The degree of unsaturation, or index of hydrogen deficiency, is the number of pi bonds plus rings in a molecule. Specify the degree of unsaturation (index of hydrogen deficiency) of the following formulas: (a) C24H30 (b) C15H22 (c) C4H,Cl,N
Q: The CFSE for octahedral [CoCl]ª¯ is 18,000 cm1. The CFSE for tetrahedral [CoCl4]²¯ will be.. а.…
A: When the ligands are same in Octahedral and Tetrahedral then the CFSE of Octahedral and Tetrahedral…
Q: Hexasulfur (S6) is an allotrope of S that presents a chair conformation, similar to cyclohexane.…
A: By rotation arround 360°/n, at particular centre, if we get identical molecule, then we called…
Q: all hydrogens are homotopic for which of the following conditions -just structure A -just structure…
A: Stereochemistry is branch of chemistry in which we deal with three dimensional arrangement of atoms…
Q: Predict the structure of I(CF3)Cl2. Do you expect the CF3 group to be in an axial or equatorial…
A: Generally the groups which are more electronegative will occupy the axial position while depicting…
Q: 6. The structure shown below is amoxicillin, one of the most commonly prescribed penicillin-type…
A: As you have asked multiple question but we will answer both the parts.
Q: DHA is a fatty acid derived from fish oil and an abundant fatty acid in vertebrate brains.…
A: One molecule of DHA undergoes ozonolysis to obtain following products.
Q: N2O4 is planar. Show that it possesses three planes of symmetry.
A: We would draw structure of N2O4 and show plane which pass symmetrically .
Q: For anion NO2-, (a) predict the stereochemistry. Justify your prediction with VSEPR theory and by…
A: VSEPR theory predict the geometry , shape and hybridisation of molecules by the number of electron…
Q: The degree of unsaturation, or index of hydrogen deficiency, is the number of pi bonds plus rings in…
A: The degree of unsaturation, or index of hydrogen deficiency, can be calculated as: DOU =(Number of C…
Q: Why are the C2H7 molecular formula impossible?
A:
Q: b. Explain in molecular orbital terms the changes in H-H inter-nuclear distance that occurs as the…
A: Bond Order is the number of bonding electron pairs between any two atoms.
Q: Which of the following molecules are chiral?
A: Chirality is the property shown by optically active molecules. Also molecules which are non…
Q: Propose structures for molecules that meet the following descriptions: a. Contains two…
A: Alkane C are SP3 hybridized alkene C atoms are SP2 hybridized while alkyne C are SP hybridized .…
Q: 4. Stereochemistry (a) Draw the possible stereoisomers of [Co(NH3)2(H2O)2(CN)2] and assign their…
A:
Q: Construct the molecular-orbital energy level diagramsof (a) ethene and (b) ethyne on the basis that…
A:
Q: Explain the following: (a) The peroxide ion, O22 - , has a longerbond length than the superoxide…
A: (1) The peroxide ion is O22- (consists of two oxygen atoms with -2 charge). Bond order of…
Q: About the special type of hybridization involved in complexation (all are correct except Select one:…
A: In different type of hybridization, different type of structures of complexes or coordination…
Q: Vhich of the following will exhibit cis-trans isomerism? e°: [Ar]4s²3d% Edº: [Kr]5s²4d10 tº:…
A: The question is based on the concept of isomerism. We have to identify cis-trans isomerism in the…
Q: Propose structures for molecules that meet the following descriptions: (a) Contains two…
A: Carbon atom attached to four other atoms has sp3 hybridization getting a tetrahedral arrangement.…
Q: Why are these two conformational isomers? I was under the impression that in molecules with only…
A: The compounds given are,
Q: Calculate the degree of unsaturation (DU) for the molecule with the formula of C5H12N2O.
A:
Q: What is the index of hydrogen deficiency for the following molecule? S. S. NH2 H2N O 1 O 2 O 3
A:
Q: Predict the mtiplicity for the hydrogen at poSimon C, consiler the'3 arfd 4 bortd' coupling Singlet…
A:
Q: Explain the following: (a) The peroxide ion, O22 - , has a longerbond length than the superoxide…
A: Peroxide ion has two atoms of oxygen with charge of -2. The bond order could be calculated as,…
Q: The degree of unsaturation, or index of hydrogen deficiency, is the number of pi bonds plus rings in…
A:
Q: Identify the s-trans conformation of (3Z,5Z)–4,5-dimethyldeca–3,5–diene.
A: E,Z nomenclature is given to double bond in which each olefinic (double bond) has different groups.…
Q: What is the degree of unsaturation of C8H10ClNO? a. 4 b. 5 c. 6 d. 7
A:
Q: Propose structures for molecules that meet the following descriptions: Contains two sp2 hybridized…
A: Given : there are 2 sp2 and 2 sp3 hybridised carbons in the molecule.
Q: Aromatic compounds must have a p orbital on every atom in the ring. Define this ?
A: If the compound is aromatic, there are some following criteria for aromaticity which are as follows:…
Q: The degree of unsaturation, or index of hydrogen deficiency, is the number of pi bonds plus rings in…
A: We have,
Q: 1. Calculate the degrees of unsaturation for the molecular formula C6H12O and draw two possible…
A: degree of unsaturation is equal to the total number of π-bonds and rings present in an organic…
Q: Draw a molecular orbital picture for the LUMO of 1,3-Butadiene
A:
Q: how many possible consitutional isomers are there for C2H4Cl2?
A: Isomer are pair of compound which have same molecular formula but different structural formula…
Q: Rank the orbitals of 3-bromopropene in the order of increasing energies. a. 1s TC b. 1s c. 1s d. 1s…
A: The energy level of the molecule starts from 1s being the least in energy and then gradually sigma…
Q: Which of the following structure represents the three dimensional line structure of the Newman…
A:
Q: Which one of the following represents the lowest energy pi-bonding molecular orbital of…
A: in 1,3 butadiene 2 pi bonds are.present. 4 pi electrons
Q: 4. Using a Frost circle, approximate the relative energy level of the a molecular orbitals for…
A: Using frost circle approximates the relative energy level of the pi molecular orbitals for following…
Q: N. Pyrazine, C4H4N2, is used in the synthesis of vitamins and drugs. The molecule can be hought of…
A: Since you have asked multiple subparts question, we will solve first three subparts for you. If you…
Q: Methotrexate, a drug that inhibits the metabolism of folic acid, is used in the treatment of a…
A: (a) The hybridization of nitrogen atoms marked by star is sp2 hybridized and the other nitrogen…
Q: The degree of unsaturation, or index of hydrogen deficiency, is the number of pi bonds plus rings in…
A: We have to calculate the IHD.
Q: Compound C show below is the starting material used in a chemical reaction. a) Name the two…
A: The specific group in structure when attached to any structure gives specific functional…
Q: Determine the symmetry of the IR active C-H stretching modes for cyclohexane chair conformation
A:
Q: calculate the degrees of unsaturation in the molecule C22H32Cl3FN2O5
A:
Q: Give a molecular orbital description for the 1,3,5-heptatriene:
A: Molecular orbital diagram:- It shows how atomic orbitals constitute together to form molecular…
Q: Calculate the number of degrees of unsaturation in a compound ofmolecular formula C4H6, and propose…
A: Unsaturated compounds are those which contain double bonds or triple bonds. Unsaturated compounds…
Q: a) Draw the open structure of BH2ÇI. (At. No of B: 5) b) Predict the symmetry species of IR…
A:
Q: Cisplatin (cis-diamminedichloroplatinum(II)) is the first platinum-based antitumor agent, and it is…
A:
Q: One isomer of retinal is converted to a second isomer by the absorption of a photon: CH3 H CH3 H H;C…
A: a). Chemical structure of the retinal: In the structure of 11-cis (in 4-cis) retinal, whenever a…
Q: Calculate the degree of unsaturation (DU) for a molecule with the formula of C5H12.
A:
Q: 2. (a) Define and give one example of a geometry that is coordinatively unsaturated. (b) Define and…
A: COORDINATIVELY UNSATURATED GEOMETRY: A compound which has metal central atoms that are bonded to…
Q: 3.- Based on the molecular orbitals of hydrogen, and the hybridized orbitals of single, double and…
A: Explanation: In 1st structure C—H bond is rounded in blue. The 'p' orbital of sp3 carbon and 's'…
Q: The degree of unsaturation, or index of hydrogen deficiency, is the number of pi bonds plus rings in…
A: Given compounds: (a) C9H12 (b) C14H18 (c) C10H8N2 We have to find the degree of unsaturation of…
Q: 1.The [CoCl4]2- anion is intensely blue colored and [CoCl6]4- is a lightly colored pink. explain…
A: The color of the coordination complexes arises due to the transition of electrons between different…
Q: When ß-carotene is oxidised in vivo, it breaks in half and forms two molecules of retinal (vitamin…
A: Introduction : In an atom each shell has its own energy level , the shell nearest to the nucleus…
Q: The degree of unsaturation, or index of hydrogen deficiency, is the number of pi bonds plus rings in…
A: Deree of unsaturation is also known as index of hydrogen deficiency , double bond equivalents, or…
Q: (a) Draw the orbital picture for :C=0: showing the explicit overlap of the contributing AOs. (b) How…
A: Molecular orbitals are formed by the Linear Combination of Atomic Orbitals (LCAO). Number of…
Q: Name the following Cd(C2H3O2)4
A: Let the oxidation state of cadmium in the molecule be x. The charge on single acetate ion is -1. The…
Q: Calculate the degree of unsaturation (DU) for a molecule with the formula of C3H10.
A:

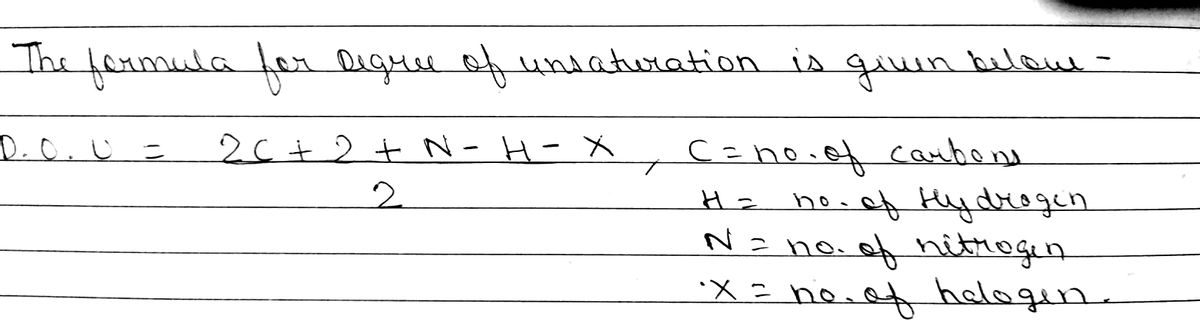
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Arrange the following compounds in increasing order of their property as indicated :(i) CH3COCH3, C6H5COCH3, CH3CHO(reactivity towards nucleophilic addition reaction)(ii) Cl—CH2—COOH, F—CH2—COOH, CH3—COOH (acidic character)(a) (CH3)3CBr classify the compound as a methyl, primary, secondary, or tertiary halide.Biphenyl has the following structure.(a) Is biphenyl a (fused) polynuclear aromatic hydrocarbon?(b) How many pi electrons are there in the two aromatic rings of biphenyl? How does this number compare with that for naphthalene?(c) The heat of hydrogenation for biphenyl is about 418 kJ>mol (100 kcal>mol). Calculate theresonance energy of biphenyl.(d) Compare the resonance energy of biphenyl with that of naphthalene and with that of two benzene rings. Explain thedifference in the resonance energies of naphthalene and biphenyl.
- (a) Describe the molecular geometry expected for 1,2,3-butatriene (H2C=C=C=CH2). (b) Two stereoisomers are expected for 2,3,4-hexatriene (CH3CH=C=C=CHCH3). What should be the relationship between these two stereoisomers?(a) A phenyl group has the molecular formula CH- and is represented by the symbol Bn. True False (b) Para substituents occupy adjacent carbons on a benzene ring. (c) True False (e) 3-Bromobenzoic acid can be separated into cis and trans isomers. True O False (d) 1-Phenylcyclohexene is a planar molecule. True False Benzene, naphthalene, and phenanthrene are polynuclear aromatic hydrocarbons (PAHs). TrueCompound A exhibits a peak in its 1H NMR spectrum at 7.6 ppm, indicating that it is aromatic. (a) How are the carbon atoms of the triple bonds hybridized? (b) In what type of orbitals are the π electrons of the triple bonds contained? (c) How many π electrons are delocalized around the ring in A?
- (b) Complete the following reactions : (i) D H3C CH3 H H كما .NO2 B | Bra/Dioxane .COCH3 AIdentify which of the statements is/are correct. (i) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O. (j) The molecular formula of the smallest carboxylic acid is C2H4O2.Write the reagent or draw structures of the starting material or organic product(s) in the following reactions. If more than one product is formed, identify the major product where possible. (a) (b) HO OH OH H2SO4 ? Cl₂ ? FeCl3
- (a) (CH3)3CBr give the common name (if possible).Draw a three-dimensional structure for each compound, and star all asymmetric carbon atoms. Draw the mirror image for each structure, and state whether you have drawn a pair of enantiomers or just the same molecule twice. Build molecular models of any of these examples that seem difficult to you. (a) ОН (b) (c) NH, ОН СH—CH—СООН | pentan-2-ol pentan-3-ol alanine (d) 1-bromo-2-methylbutane (e) chlorocyclohexane (f) cis-1,2-dichlorocyclobutane (h) (i) H CH; "H H. H CH, H(a) Draw the structure of the following :(i) p-Methylbenzaldehyde (ii) 4-Methylpent-3-en-2-one(b) Give chemical tests to distinguish between the following pairs of compounds :(i) Benzoic acid and Ethyl benzoate, (ii) Benzaldehyde and Acetophenone.(iii) Phenol and Benzoic acid.

