecule? (d) Howmany valence electrons remain in nonbonding pairs in themolecule? (e) What is the hybridization at each carbon atomin the molecule?arrow_forwardSelect a nonpolar molecule in which the dipole moment of polar covalent bonds cancels each other. Explain your reasoning. (a) H2O (b) NH3 (c) CCl4 (d) HClarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





