
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 4.13B, Problem 4.20P
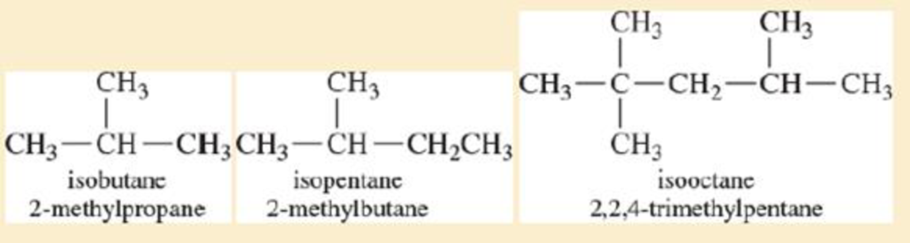
Use the bond-dissociation enthalpies in Tabte4-2 (page 167) to calculate the heats of readier for the two possible first propagation steps in the chlorination of isobutene. Use this information to draw a reaction-energy diagram like Figure4-8, comparing the activation energies for formation of the two radicals.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A reaction profile (not to scale!) for the reaction
03 + NO-
is shown below:
→02 + NO2
E (kJ)
11
O3 + NO
200
O2 + NO2
Reaction Coordinate
Which of the following are true?
Choose all that apply.
OThe reaction is exothermic.
The energy of the products is lower than the energy of the reactants.
O If the energy of the activated complex were increased, Ea would increase.
AE is positive.
Draw and label the enegry diagram for the reaction with the following: E(reactants) = 1000KJ, E(a)= 40KJ, delta H = -20KJ
Is this reaction endo/exothermic?
What is the energy of the products?
What is the activation energy?
sing energy kJ
7. In the reaction coordinate diagram, label the
boxes with the appropriate terms. Compare and
contrast the uncatalyzed and catalyzed reactions.
-Ea for uncatalysed reaction
-Activation energies for the
steps of a catalysed reaction
Ea3
'Ea1
tants
nroducts
Chapter 4 Solutions
Organic Chemistry (9th Edition)
Ch. 4.3A - Draw Lewis structures for the following free...Ch. 4.3B - a. Write the propagation steps leading to the...Ch. 4.3C - Prob. 4.3PCh. 4.3C - Prob. 4.4PCh. 4.4 - The following reaction has a value of G =...Ch. 4.4 - Under base-catalyzed conditions two molecules of...Ch. 4.5B - When ethene is mixed with hydrogen in the presence...Ch. 4.5B - For each reaction, estimate whether S for the...Ch. 4.7 - a. Propose a mechanism for the free radical...Ch. 4.7 - a. Using bond-dissociation enthalpies from...
Ch. 4.8 - The reaction of tert-butyl chloride with methanol...Ch. 4.8 - Under certain conditions, the bromination of...Ch. 4.8 - When a small piece of plat num is added to a...Ch. 4.10 - Prob. 4.14PCh. 4.10 - Prob. 4.15PCh. 4.12 - The bromination of methane proceeds through the...Ch. 4.12 - a. Using me BDEs in Table4-2 (page 167 ), compute...Ch. 4.13A - What would be the product ratio in the...Ch. 4.13A - Classify each hydrogen atom in the following...Ch. 4.13B - Use the bond-dissociation enthalpies in Tabte4-2...Ch. 4.13B - Prob. 4.21PCh. 4.13B - Prob. 4.22PCh. 4.14 - a. Compute the heats of reaction for abstraction...Ch. 4.14 - 2,3-Dimethylbutane reacts with bromine in the...Ch. 4.14 - Prob. 4.25PCh. 4.15 - Prob. 4.26PCh. 4.15 - Prob. 4.27PCh. 4.16A - Prob. 4.28PCh. 4.16A - Prob. 4.29PCh. 4.16B - Prob. 4.30PCh. 4.16C - Prob. 4.31PCh. 4.16C - Acetonitrile (CH3C N) is deprotonated by very...Ch. 4.16D - Prob. 4.33PCh. 4 - The following reaction is a common synthesis used...Ch. 4 - Consider the following reaction-energy diagram. a....Ch. 4 - Draw a reaction-energy diagram for a one-step...Ch. 4 - Draw a reaction-energy diagram for a two-step...Ch. 4 - Prob. 4.38SPCh. 4 - Treatment of tert-butyl alcohol with concentrated...Ch. 4 - Label each hydrogen atom in the following...Ch. 4 - Prob. 4.41SPCh. 4 - Prob. 4.42SPCh. 4 - Prob. 4.43SPCh. 4 - Prob. 4.44SPCh. 4 - Prob. 4.45SPCh. 4 - Prob. 4.46SPCh. 4 - For each compound, predict the major product of...Ch. 4 - When exactly 1 mole of methane is mixed with...Ch. 4 - Prob. 4.49SPCh. 4 - Prob. 4.50SPCh. 4 - Prob. 4.51SPCh. 4 - When dichloromethane is treated with strong NaOH,...Ch. 4 - Prob. 4.53SPCh. 4 - When a small amount of iodine is added to a...Ch. 4 - Prob. 4.55SPCh. 4 - When healthy, Earths stratosphere contains a low...Ch. 4 - Prob. 4.57SPCh. 4 - lodination of alkanes using iodine (I2) is usually...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Consider the following statements: In general, the rate of a chemical reaction increases a bit at first because it takes a while for the reaction to get warmed up. After that, however, the rate of the reaction decreases because its rate is dependent on the concentrations of the reactants, and these are decreasing. Indicate everything that is correct in these statements, and indicate everything that is incorrect. Correct the incorrect statements and explain.arrow_forwardWhich statement accurately describes a reaction with very high heat energy? O few molecular collisions occurring quickly O many molecular collisions occurring slowly few molecular collisions occurring slowly many molecular collisions occurring quickly https://prodpcx-cdn-learnosityassessmentviewer.emssvc.connexus.com/index.html# Type here to search 立arrow_forwardThe 3rd order reversible reaction given below has an activation energy of the forward reaction equal to -337.177 cal/mol. 2NO(g) + 02(g) ↔ 2N02(g) If the specific rates of the reverse reaction, expressed in moles/L-s, are 73.9 at 600K and 307 at 645K, calculate the heat of reaction at constant volume? A) None of these B) -24,673.08 cal/mol C) -3371.77 cal/mol D) -23,998.72 cal/mol E) -24,335.90 cal/molarrow_forward
