Calculate the packing efficiency of the face-centered cubic unit cell. Use the labeling task to show your work, where V represents volume. Label the diagram by dragging the labels to the appropriate targets. Reset Help 4 atoms /3m 8. 2 atoms 16 T Vatoms 3/2 unit cell atom unit cell Vunit cell = 1 = (2) 68 Vatoms Vunit cell 52 %Valled X 100% X 100% 3 (2/2r)" 74 |(2/2r)* x 100% = 68 % 2 atoms 8r3 1 atom II
Calculate the packing efficiency of the face-centered cubic unit cell. Use the labeling task to show your work, where V represents volume. Label the diagram by dragging the labels to the appropriate targets. Reset Help 4 atoms /3m 8. 2 atoms 16 T Vatoms 3/2 unit cell atom unit cell Vunit cell = 1 = (2) 68 Vatoms Vunit cell 52 %Valled X 100% X 100% 3 (2/2r)" 74 |(2/2r)* x 100% = 68 % 2 atoms 8r3 1 atom II
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter11: Liquids And Solids
Section: Chapter Questions
Problem 11.87QE
Related questions
Concept explainers
Bond Parameters
Many factors decide the covalent bonding between atoms. Some of the bond parameters are bond angle, bond order, enthalpy, bond length, etc. These parameters decide what kind of bond will form in atoms. Hence it is crucial to understand these parameters in detail and understand how changing these parameters affects the kind of bonding or various characteristics.
Bond Dissociation Energy
The tendency of an atom to attract an electron is known as its electronegativity.
Question
Question in picture

Transcribed Image Text:Part A
Calculate the packing efficiency of the face-centered cubic unit cell. Use the labeling task to show your work, where V represents volume.
Label the diagram by dragging the labels to the appropriate targets.
Reset Help
4 atoms
V37
2 atoms
16
Vatoms
unit cell
unit cell
atom
3/2
Vunit cell
= 3 =
68
Vatoms
X 100% =
X 100%
52
%Vfilled =
Vunit cell
(2/2r)³
74
(2/2r)3
X 100% =
%
68
2 atoms
8r3
1 atom
Expert Solution
Step 1
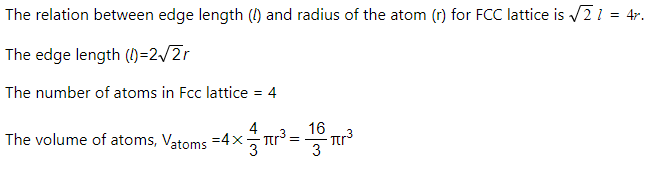
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning