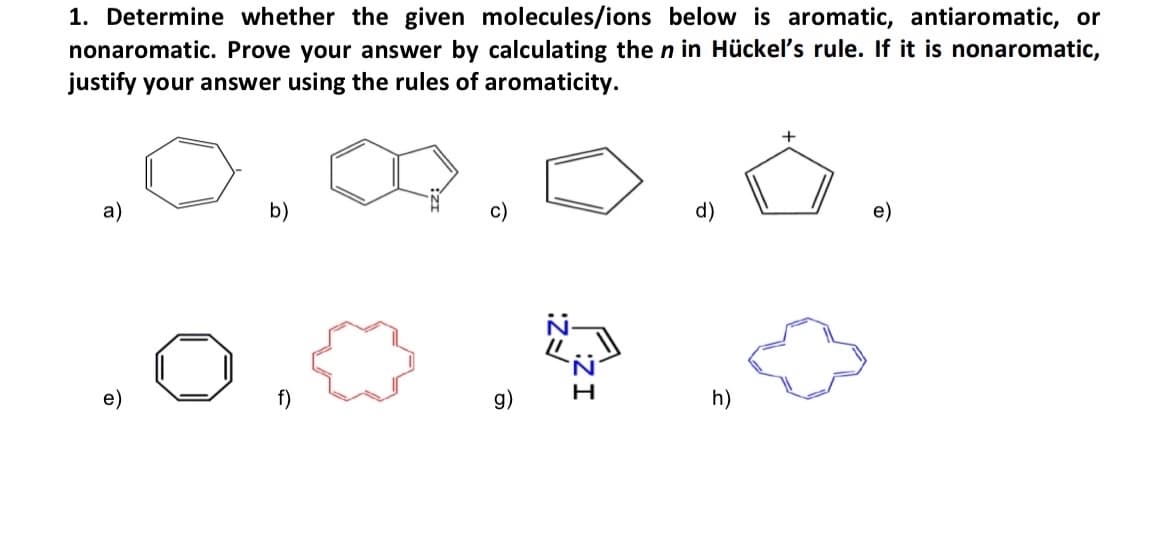
1. Determine whether the given molecules/ions below is aromatic, antiaromatic, or nonaromatic. Prove your answer by calculating the n in Hückel's rule. If it is nonaromatic, justify your answer using the rules of aromaticity. a) b) g) c) IZ: FZ: d) h) + e)
Q: The young and great expert Hand written solution is not allowed.
A:
Q: Uric acid is used for the diagnosis of what conditions? Question 31 options:…
A: The objective of the question is to identify the medical conditions that can be diagnosed using uric…
Q: ៣ AA Learning E4 CH 16 17 and 18 prod03-cnow-owl.cengagenow.com Learning Learning Learning × Online…
A: Step 1: Step 2: Step 3: Step 4:
Q: High serum total protein with high levels of both albumin and globulins is usually seen in…
A: In Waldenström's macroglobulinemia:1. Malignant B-lymphocytes proliferate and overproduce monoclonal…
Q: in the reaction 2SO(g) -- 2SO2(g), Kc= 4.0 x 10 -2 at 300 C. if the starting amount of SO2 was .5 M.…
A: The objective of the question is to find the equilibrium concentrations of all species in the given…
Q: How many grams of solid ammonium bromide should be added to 1.50 L of a 0.0879 M ammonia solution to…
A: The objective of this question is to determine the amount of solid ammonium bromide that needs to be…
Q: In carbohydrate metabolism, the hexose monophosphate shunt is used for: Question 67 options:…
A: The objective of the question is to understand the role of the hexose monophosphate shunt (also…
Q: Draw the arrow-pushing mechanism of the following reaction: OH HO H+ Draw the arrow-pushing…
A:
Q: help question 10
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Calculate the pH of solution of the following weak acid. Use Table 14.2 and Appendix H in your…
A: Step 1:Amphiprotic ion is a specie that can accept as well as donate the H+ ion. It means it can act…
Q: × Incorrect. Identify the major product(s) for the following reaction. Select all that apply. If the…
A: In bromination of alkyl in presence of hv, bromine atom will atach to secondary carbon as at…
Q: 9. For a spherical harmonic given by (a) What is the value of 1? (b)What is the value of my ? 3 Y…
A: Step 1:Ans:In spherical harmonic case the value of l and m decide in simply way we say that (1) The…
Q: An air-gap parallel plate capacitor whose plates have an area of A=0,38m² are separated by a…
A: Step 1:Step 2:
Q: The lone pairs of which heteroatoms(s) contribute to the aromaticity of the following molecule? N $
A: Step 1:The heteroatoms present in the given ring structure are:Oxygen and nitrogen. Nitrogen…
Q: Question 1: Aromaticity a. Draw a fully labelled MO energy diagram for each (match clearly). b.…
A: Step 1: The given molecule have the following IUPAC name: • Pyrrol-3-one • 1-Methyl-1H−borepine…
Q: Question 31 Predict the FINAL (?) product for each of the following reaction or synthetic chain:…
A: Step 1: Step 2:Mechanism Step 3: Step 4:
Q: The scientific report about Nitration of methyl benzoate experiment
A: ### Introduction to Electrophilic Aromatic SubstitutionElectrophilic aromatic substitution (EAS) is…
Q: None
A:
Q: What is the selenide ion concentration [Se2-] in a 0.100 M H2Se solution that has the stepwise…
A: Step 1:To find the concentration of the selenide ion ([Se²⁻]) in the solution, we need to consider…
Q: The mechanical properties of a metal may be improved by incorporating fine particles of its oxide.…
A: Given data:Emetal=50GPa moduli of elasticity of the metalEoxide=380GPa moduli of elasticity of the…
Q: The following data was obtained by measuring the volume of a trapped sample of gas at different…
A: Temperature T is plotted along x axis and volume V is plotted along y axis . The graph is shown in…
Q: יוון UIPAC name of this compound?
A: The IUPAC nomenclature of organic compounds follows this naming format:Writing the name of organic…
Q: Describe how you could achieve a higher degree of separation for the bands of food dyes on a TLC…
A: The objective of the question is to understand how to achieve a higher degree of separation for the…
Q: Consider three hypothetical solids:AX,AX2 and AX3 (each X forms X). Each of these solids has the…
A: Solution : Solubility is defined as the maximum amount of solute dissolved in an aqueous…
Q: 5(a) The Joule-Thomson experiment (JTE) is an example of an isenthalpic process i) Define an…
A: The objective of the first part of the question is to define an isenthalpic process, show that the…
Q: Give the mechanism for the reaction - draw it out
A:
Q: Draw the products and label them as major or minor.
A: See image.
Q: the spectra is of a drug either cannabinoid, opiate, MDMA etc, please explain the spectra and what…
A: The upper mass chromatogram speaking to the mass window 183-187 Da appears the specified item to be…
Q: None
A: Step 1:To determine the torsional strain in a planar cyclohexane molecule, we need to consider the…
Q: Problem 34 of 50 Submit Draw the product that could be formed when 1,3-butadiene reacts with…
A: Step 1:Mechanism Diels-Alder Reaction:It's a pericyclic reaction, meaning all bond breaking and…
Q: 30. The following data were collected for a certain reaction of the type: A → products Time, min…
A:
Q: Part A Consider the reaction A+2B C whose rate at 25 °C was measured using three different sets of…
A: Step 1: Step 2: Step 3: Step 4:
Q: Reactivity. Explain the regioselectivity and stereochemistry observed in each of the following…
A:
Q: Calculate creatinine clearance from the following results.urine creatinine=75 mg/dLurine volume=850…
A: The objective of this question is to calculate the creatinine clearance rate, which is a measure of…
Q: provide A and B both answer with explanation
A:
Q: - Aktiv Chemistry ← → C M Gmail b Answered: Draw the major pro × | + app.aktiv.com ☑ ੩ ॥ YouTube…
A: Thank you.
Q: Synthesis. Supply the missing reagents required to accomplish each of the following syntheses. Be…
A:
Q: Draw the major product of this aldol condensation reaction. Ignore inorganic byproducts. + H a H Q…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Which of the following aqueous solutions are good buffer systems? (Select all that apply.) 0.20 M…
A: The objective of this question is to identify which of the given aqueous solutions are good buffer…
Q: None
A: Step 1: Step 2:Left oxygen indicated by red outlined oxygen, and it have two lone pairs and two…
Q: Suppose all the hydrogen atoms in this molecule are replaced by chlorine atoms: དཡིག་བྱ། །ད Draw a…
A: Thank you.
Q: The electrophilic addition of HBr to 3-cyclohexylbut-1-ene gives a mixture of two constitutional…
A: [1,2]-h shift takes place
Q: (20 pts) Alcohol A undergoes a remarkable acid-catalyzed conversion to B. Me Me Me HO Me I Me S Me…
A: Step 1: Step 2: Step 3: Step 4:
Q: Depending on the method you are using, the method for measuring lactate dehydrogenase either…
A: The question is asking about the biggest factor that influences the direction of the reaction in the…
Q: write the ionic equation for 2NaCl + ZnS
A: ZnCl2(aq) + Na2S(aq) → 2 NaCl(aq) + ZnS(s) Total Ionic Equation: [Note: When ions appear on both…
Q: Draw the product of the reaction shown < below. Use a dash or wedge bond to indicate the…
A:
Q: :$;$;$;:$:&;&;&&;&
A:
Q: Question 35 Predict the FINAL (?) product for each of the following reaction or synthetic chain:…
A:
Q: O O H3O+ OH H2O
A: The given equation is a chemical reaction involving oxygen (O), hydronium ion (H3O+), and hydroxide…
Q: Living chain polymerization of monomer X follows the first-order kinetics. The corresponding…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Explain how to construct the molecular orbitals of a conjugated cyclic system similar tobenzene and cyclobutadiene. Use the polygon rule to draw the energy diagram, and fillin the electrons to show whether a given compound or ion is aromatic or antiaromatic.What is the Huckel rule of aromaticity. Explain and give an example. short and clear answer pleaseOChem Help... Label the following as aromatic, antiaromatic or nonaromatic. Next, show how Huckel's rule is applied in each case. See attached photo.
- Predict whether the cyclic system is aromatic , anti-aromatic, or non-aromatic by counting electrons and explain why ? Also circle all the basic nitrogen’s wherever possible . Show all lone pairs which are involved in cyclic delocalization?11.) Using Frost's circle, please determine whether the following molecule is aromatic, antiaromatic or non-aromatic (Please provide detailed explanation with diagram).Are the following molecules aromatic, antiaromatic, or non-aromatic? Why?
- Consider the following compound, which has been synthesized and characterized:(a) Assuming this molecule is entirely conjugated, do you expect it to be aromatic, antiaromatic, or nonaromatic?(b) Why was this molecule synthesized with three tert-butyl substituents? Why not makethe unsubstituted compound and study it instead?(c) Do you expect the nitrogen atom to be basic? Explain. Why doesn’t nitrogen’s lonepair overlap with the double bonds to give a total of six electrons in the pi system?(d) At room temperature, the proton NMR spectrum shows only two singlets of ratio 1:2. The smaller signal remainsunchanged at all temperatures. As the temperature is lowered to -110 °C, the larger signal broadens and separatesinto two new singlets, one on either side of the original chemical shift. At -110 °C, the spectrum consists of threeseparate singlets of areas 1:1:1. Explain what these NMR data indicate about the bonding in this molecule. How doesyour conclusion based on the NMR data agree with…Circle any of the following which you think are aromatic systems. For the ones that are aromatic systems, give the "n" (Huckel number.) It's possible tk have more than one. Thank you!Using the Frost Circle method to outline the molecular orbitals of cyclobutadiene. and identify whether it is aromatic, antiaromatic or non-aromatic. Explain. (Note: Show the outline of MO of cyclobutadiene using the Frost circle method)
- (a) Draw an energy diagram for the cyclopropenyl MOs. (The polygon rule is helpful.) Label each MO as bonding, nonbonding, or antibonding, and add the nonbonding line. Notice that it goes through the approximate average of the MOs.(b) Add electrons to your energy diagram to show the configuration of the cyclopropenyl cation and the cyclopropenyl anion. Which is aromatic and which is antiaromatic?organic chemistry Diels Alder.. ( Explain well all point of question with proper step by step type Answer. ).Determine whether the following molecules are aromatic (A), non-aromatic (NA), or anti-aromatic (AA), and show the use of a Frost circle in determing aromaticity. You may assume planarity.

