Q: If I wanted to separate a mixture containing formic acid and naphthol that were dissolved in diethyl…
A: Both formic acid and naphthol are acidic in nature but pKa value of formic acid is 3.75 and pKa…
Q: Suggest a reasonable mechanism for the conversion of a nitrile (RCN) to the corresponding amide in…
A: Nitriles (RCN) can be converted to the corresponding amid in the presence of aqueous acid. Consider…
Q: Illustrate the Application of Enolate Alkylation: Tamoxifen Synthesis ?
A: Given reaction, Application of enolate alkylation: Tamoxifen Synthesis
Q: Show how Fischer esterification might be used to form the following esters. In each case,suggest a…
A: The Fishcer esterification is a process in which there is a acid catalyzed nucleophilic acyl…
Q: can some please draw and explain in detail the reaction mechanism BENZIMIDAZOLES from…
A: Following are the appropriate answers of the given questions.
Q: Apply retrosynthetic analysis and create a synthetic strategy for the synthesis of…
A: Retrosynthesis : it is the way how target molecule can be returned to the starting material. Forward…
Q: Why, in the last step of the mechanism for hydroxide-ion promoted hydrolysis of an amide, is the…
A: We know that the hydrolysis of an amide will yield a carboxylic acid and the base catalyst will…
Q: CIe II NO L-tyrosine F. ZEZ-
A:
Q: Explain what will happen to the (i) yield and (ii) purity, of aspirin if the following steps were…
A: The functional group is a key component associated with the molecule that transforms the molecule…
Q: Give the structures of the two substrates that are required to synthesize the following drug against…
A: The molecular structure of the required drug is provided. It can be synthesized by Knorr pyrrole…
Q: To synthesize m-ethylbenzenesulfonic acid, a student attempted the Friedel–Crafts alkylation of…
A: The given reaction of benzenesulfonic acid undergoing alkylation to form m- ethylbenzenesulfonic…
Q: Pivampicillin is a methylpivoxyl ester of ampicillin. It is prepared as a pro-drug to improve the…
A: Amphicillin is antibiotics used to treat for many infection . In ampicillin having carboxylic acid…
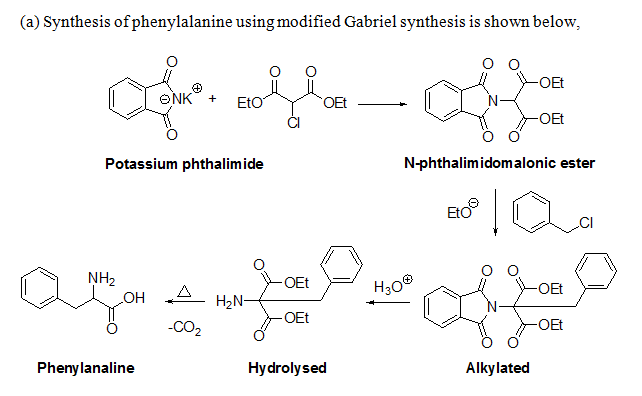
Q: Show how you will use Modified Gabriel’s Synthesis and Streckers’s Synthesis to prepare…
A: Modified Gaberial Synthesis : Potasium Phthalimide reaction with chloromalonic ester gives…
Q: Define the following terms and provide a specific example for each: a. Haloform reaction b. Phase…
A: Please find the attachment.
Q: How Sulfinpyrazone will be synthesized? Give full reaction
A: Sulfinpyrazone : Sulfinpyrazone is a phenylbutazone derivative. Direct effect of sulfinpyrazone was…
Q: Why is retrosynthesis seen as a very important approach in the synthesis of organic compounds,…
A: Retrosynthesis means destructing a complex organic compound to simple forms of starting material. It…
Q: Write the two acid-base equations that allow the separation of a mixture of trans-stilbene and N,…
A: Interpretation: To write the two acid-base equations that allow the separation of a mixture of…
Q: Make a retrosynthetic analysis on the synthesis of m-nitroacetanilide. Use the result to identify…
A: Retrosynthetic analysis is an analysis technique try to solve the problem for the synthesis of…
Q: Show how you would use a Strecker synthesis to make phenylalanine.
A: Strecker synthesis is a two step procedure that is used to synthesize alpha amino acids from…
Q: Please neatly show the mechanism and the balanced equation for the addition of NaBH4 to benzil in a…
A: To show the balanced equation and for reduction of benzil by NaBH4.
Q: 2. Draw the scheme of nifedipine synthesis (name all the products).
A: The drug can be defined as a medcation. They are used to treat and cure diseases. One of the…
Q: Show how you will use Strecker's synthesis to prepare phenylalanine in the laboratory
A: Strecker amino acid synthesis- This method is useful for preparation of amino acids. An aldehyde…
Q: 1. Answer all parts of this question. Ci epichlorohydrin propanolol (a) Provide a two-step…
A: Synthetic organic chemistry involves the conversion of the reactants into the appropriate products…
Q: Write the step by step mechanism with curved arrows of the hydrdysis of the amide in' this section.
A: Any organic group which has functional group CONH2 are amide
Q: Show how you will use modified Gabriel's synthesis to prepare phenylalanine in the laboratory
A: The preparation of phenylalanine using modified Gabriel's synthesis has to be shown.
Q: Provide a stepwise synthesis for benzyl chloride to benzoic acid. (including arrow-pushing…
A: In the above question, we have to provide a synthesis mechanism for conversion of benzyl chloride to…
Q: (b) With proper reasoning, provide the retrosynthesis and forward synthesis for the following…
A:
Q: b) Menthone is converted to a mixture of menthone and isomenthone on treatment with 90% Sulfuric…
A:
Q: 1. Discuss a disadvantage of Friedel - Crafts acylation reaction 2. Propose a mechanism for the…
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: Deduce a synthetic strategy for the synthesis of m-nitroacetanilide using retrosynthetic analysis.…
A:
Q: The reactions given in the synthesis scheme showing the synthesis of D, a phremone of the moth…
A:
Q: Explain why a much better yield of primary amine is obtained from the reaction of an alkyl halide…
A: Alkyl halide reacts with sodium azide to form alkyl azide. Here the formed alkyl azide is not…
Q: Show the synthesis of butylamine from ammonia in one step. Make sure to specify the stiochiometry of…
A: Alkyl halides undergo substitution reaction with ammonia to give ammonium salts because ammonia act…
Q: Suppose 1-bromo-4-methoxybenzene was added to the initial mixture ( C 4-ethyl aminobenzoate, B…
A: See the answer below.
Q: Using aniline to perform a Friedel-Crafts acylation. However, the reaction failed and the aluminum…
A: Friedel Crafts reaction is an organic coupling reaction involving electrophilic substitution at the…
Q: H S. NH2 H S- H3CO2C H3CO2C Nafcillin IZ
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: Suggest a synthesis of the following compound by using the acetoacetic ester synthesis. Show all…
A: Target molecule
Q: When o-vanillin and p-toluidine are mixed, the mixture turns to a bright orange powder as the imine…
A: We have find out the answer.
Q: d-caprolactone with 2 equivalents of ethylamine b. Use mechanism arrows to illustrate the reaction…
A:
Q: What reaction sequence will form dibenylamine from benzoic acid in high yield
A: The details solution for this reaction is provided below in attach image.
Q: Give evidence and prove that E2 reactions are predominantly anti-elimination
A: E2-Elimination: In this type of bimolecular elimination, Bond between carbon and hydrogen break with…
Q: Give the mechanism of the Julia olefination reaction with three examples.
A: Answer - Julia olefination - The Julia olefination is the chemical reaction used in organic…
Q: Grignard Reaction, 3-hexanol retrosynthesis explain two mtheds.
A:

Step by step
Solved in 3 steps with 3 images

- Show how you will useModified Gabriel’s Synthesis and Streckers’s Synthesis to prepare phenylalanine in the laboratory.tert-Butoxycarbonyl azide was developed as a reagent for peptide synthesis at OWL's home institution, the University of Massachusetts, by Prof. L.A. Carpino. It is prepared by treating tert-butoxycarbonyl chloride with sodium azide. Propose a structure for the initially-formed intermediate in this reaction.Suggest a test you will use to show that a given food substance contains protein. Show how you will use; A modified Gabriel's synthesis A Streckers' synthesis to prepare phenylalanine in the laboratory.
- Synthesize the compund (Amlodipine) via Hantzsch dihydropyridine synthesis.(D)The best route to synthesise aromatic primary amines is by reduction of the corresponding nitro compounds. Draw the reaction scheme for the preparation of p-toluidine.From the given structures which is(a) an acid halide? (b) a secondary amide (c) an aldehyde incapable of self-aldol consensation?
- (a) Vanillic acid is a phenolic compound found in vanilla and many other plant extracts. It is a flavouring and scent agent that produces a pleasant, creamy odour. Show the biosynthesis route for the formation vannillic acid from phenylalanine (Shikimate pathway). HO. NH2 Phenylalanine Vanillic acidFollowing is an outline of a synthesis of the bronchodilator carbuterol, a beta-2 adrenergic blocker with high selectivity for airway smooth muscle receptors. Q.Suggest a structural relationship between carbuterol and ephedrineDraw the expected product of the reaction of the following sugars with excess methyliodide and silver oxide.(a) a-d-fructofuranose (b) b-d-galactopyranose