- Second Year Second Semester 2018-2019 First Month Exam School of Chemistry Organic Chemistry QI/ Consider the following reaction: "OCICH,, Br (CH,,COH ン しい」 What happens to the reaction rate guh each of the following changes? is (1) The concentration of OC(CH is decreased.(2) The base is changed to "OH. (3) The halide is changed to CH;CH:CH2CH,CH(Br)CH3. (4) The leaving group is changed to I.arrow_forwardHeterogeneous catalysts are more advantageous to use than homogeneous catalysts because they can interact with the reactants more intimately, resulting in faster reactions. Select one: O True O False The rate of a reaction remains constant from beginning to end. Select one: O True O False For a reaction with a rate constant of 0.533 M-1 s1 at 600 K, the overall reaction order is 2. Select one: O True O False The equilibrium constant (Kea) is greater than 1 for an exothermic reaction that involves a positive entropy change. Select one: O True O Falsearrow_forward4. Xylene can be produced from toluene as written schematically: CHAPTER 1 The Basics of Reaction Kinetics for Chemical Reaction Engineering 47 CH3 CH3 CH3 CH AG= 14.85 kJ mol" toluene ortho-xylene benzene CH3 CH3 CH, AG'= 10.42 kJ mol CH3 toluene meta-xylene benzene CH3 CH3 CH3 AG'= 15.06 kJ mol toluene benzene CH3 para-xylene The values of AG° were determined at 700 K. What is the equilibrium composition (including all xylene isomers) at 700 K and 1.0 atm pressure? Propose a method to manufacture para-xylene without producing significant amounts of either ortho- or meta-xylene.arrow_forward
- 2. Select the energy profile templates below (A or B) that correctly represents the enthalpy change calculated for the decomposition of ammonium nitrate and label it showing the reactants, products, change in enthalpy for the reaction, AHRXn and the activation energy, Ea. Potential Energy Reactants A Reaction Progress->> Products ΔΗ Potential Energy Reactants ΔΗ B Products Reaction Progress-> 3. Use your understanding of reaction kinetics and collision theory to explain why an increase in temperature causes an increase in reaction rate. 4. Evaluate the changes in physical state that occur during the decomposition of ammonium nitrate and predict the sign on AS for the reaction. 5. Use the standard entropies, S° to determine the AS (in J K-¹ mol¹ ) for the reaction. Does the sign on AS match your prediction?arrow_forwardLabel the reaction profiles below as exothermic or endothermic and use them to calculate the activation energy of the forward and reverse reactions and the value of AH for the reaction. This is a reaction of profile of an 250 exothermic/endothermic reaction 200 PE 150 (kJ) Eaf = ...... 100 Ea, = 50 AH = Reaction pathway 100 This is a reaction of profile of an 80 exothermic/endothermic reaction 60 PE (kJ) A+B 40 Ear = C+D 20 Ear = ΔΗ Progress of the reactionarrow_forwardA reaction profile (not to scale!) for the reaction 03+02 02 is shown below: E (kJ) 03 + O 392 Which of the following are true? Choose all that apply. 19 Reaction Coordinate O2 + O2 AE is positive. The energy of the products is lower than the energy of the reactants. The reaction is exothermic. If the energy of the activated complex were decreased, Ea would decrease. Previous Next>arrow_forward
- Consider the following reaction mechanism. Step 1: 2X+Y W+ 2 Z fast Step 2: Z U+V slow Step 3: V+Z 2W + Y fast Which of the following is/are the intermediate/s in the given reaction mechanism? O V only O V and Y O Y only O Z and V At 298 K, if the bond dissociation of Cl2g) to Clo) is +57 kJ/mol while the electron affinity of Clg) to form Cl (9) is -349 kJ/mol, then the Gibbs energy of the reaction, Cl2g) + 2e 2 Clg), is negative. Select one: O True O Falsearrow_forwardConsider the mechanism shown here: 2 NO N202 N202 + H2 → N20 + H20 (slow step) Nz© + H, → Nz + H20 Write the balanced, overall reaction for this mechanism. You do not need to subscript the #atoms in each molecule, use an "equals sign" for the arrow.arrow_forwardGiven reaction rate data for: H;O2 + 31- + 2H+ → k + 2H;O Trial [H2O2] M [1'] M [H*] M Rate (M/s) 0.00050 0.010 0.020 0.010 0.010 0.010 0.020 0.010 1.15 x 106 2.30 x 106 2.30 x 106 1.15 x 106 1 2 0.00050 0.00050 4 0.010 0.00100 Write the rate law for the reaction.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Kinetics: Initial Rates and Integrated Rate Laws; Author: Professor Dave Explains;https://www.youtube.com/watch?v=wYqQCojggyM;License: Standard YouTube License, CC-BY