
Concept explainers
Rank the
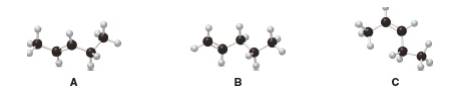
Interpretation: The given alkenes shown in the ball-and-sticks models are to be ranked in order of increasing stability.
Concept introduction: Stability of alkenes is governed by Zaitsev rule. More substituted alkenes are more stable.
Answer to Problem 24P
The stability order of given alkenes is
Explanation of Solution
According to Zaitsev rule the more substituted or more alkylated alkene is more stable which makes the order of stability as tetra-substituted > tri-substituted > di-substituted > mono-substituted.Trans alkenes are more stable than cis alkenes as there substitutions are attached far from each other which makes them less sterically hindered. In the given question A is trans-disubstitued alkene, B is mono-substituted alkene while C is cis-disubstitued alkene.
Explain how you got molecular structure from the given ball and stick model.
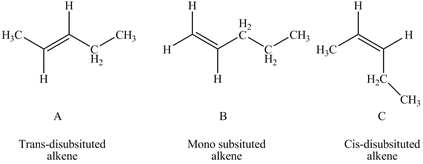
Figure 1
So, A and C are more stable than B.A is more stable than C as A is trans isomer while C is cis isomer. Therefore, stability order of given alkenes is
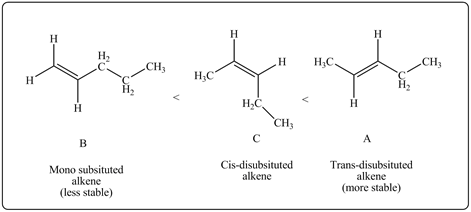
Figure 2
The stability order of given alkenes is–
Want to see more full solutions like this?
Chapter 8 Solutions
Organic Chemistry (6th Edition)
Additional Science Textbook Solutions
General, Organic, & Biological Chemistry
Introduction to Chemistry
Essential Organic Chemistry (3rd Edition)
Chemistry: The Central Science (13th Edition)
Chemistry In Context
Chemistry by OpenStax (2015-05-04)
- Match each alkene to its heat of hydrogenation. Alkenes: 3-methylbut-1-ene, 2-methylbut-1-ene, 2-methylbut-2-ene ΔH° (hydrogenation) kJ/mol: –119, –127, –112arrow_forwardDraw the structure of each alkene of molecular formula C7H14 that has a tetrasubstituted double bondarrow_forwardRank the alkenes below from most stable to least stable.arrow_forward
- Match each alkene to its heat of hydrogenation.Alkenes: 3-methylbut-1-ene, 2-methylbut-1-ene, 2-methylbut-2-ene?H° (hydrogenation) kJ/mol: –119, –127, –112arrow_forwardA is a toxin produced by the poisonous seaweed Chlorodesmis fastigiata. (a) Label each alkene that exhibits stereoisomerism as E or Z. (b) Draw a stereoisomer of A that has all Z double bonds.arrow_forwardDehydration of 1,2,2-trimethylcyclohexanol with H2SO4 affords 1-tert-butylcyclopentene as a minor product. (a) Draw a stepwise mechanism that shows how this alkene is formed. (b) Draw other alkenes formed in this dehydration. At least one must contain a ve-membered ring.arrow_forward
- A is a toxin produced by the poisonous seaweed Chlorodesmis fastigiata. (a) Label each alkene that exhibits stereoisomerism as E or Z.arrow_forward4) Which of the following alkene is the most stable? A) B) D)arrow_forwardClassify the carbocations as 1º, 2º, or 3º, and rank the carbocations in each group in order of increasing stability.arrow_forward
- Rank the alkenes shown below based on stability, should be indicated as (most stable (left) to least stable right): i.e.: A > B > C > Darrow_forwardDraw the constitutional isomer formed when the attached alkenes are treated with each set of reagents: [1] H2O, H2SO4; or [2] BH3 followed by H2O2, −OH.arrow_forwardDraw the alkene that would react with the reagent given to account for the product formed. ? + + H₂O **** H₂S04 • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. CH3 CH3 CHCCH3 | | OH CH3 +1arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





