
Chemistry In Context
9th Edition
ISBN: 9781259638145
Author: Fahlman, Bradley D., Purvis-roberts, Kathleen, Kirk, John S., Bentley, Anne K., Daubenmire, Patrick L., ELLIS, Jamie P., Mury, Michael T., American Chemical Society
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9.6, Problem 9.15YT
Skill Building Esters and Polyesters
You have seen that terephthalic acid and ethylene glycol can react. Now consider ethanoic acid (acetic acid) and ethanol (ethyl alcohol):
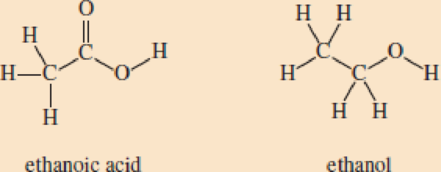
- a. Show how this
carboxylic acid and alcohol can react to form an ester. Hint: Remember a water molecule is formed as a product.- b. Could ethanoic acid and ethanol react to form a polyester? Explain your reasoning.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
M
0
K
.....
P
NH *
please answer letter b. Please help. Tysm❤️B. Choose one type of solid (metallic crystals, ionic crystals, molecular crystals, and covalent network crystals) and analyse explain in two sentences why it has these properties (strength or hardness, melting point, electrical conductibility, heat conductibility, and brittleness). Example: Metallic crystals have high melting point because a large amount of energy is needed to melt the crystal since the forces of attraction to be broke are numerous and extend throughout the crystal.
Using two monomers only (reactants). Each one of these two monomers should have two functional groups of Carboxyl on the right side and Hydroxyl on the left side attached to them. Hint: The total number of carbon atoms for each one of these two monomers, including the one for the carboxyl, must be four (4) only.
a) Draw the proper polymer structure of the products.
b) Name the products.
c) State the type of polymerization.
Chapter 9 Solutions
Chemistry In Context
Ch. 9.1 - Scientific Practices Tennis Anyone? Examine this...Ch. 9.3 - Prob. 9.2YTCh. 9.3 - Prob. 9.3YTCh. 9.4 - Prob. 9.4YTCh. 9.4 - Prob. 9.5YTCh. 9.4 - Prob. 9.6YTCh. 9.4 - Prob. 9.7YTCh. 9.4 - Prob. 9.8YTCh. 9.4 - Prob. 9.9YTCh. 9.5 - Prob. 9.10YT
Ch. 9.5 - Skill Building Benzene and Phenyl The difference...Ch. 9.5 - Prob. 9.13YTCh. 9.5 - Skill Building Polystyrene Possibilities Show the...Ch. 9.6 - Skill Building Esters and Polyesters You have seen...Ch. 9.6 - Prob. 9.16YTCh. 9.7 - Skill Building Kevlar Kevlar is a polyamide used...Ch. 9.8 - Prob. 9.20YTCh. 9.8 - Your Turn 9.22 Skill Building Burning a Plastic...Ch. 9.8 - Your Turn 9.23 Scientific Practices Landfill...Ch. 9.9 - Examine the values in Table 9.4 from the American...Ch. 9.9 - Prob. 9.25YTCh. 9.9 - Prob. 9.26YTCh. 9.9 - Prob. 9.28YTCh. 9.10 - Skill Building The Chemistry of PLA We dont show...Ch. 9.11 - Your Turn 9.31 Scientific Practices Glass or...Ch. 9.11 - Prob. 9.32YTCh. 9.11 - Skill Building Meet DEHP DEHP belongs to a common...Ch. 9 - Prob. 1QCh. 9 - Prob. 2QCh. 9 - Prob. 3QCh. 9 - Prob. 4QCh. 9 - Prob. 5QCh. 9 - Prob. 6QCh. 9 - Prob. 7QCh. 9 - Prob. 8QCh. 9 - Prob. 9QCh. 9 - Prob. 10QCh. 9 - Prob. 11QCh. 9 - Prob. 12QCh. 9 - Prob. 13QCh. 9 - Prob. 14QCh. 9 - Prob. 15QCh. 9 - Prob. 16QCh. 9 - Prob. 17QCh. 9 - Prob. 18QCh. 9 - Prob. 19QCh. 9 - Prob. 20QCh. 9 - Prob. 21QCh. 9 - Prob. 22QCh. 9 - Prob. 23QCh. 9 - Prob. 24QCh. 9 - Prob. 25QCh. 9 - Prob. 26QCh. 9 - Prob. 27QCh. 9 - Prob. 28QCh. 9 - Prob. 29QCh. 9 - Prob. 30QCh. 9 - Prob. 31QCh. 9 - Prob. 32QCh. 9 - Prob. 33QCh. 9 - Prob. 34QCh. 9 - Prob. 35QCh. 9 - Prob. 36QCh. 9 - Prob. 37QCh. 9 - Prob. 38QCh. 9 - Prob. 39QCh. 9 - Prob. 40QCh. 9 - Prob. 41QCh. 9 - Prob. 42QCh. 9 - Prob. 43QCh. 9 - Prob. 44QCh. 9 - Prob. 45QCh. 9 - Prob. 46QCh. 9 - Prob. 47QCh. 9 - Prob. 48QCh. 9 - Prob. 49QCh. 9 - Prob. 50QCh. 9 - Prob. 51QCh. 9 - Prob. 52QCh. 9 - Prob. 53QCh. 9 - Prob. 54QCh. 9 - Prob. 55QCh. 9 - Prob. 56QCh. 9 - Prob. 57QCh. 9 - Prob. 58QCh. 9 - Prob. 59Q
Additional Science Textbook Solutions
Find more solutions based on key concepts
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living By Chemistry: First Edition Textbook
Write a Lewis formula for each of the following organic molecules: C2H3Cl (vinyl chloride: starting material fo...
Organic Chemistry - Standalone book
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
How could you separate a mixture of the following compounds? The reagents available to you are water, either, 1...
Organic Chemistry
Characterize each of the following structures as aromatic, nonaromatic, or antiaromatic:
Answer: _____
Organic Chemistry As a Second Language: Second Semester Topics
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 1. What is polymer? a. A large molecule made by twisted chains. b. A small molecule made of one atom. c. A large molecule made by human. d. A large molecule made by monomers.arrow_forwardIn the experiment of reaction of haloalkanes ( reaction with aqueous alkali ): An oily firm may appear on the water surface. Suggest what it is ( It is alcohol ) and how it got there? Explain it. Guess what is it and explain oil film questionarrow_forwardUsing two monomers only (reactants). Each one of these two monomers should have two functional groups of Carboxyl on the right side and Hydroxyl on the left side attached to them. Hint: The total number of carbon atoms for each one of these two monomers, including the one for the carboxyl, must be four (4) only. a) Draw the proper polymer structure of the products. b) Name the products.arrow_forward
- How would you do this I’m stuck on the structurearrow_forwardWhich is more elastic, the springs or the rubber band? Why? Discuss in your lab report. What is the yield point of the rubber band? What it represent? Discuss in your lab report. Why are the shapes of the graphs for the springs and rubber band different? What does it imply? Discuss in your lab report.arrow_forward{rovide a short and concise explanation: a.Recognize the difference between crystalline and non-crystalline confectionery. b. What exactly is the logic behind the word "interfering" agents in the confectionery industry? Give at least two examples of interfering with crystallization and explain how each one works. c. Explain why sugar is important in meal preparation.arrow_forward
- 15. Network polymer structure is. O A. Branching. B. Liner. C. Cross-link. 16. Strong bonding in organic chemistry is. O A. Covalent bonding. OB. Hydrogen bonding. C. Van der Waals. Is are cured into permanent shape is Cannot be re- 17. melted. A. Thermoplastics B. Thermosetting Plasticsarrow_forwardPart A: The graphic shows an organic molecule and highlights a functional group that is bonded to it. This functional group is an example of ______ group. Part B: select which Characteristic this functional group impacts on the molecule. A. Forms disulfide bonds B. Nonpolararrow_forward2.The force between methylamine and acetate will be greater in methanol than in water because ___________. * a.Methanol has a lower dielectric constant than water b.Methanol, but not water, is structurally similar to methylamine c.Acetate and methylamine are more soluble in methanol than in water d.Dipole-dipole interactions between acetate and methylamine are induced in methanol, but not in water 3.Within the aqueous environment of an animal cell, sugars are stored as polymers rather than monomers. If the sugars were stored as monomers instead of polymers, which of the following properties would be least affected? * a.pH b.Viscosity c.Freezing point d.Osmotic pressurearrow_forward
- Write TRUE if the statement is accurate and FALSE if the statement is flawed._________ 1. There are negligible, attractive, or repulsive forces between molecules._________ 2. The average kinetic energy of a molecule is constantChoose the letter of the correct answer.3. Jane can still pump air in the party balloon even though it is already inflated. What explain thisphenomenon?a. Balloons look better if its size is bigger.c. the air inside the balloon is hot.b. Balloons are made up of plastic. d. air molecules can be compressedarrow_forwardUser Guard™ What kind of functional group is hydrolyzed to form soap? A. Aldehyde B. Amine C. Acid chloride D. Ester Q Search ****** ******* ********* *** app.honorlock.com 99+arrow_forwardIdentification Type 1. Agroup of three numbers that indicate the orientation of a plane or set of parallel planes of atoms in a crystal is called what? 2. A long chain giant organic molecules are assembled from many smaller molecules called monomers or mers. It comes from the Greek word poly meaning “many" and meros meaning "part". 3. It is the experimental science of determining the arrangement of atoms in solids. 4. List the classifications of Polymers based on composition. 5. List classification of Polymers based on physical property related to healing 6. What is a type of rock that contains minerals with important elements including metals? 7. What is the difference between Primary metals and Secondary metals? 8. Give an example of natural Polymer. 9. Give an example of Synthetic Polymer. Matching Type 1. Match the terms given in Column I with the compounds given in Column II. Column I Column II (1) Acid rain (ii) Photochemical smog (iii) Combination with haemoglobin (iv) Depletion…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY