
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16.8C, Problem 16.12P
Explain why each compound or ion should be aromatic, antiaromatic, or nonaromatic.
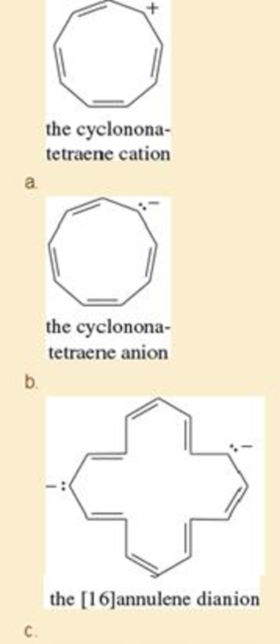
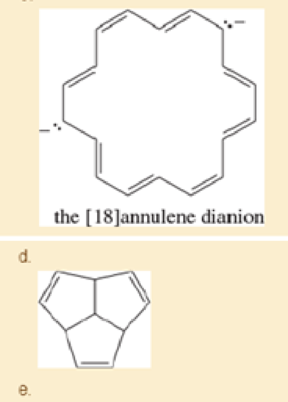
- f. the [20]annulene dication
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The purine heterocycle occurs commonly in the structure of DNA.
a. How is each N atom hybridized?
b. In what type of orbital does each lone pair on a N atom reside?
c. How many a electrons does purine contain?
d. Why is purine aromatic?
purine
Label each compound as aromatic, antiaromatic, or not aromatic. Assume all completely conjugated rings are planar.
Å
a.
b.
C.
d.
Zolpidem (trade name Ambien) promotes the rapid onset of sleep, making it a widely prescribed drug for treating insomnia.
a.In what type of orbital does the lone pair on each N atom in the heterocycle reside?
b. Explain why the bicyclic ring system that contains both N atoms is aromatic.
c.Draw all reasonable resonance structures for the bicyclic ring system.
Chapter 16 Solutions
Organic Chemistry (9th Edition)
Ch. 16.2 - Prob. 16.1PCh. 16.2 - Prob. 16.2PCh. 16.2 - a. Draw the resonance forms of benzene,...Ch. 16.2 - Show the product of the Diels-Alder dimerization...Ch. 16.4 - Prob. 16.5PCh. 16.6 - Make a model of cyclooctatetraene in the tub...Ch. 16.6 - Prob. 16.7PCh. 16.6 - Prob. 16.8PCh. 16.7 - Prob. 16.9PCh. 16.8A - a. Draw the molecular orbitals for the...
Ch. 16.8A - Repeat Problem16-10 for the cyclopentadienyl ions....Ch. 16.8C - Explain why each compound or ion should be...Ch. 16.8C - The following hydrocarbon has an unusually large...Ch. 16.8C - Prob. 16.14PCh. 16.8C - Prob. 16.15PCh. 16.9B - Prob. 16.16PCh. 16.9C - Show which of the nitrogen atoms in purine are...Ch. 16.9C - The proton NMR spectrum of 2-pyridone gives the...Ch. 16.9D - Prob. 16.19PCh. 16.9D - Prob. 16.20PCh. 16.10 - Prob. 16.21PCh. 16.12 - Ciprofloxacin is a member of the fluoroquinolone...Ch. 16.13 - Draw and name all the chlorinated benzenes having...Ch. 16.13 - Name the following compounds:Ch. 16.15 - The UV spectrum of 1-phenylprop-2-en-1-ol shows an...Ch. 16 - Prob. 16.26SPCh. 16 - Name the following compounds:Ch. 16 - Draw and name all the methyl, dimethyl, and...Ch. 16 - Four pairs of compounds are shown. In each pair,...Ch. 16 - One of the following hydrocarbons is much more...Ch. 16 - In Kekuls time cyclohexane was unknown, and there...Ch. 16 - Prob. 16.32SPCh. 16 - Azulene is a deep-blue hydrocarbon with resonance...Ch. 16 - Prob. 16.34SPCh. 16 - Prob. 16.35SPCh. 16 - Prob. 16.36SPCh. 16 - Prob. 16.37SPCh. 16 - Prob. 16.38SPCh. 16 - Prob. 16.39SPCh. 16 - Biphenyl has the following structure. a. Is...Ch. 16 - Anions of hydrocarbons are rare, and dianions of...Ch. 16 - How would you convert the following compounds to...Ch. 16 - Prob. 16.43SPCh. 16 - Prob. 16.44SPCh. 16 - A student found an old bottle labeled thymol on...Ch. 16 - Prob. 16.46SPCh. 16 - Prob. 16.47SPCh. 16 - Prob. 16.48SPCh. 16 - The proton NMR chemical shifts of the hydrogens in...Ch. 16 - Prob. 16.50SPCh. 16 - NMR has been used to probe many molecular...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In electrophilic aromatic substitution, a group attached to the aromatic ring does not affect ring reactivity. A.RightB.Falsearrow_forwarda.How many π electrons does C contain? b.How many π electrons are delocalized in the ring? c.Explain why C is aromatic.arrow_forwardExplain why each compound is aromatic, anti aromatic or non aromatic? Explainarrow_forward
- Which of the foll owing lone pairs of purine participate in its aromaticity? A N. N.B N. Carrow_forwardWhich of the following molecules is anti-aromatic? A. Cyclopentadienyl cation B. cycloheptatriene C. silopentadiene D. cycloheptatrienyl cation E. cyclopentadienyl anionarrow_forward2) Which one of the following molecules is anti-aromatic? a. b. C. d. e.arrow_forward
- 1. Is the molecule below aromatic? If so, circle the atoms that are part of the aromaticity. Also indicate the orbital that each lone pair occupies. .N. H2Narrow_forwardplease explain why they are aromatic.?arrow_forwardWhich compound is not aromatic? A. only A and D B. A, B and C C. only B D. only Aarrow_forward
- Use Huckel’s rule to identify if its aromatic or not. Draw the resonance structure of the ff. compounds. note: I need the answer immediately. I will send a good rate right away as well.arrow_forwardWhich of the following compounds is aromatic? A. only A and B B. only A and C C. only B and C D.only Barrow_forwardWhich molecules below are anti- aromatic? D A. Structure D Structure B Structure A Structure C B. C. D.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY