Q: At a certain temperature, the equilibrium constant K for the following reaction is 1.3: CO(g) +…
A: Equillibrium constant :For a chemical reaction, the equilibrium constant can be defined as the…
Q: Draw the products of the transformation shown by the fishhook notation. Include all hydrogen atoms…
A: In the given question we have to draw the product by homolytic cleavage of butyl radical to form…
Q: Which of the following IMFs could form between an aldehyde and a molecule of H₂O? [Select all that…
A: Intermolecular forces are the attractive or repulsive forces that exist between molecules. These…
Q: Why the extended Pi(π) conjugated are suitable for Fluorescence properties? Please shortly answer at…
A: Introduction Fluorescence is a phenomenon where a substance absorbs light of a particular wavelength…
Q: Rank the following elements according to their ionization energy. element magnesium sodium beryllium…
A: It is based on the concept of periodic property and it's trends. Here we are required to arrange the…
Q: Give the IUPAC name for each compound. Part 1 of 3
A: we have to name the given structure
Q: : Br-CH₂-CH3 + 30-H
A: A chemical reaction
Q: CH3 HO H H CI CI A) diastereomers B) enantiomers C) same compound D) unrelated compounds OH The…
A: Answer- A) Diastereomers
Q: Ph,PCHP, THF
A: This reaction is an example of Wittig Reaction.
Q: Which among the followung has the strongest IMFA? a. C4H10 b. CH3CI C. d. 12 HF Which of the…
A: The question is based on the concept of chemical bonding. we need to identify the nature of…
Q: ← Curved arrows are used to illustrate the flow of electrons. Using the provided starting and…
A:
Q: Please explain the answer OH 1. TsCl, pyr 2. OH, NaOH
A:
Q: The following sequence shows a suggested mechanism for a reaction: Step 1: NO (g) + Cl₂ (g) - →…
A: This question belong to chemical kinetics. Rate of complex reaction. Only slow reaction will decide…
Q: Re-order each list in the table below, if necessary, so that the atoms or ions in it are listed in…
A: It is based on the concept of periodic property and trends Here we are required to arrange the…
Q: An unknown substance X (AHvap = 22.1 kJ/mol) has a vapor pressure of 76.0 mmHg at 300 Determine the…
A:
Q: Consider the following reaction where Kc 7.00 x 10 at 673 K. NH₁I(s) ⇒ NH3 (9) + HI(g) A reaction…
A: The question is based on the concept of chemical equilibrium. We calculate the value of reaction…
Q: A bottle is labeled only "Compound B (C6H₁2)" and contains a colorless liquid. You have been called…
A: The given molecular formula of the compound is C6H12. The degree of unsaturation of the compound is…
Q: [NO2] (M) 2.5 2 1.5 1 0.5 0 0 10 20 30 40 50 Time (s) 60 70 nation to answer 80 90 100
A: The explanation is given below-
Q: Give the IUPAC name for each compound.
A: The IUPAC name of the compound can be written on the basis of the number of carbon atoms in the main…
Q: Question 12 Dr. A dilutes 35.0 mL of 2.4 M H2SO4 to a new volume of 850.0 mL. What is the new…
A: 12) given, Before dilution: Concentration of H2SO4 solution (M1) = 2.4 M Volume of H2SO4 solution…
Q: Provide the correct systematic name for the compound shown here.
A:
Q: Indicate, by letter(s), the position(s) on the ring at which substitution occurs when the aromatic…
A: In presence of Lewis acid like FeBr3, Br2 undergo bromination reaction. Here FeBr3 activates Br2…
Q: Copy the above Lewis structure CT use for Teflon and annotate it to indicate the overall molarity…
A:
Q: Please give me some paragraph explanation regarding my thesis title below and its uses in the…
A: A carbon monoxide and carbon dioxide abatement and notification system is a system designed to…
Q: Consider hydrogen atoms with electrons randomly distributed in the n= 1, 2, 3 and 4 orbits.…
A: • Here is given that the electrons for the hydrogen atoms are randomly distributed is the given…
Q: Draw the condensed structures of the following compounds.. a. 2-bromo-3-methylpentane b.…
A: In condensed structural formula each carbon was represented individually and repeating units are…
Q: Fill in the information missing from this table: subshell 4f 3s 5d Some electron subshells principal…
A: The given problem is based on the four types of quantum numbers.
Q: What do you think is the pH of the solution at its equivalence point will be seven if we are…
A: is the pH of the solution at its equivalence point will be seven if we are titrating a strong acid…
Q: To what volume should you dilute 125 mL of an 8.00 M CuCl2 solution so that 50.0 mL of the diluted…
A: Here according to given conditions in question we are doing that:-
Q: The rate of a certain reaction is given by the following rate law: rate = k [NO] 2 [0₂] Use this…
A: Rate law expression: Let us understand the rate law expression by taking an example, X + Y ➝ Z The…
Q: WHAT IS THE pH and pOt of each solution given the following values of [0]'? WHICH SOLUTIONS ARE…
A:
Q: Give the IUPAC name for each compound. Part 1 of 2 X 5
A:
Q: HỘI (PHAN)
A:
Q: 1a. Briefly explain 3 advantages and disadvantages of using ¹H-NMR for the quantitative analysis of…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three sub-parts…
Q: The actual density of pennies made before 1982 is about 8.8 g/mL. The actual density of pennies made…
A: This answer provides suggestions on what could be done differently to obtain results closer to the…
Q: Use the reaction coordinate graph below to answer questions 11-13: 11. Which step is the rate…
A: Given is Graph between activation energy and co-ordinate of reaction . The reaction goes in many…
Q: Which would you predict to have the lower vapor pressure at a given temperature?
A: The vapor pressure of a substance depends on its intermolecular forces and molecular weight.…
Q: Which is an example of an Arrhenius acid/base A. NaOH b. HCI c. KOH d. All of the above
A:
Q: Write equations for the preparation of the following alkenes by the Wittig reaction. Start with any…
A: In witig reaction, a carbonyl compound reacts with triphenyl phosphonium ylide to produce an alkene…
Q: The rate constant k, for a reaction was measured at several temperatures and the slope of the ln(k)…
A: Arrhenius equation is lnk =-EaR1T+lnAk is rate constantEa is activation energyR is gas constantT is…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Drag To Pan
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: 5. The structure of anthocyanin, a naturally occurring compound that acts as a pigment in many…
A: Anthocyanidin gives red color at pH=3 (Acidic condition) Anthocyanidin gives purple color at pH=6-7…
Q: name the following compounds following the IUPAC nomenclature.
A: Rules for IUPAC nomenclature of ester: • The name of the ester depends on the alcohol and carboxylic…
Q: Macmillan Learning For the given generic reaction, the concentration of A and the amount of time…
A: l
Q: All of the following are intermediates in the following transformation EXCEPT: OCH3 Li, NH3 tBuOH…
A:
Q: Give the products for each of the following reactions. ~x.O (a) (b) (c) [ + H3COC- OCH 3 || -COCH3
A: The given reactions are examples of Diels alder reaction. A cyclohexene derivatives are the possible…
Q: Organic... art 2 of 3 X S
A: One question is Clea visible other one is not so i answer only the first one.
Q: Ethylene (C2H4) plays an important role in the ripening of certain fruits and vegetables. It is…
A:
Q: When the following molecular equation is balanced using the smallest possible integer coefficients,…
A:
Q: calculate the empirical formula of each compound with the following percent composition. 1. 79.8%C,…
A: Empirical formula of a chemical compound is the simplest whole number ratio of atoms present in that…

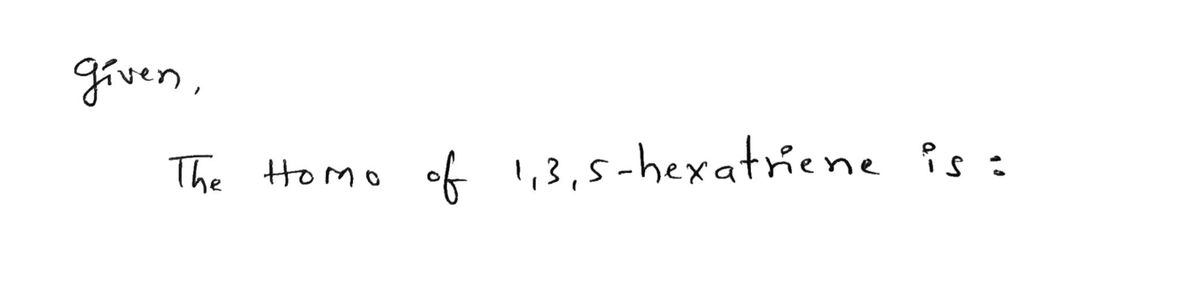
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Which structure (or structures) represents (or represent) the enantiomer of the molecule below? I OCH 3 &&&* IV I III and IV ||| || OCH3 OH OCH 3 & II || Br III OCH3 H3CO/I IVDraw a dash-wedge structure for (1R)-1-bromo-1,3,3-trimethylcyclohexane.How many degrees of unsaturation (double bond equivalents) does C4H10ClNO have?
- Rank the conformers of 1,2,4-trimethylcyclohexane in order of decreasing stability, putting the most stable first. II II IV Multiple Choice III > || > IV > | III > IV > || > | |> || > IV > II| |> || > I| > IVH CH, is molecule chiral or achiral? CH, H.Which of the following is the highest energy chair conformer of the most stable isomer of 1-ethyl-2,4-dimethylcyclohexane? A | B II C) III IV = || ||| > IN
- H₂N S=0 atom 1 CH3 ; atom 2 isoxazole ring Assuming the heterocyclic ring above to be planar: Give the number of electrons that are contributed to the pi system of the heterocyclic ring by: bextra, a COX-2 inhibitor, used in the treatment of arthritis ✓; atom 3 NA . Enter NA for atom 3 if the structure above has only two numbered atoms. How many electrons are in the pi system of the heterocyclic ring? (In the case of fused rings add all the pi electrons.) Is the heterocyclic ring with the numbered atoms aromatic?[• -- H (b) H,C 3-methylhexane CH, (c) B கூ CH, CH, HgCG; சீகூ H₂CH H 3-ethyl-1-methyloctane 3,3 diethyl pentane 5Å, Å,CH, W CH3 CH2 CH₂ W21 Su2... CH3 X -CH3 G b F E L cWhich of the following is the correct Newman projection for the following compound as viewed down the indicat conformation shown? CO II O III O IV OV CH3 CH3 CH31 CH3 CI || Br Br H CH3 CH3 ||| Br CH3 CH3 IV CI Br CH3
- CH3Li CH3MgBr (CH3)2CuLi CH3OH4.(a). Draw the structures of (i) cis-1,4-Dibromocyclohexane and (ii) trans-1,4- Dibromocyclohexane. 4(b). Draw the most stable conformers of (i) cis-1,4-Dibromocyclohexane and (ii) trans-1,4-Dibromocyclohexane. 4(c)Which is more stable? (i) cis-1,4-Dibromocyclohexane or (ii) trans-1,4- Dibromolcyclohexane. Explain the reason for your choice.Classify the pairs of molecules as not the same molecule, structural isomers, diastereomers, enantiomers or identical. Circle any molecules that are not achiral (a) (b) (c) Br... H CI H H Br H H3C Br H. CI Br H Br CI I I Br" "H H Br H CH3 Cr Br

