Q: Br starting material steps product NH2
A: Given reaction can be done by two stepsfirst step : nucleophilic substitution reactionsecond step :…
Q: A B H O Type your answers in all of the blanks and submit X₁ X N- 0 The following compound has both…
A: The objective of the question is to predict the site of reaction on a compound that has both an…
Q: Rank the 0.06 m aqueous solutions of monoprotic acids from the weakest acid to the strongest acid.…
A: Solution and their Ka values0.06 m acetylsalicylic acid (HC9H7O4) 3.31×10-4 0.06 m hydroazoic…
Q: If 2.82 g of the triglyceride below is saponified with excess NaOH, calculate the theoretical yield…
A: Mass of triglyceride = 2.82 whereR=15 NaOH = excessCalculate the theoretical yield of soap.
Q: Br H Cl H Enter the correct IUPAC nomenclature. cis-1-bromo
A: For naming a compound according to IUPAC, there are some rules. They are -Find the longest chain or…
Q: right? Is the ship between the parent structure on the left and the structure on the
A: Organic compounds can be defined as compounds that contain carbon and hydrogen atoms. We have been…
Q: Name these organic compounds: Н HIC | H | - - H | | | structure - H H H - | - | - H HIC I H H-C C-H…
A: IAnswer:IUPAC is the International Union of Pure and Applied Chemistry which has given set of rules…
Q: Fill in the left side of this equilibrium constant equation for the reaction of phosphoric acid…
A: ka = {[H3O+][H2PO4-]}/[H3PO4]. Explanation:When the phosphoric acid reacts with the water it…
Q: An electrode was constructed in lab that measures equilibrium silver ion concentrations in solutions…
A: The concentration of Ag+ ions will decrease by the same amount as the concentration of Cl- ions…
Q: Predict the major products of the following organic reaction: + ? Some important Notes: • Draw the…
A: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a conjugated diene and a…
Q: The solubility of Ce(IO₃)₃ (cerium (III) iodate) in 0.20 M KIO₃ (potassium iodate) is 4.4 x 10-8 M.…
A:
Q: Predict the major products of the following organic reaction: + NC Δ ? Some important Notes: • Draw…
A: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a conjugated diene and a…
Q: 21. Propose a synthesis of the desired product from the given starting material(s).
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: organic starting materials NaOH B-hydroxycarbonyl HO
A: Given,The reaction is:
Q: Calculate the pH for 0.45 M NaOCI. The K₂ value for HOCI is 3.0 x 10-8. K₁ for OCI = [OH-] = pH =
A:
Q: Propane (C3H8) is a gas at room temperature, but it exists as a liquid under pressure in a propane…
A: The objective of the question is to write a balanced chemical equation for the reaction of propane…
Q: What would be the base and alcohol used in the following Claisen condensation reaction? Draw the…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: 6) What is the product of the following reaction? CH A) CHOCH LOCH OCH CH₂OCH сно
A: It is an example of nucleophilic substitution reactionnucleophile can attack at 2nd or 4th position.…
Q: How many atoms from the following molecule will be detected in a 1H NMR experiment? We are not…
A: In 1H-NMR all the hydrogen atoms will be detected. The number of hydrogen atoms detected in 1H-NMR…
Q: Consider the reaction below. Then select all that are true about this reaction. CH,CH₂ÖH H CH,CH,OH…
A: Answer:In any reaction, specie that loses its H+ ion is called Bronsted-Lowry acid and specie that…
Q: The alkene shown was synthesized via a Wittig reaction. In the labeled boxes, draw the alkyl bromide…
A: Witting reactionCarbonyl compounds react with ylide which gives olefines. Ylides can prepared by…
Q: Identify the geometry of each alkene as E, Z, or neither (non-stereoisomeric). HO X and Olis Z; Il…
A:
Q: Draw the major organic product formed in this reaction. HBr ROOR
A: Alkenes undergo free radical addition when treated with HBr in the presence of peroxide.This…
Q: ct Question 4 0 / 9 pts Sulfuric acid reacts with sodium hydroxide according to this reaction: H₂SO4…
A: The objective of the question is to calculate the molarity of the sulfuric acid (H2SO4) solution.…
Q: Complete the mechanism for the deprotonation of the α carbon.
A: The mechanism of a reaction is the step-by-step pathway by which the reactant is converted to the…
Q: Draw the structure of the major organic product(s) of the reaction. Хуа 1. LiAlH4, ether 2. H3O+
A: Lithium Aluminium Hydride (LAH) is versatile reducing agent used for the reduction of functional…
Q: Kainic acid, an amino acid isolated from red seaweed, is used by laboratory scientists to study…
A: The stability of the conjugate base is a key factor in acidity. Protons attached to atoms that allow…
Q: Machinery costs $1 million today and $100,000 per year to operate. It lasts for 8 years. What is the…
A: The objective of the question is to calculate the equivalent annual annuity of the machinery given…
Q: Consider the pair of reactions. Draw the organic products, then predict the type of substitution…
A: We have to draw the products of the given SN2 reactions.Explanation:Approach to solving the…
Q: Draw the appropriate Newman projection which leads to the major E2 ELIMINATION product. Hint: Review…
A:
Q: Can the molecule on the right-hand side of this organic reaction be made in good yield from no more…
A: Given is organic compound.Given compound is fused ring compound. As we can see one six member ring…
Q: These are synthesis questions. You need to show how the starting material can be converted into the…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Calculate the osmolarity of 2.00 M BaBr2
A: The objective of this question is to calculate the osmolarity of a 2.00 M BaBr2 solution. Osmolarity…
Q: You need to make an aqueous solution of 0.173 M zinc fluoride for an experiment in lab, using a 500…
A: The objective of the question is to calculate the amount of solid zinc fluoride needed to prepare a…
Q: Questions 1-3 refer to these structures: CH3CH2 O-CH2CH3 CH3CH2CH2CH2CH2CH2OH D A B HOC-CHỊCH…
A: Given,The structures:
Q: 5 1.5 points Potassium Chlorate decomposes into potassium chloride and oxygen gas. How many grams of…
A: 5) Weight of potassium chlorate = 8.40 g.We have to find out the amount of oxygen(O2) produced when…
Q: A solution was prepared in the lab when equal volumes of 3.00 × 10⁻⁴ M calcium chloride and 2.50 ×…
A: I have given an answer and explanation for the question that you have asked with my knowledge and…
Q: Draw the product that could be formed when 1,3-butadiene reacts with maleic anhydride. Include any…
A: Find out the product
Q: 3. Copy the five sets of data (volume NaOH and pH) from titration #3 into the table on the next…
A: The objective of the question is to calculate the pKa of an unknown weak acid using the data from a…
Q: 10.51 Draw all stereoisomers formed in each reaction. a. Br₂ b. Cl₂ ن H₂O NBS d. DMSO, H₂O H₂O H2SO4
A: As images cannot be uploaded to this section, see below for the answers:Explanation:The answers are…
Q: Calculate the cell potential for the galvanic cell in which the given reaction occurs at 25 °C,…
A: In the given question->[Sn2+] = 0.0670 M[Co3+] = 0.0365 M[Sn4+] = 0.00661 M [Co2+] = 0.00991MThe…
Q: How many signals would you expect to appear in an 1H NMR spectrum of the following compound? 3 2 O O…
A: In 1H NMR, number of different type of hydrogen represent the number of signals.It depends on the…
Q: Calculate the molar solubility of MX (Ksp=2.65×10−36) in each liquid or solution. pure water 0.25M…
A:
Q: Mass of CaCO3 standard (g) Volume of CaCO3 used (mL) Volume of EDTA Trial 0.15099 34.90 Trial 2…
A: The % mass of a molecule in a given mixture can be calculated using the following equation-
Q: Determine the pH of 0.231 M Ca(OH)2 (aq
A: Given:Concentration of Ca(OH)2 = 0.231 MWe have to determine the pH of the solution
Q: What is the angular momentum of a SF6 molecule when it is rotating with its minimum energy?
A: Angular momentum is defined as the property of a rotating object given by the product of moment of…
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A:
Q: x 2 G > : ☐ Draw the simplest curved arrow mechanism possible for the reaction shown below. You may…
A: Curved arrows indicate the flow of electrons. The tail of the curved arrow is placed at the source…
Q: An analytical chemist is titrating 165.1 mL of a 1.100M solution of acetic acid (HCH3CO2) with a…
A: The volume of acetic acid (HCH3CO2) = 165.1mL = 0.165LThe Molarity of acetic acid( HCH3CO2)…
Q: First, specify what you think is the sign of delta H and delta S (without calculating or looking at…
A: The reaction in which heat energy is released, and then the energy of products become less than that…
Draw the structure of the self-aldol addition product that would form from propanal. Don't forget to include lone pair electrons!
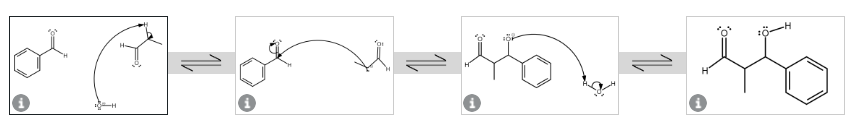
Step by step
Solved in 1 steps with 1 images




